Abstract
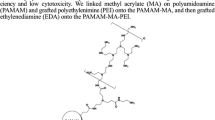
In this study, a new kind of antibiotic drug-polymer conjugate, the kanamycin-methyl acrylate-polyethylenimine (KMP) polymer, was synthesized and characterized as gene carrier. Previous reports have shown that the modification of low molecular weight polyethylenimine (LMW-PEI) improves the transfection efficiency and decreases the toxicity. Kanamycin can be hydrolyzed under an acidic environment owing to the glycosidic bond. In this physicochemical study, the properties of the kanamycin-based hybrid polymers conjugated with various LMW-PEIs, such as 0.6, 1.2, and 2kDa polyethylenimine (PEI), were investigated by 1H NMR, FT-IR, and their buffering capacity was assayed by acid-base titration method. Gel electrophoresis and picogreen assay were performed to evaluate the ability to bind with plasmid DNA. The polyplexes were nanosized with a net positive charge. The cytotoxicity and transfection efficiency were evaluated using human cervical carcinoma (HeLa) cells. KMPs exhibited lower toxicity than PEI 25 kDa, and higher transfection efficiency compared to the native LMW-PEIs.
Similar content being viewed by others
References
E. L. Scheller and P. H. Krebsbach, J. Dent. Res., 88, 585 (2009).
K. Bulaklak and C. A. Gersbach, Nat. Commun., 11, 5820 (2020).
E. Koutsilieri, A. Rethwilm and C. Scheller, J. Neural Transm. Suppl., 43 (2007).
D. Weissman and K. Kariko, Mol. Ther., 23, 1416 (2015).
K. Cornetta, M. Bonamino, J. Mahlangu, F. Mingozzi, S. Rangarajan and J. Rao, Mol. Ther., 30, 2122 (2022).
M. L. Hu, T. L. Edwards, F. O’Hare, D. G. Hickey, J. H. Wang, Z. Y. Liu and L. N. Ayton, Clin. Exp. Optom., 104, 444 (2021).
R. H. Pickler and C. L. Munro, J. Pediatr. Nurs., 10, 40 (1995).
S. K. Das, M. E. Menezes, S. Bhatia, X. Y. Wang, L. Emdad, D. Sarkar and P. B. Fisher, J. Cell. Physiol., 230(2), 259 (2015).
Y. Q. Wang, A. Asakawa, A. Inui and K. Kosai, Expert Opin. Biol. Ther., 10, 1405 (2010).
D. K. Chellappan, N. S. Sivam, K. X. Teoh, W. P. Leong, T. Z. Fui, K. Chooi, N. Khoo, F. J. Yi, J. Chellian, L. L. Cheng, R. Dahiya, G. Gupta, G. Singhvi, S. Nammi, P. M. Hansbro and K. Dua, Biomed. Pharmacother., 108, 1188 (2018).
V. Sudhakar and R. M. Richardson, Neurotherapeutics, 16, 166 (2019).
R. Miller, C. Hinderer, C. Dyer, J. Johansson, P. Bell and J. M. Wilson, Hum. Gene Ther., 30, A91 (2019).
N. T. Dastjerd, A. Valibeik, S. R. Monfared, G. Goodarzi, M. M. Sarabi, F. Hajabdollahi, M. Maniati, J. Amri and S. S. Tehrani, J. Cell. Biochem., 40, 28 (2022).
M. Ramamoorth and A. Narvekar, J. Clin. Diagnostic Res., 9, GE01 (2015).
S. Akhtar, Y. Omidi, A. Hollins, R. Drayton and M. Benboubetra, J. Gene Med., 6, S5 (2004).
H. Yin, R. L. Kanasty, A. A. Eltoukhy, A. J. Vegas, J. R. Dorkin and D. G. Anderson, Nat. Rev. Genet., 15, 541 (2014).
M. S. Huh, E. J. Lee, H. Koo, J. Y. Yhee, K. S. Oh, S. Son, S. Lee, S. H. Kim, I. C. Kwon and K. Kim, Top. Curr. Chem., 375(2), 31 (2017).
S. Salatin and M. Jelvehgari, Iran. J. Pharm. Sci., 23, 84 (2017).
S. Barua, J. Ramos, T. Potta, D. Taylor, H. C. Huang, G. Montanez and K. Rege, Comb. Chem. High Throughput Screen., 14, 908 (2011).
A. R. Teagle, J. C. Birchall and R. Hargest, Skin Pharmacol. Physiol., 29, 119 (2016).
J. L. Chen, H. Wang, J. Q. Gao, H. L. Chen and W. Q. Liang, Int. J. Pharm., 343, 255 (2007).
C. R. Dass and P. F. M. Choong, J. Control. Release, 113, 155 (2006).
A. Zakeri, M. A. J. Kouhbanani, N. Beheshtkhoo, V. Beigi, S. M. Mousavi, S. A. R. Hashemi, A. K. Zade, A. M. Amani, A. Savardashtaki, E. Mirzaei, S. Jahandideh and A. Movahedpour, J. Exp. Nanosci., 9(1), 1488497 (2018).
R. V. Benjaminsen, M. A. Mattebjerg, J. R. Henriksen, S. M. Moghimi and T. L. Andresen, Mol. Ther., 21, 149 (2013).
I. M. S. Degors, C. Wang, Z. U. Rehman and I. S. Zuhorn, Acc. Chem. Res., 52, 1750 (2019).
C. K. Chen, P. K. Huang, W. C. Law, C. H. Chu, N. T. Chen and L. W. Lo, Int. J. Nanomed., 15, 2131 (2020).
Q. Liu, R. C. Su, W. J. Yi and Z. G. Zhao, Molecules, 4, 22 (2017).
M. A. Gosselin, W. J. Guo and R. J. Lee, Bioconjug. Chem., 12, 989 (2001).
T.-H. Kim, H. Choi, G. S. Yu, J. Lee and J. S. Choi, Macromol. Res., 21, 1097 (2013).
Y. H. Lee, H. I. Park and J. S. Choi, Carbohydr. Polym., 137, 669 (2016).
K. M. Krause, A. W. Serio, T. R. Kane and L. E. Connolly, Cold Spring Harb. Perspect. Med. CSH., 6(6), a027029 (2016).
Y. Zhang, Z. Zhou, X. Zhu and M. Chen, Eur. J. Pharm. Sci., 105, 33 (2017).
M. Sainlos, P. Belmont, J. P. Vigneron, P. Lehn and J. M. Lehn, Eur. J. Org. Chem., 2003, 2764 (2003).
M. S. Chen, J. L. Wu, L. Z. Zhou, C. Y. Jin, C. L. Tu, B. S. Zhu, F. A. Wu, Q. Zhu, X. Y. Zhu and D. Y. Yan, Polym. Chem., 2, 2674 (2011).
R. Augustine, A. Hasan, R. Primavera, R. J. Wilson, A. S. Thakor and B. D. Kevadiya, Mater. Today Commun., 25, 101692 (2020).
M. X. Wang, B. Wu, S. N. Shah, P. J. Lu and Q. L. Lu, Mol. Ther. Nucleic Acids., 16, 663 (2019).
S. Godeshala, R. Nitiyanandan, B. Thompson, S. Goklany, D. R. Nielsen and K. Rege, Bioeng. Transl. Med., 1, 220 (2016).
C. K. Chen, P. K. Huang, W. C. Law, C. H. Chu, N. T. Chen and L. W. Lo, Int. J. Nanomed., 15, 2131 (2020).
Acknowledgements
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2020R1I1A3070280).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supporting Information
Additional information as noted in the text. This information is available via the Internet at http://www.springer.com/chemistry/journal/11814.
Electronic Supplementary Material
Rights and permissions
About this article
Cite this article
Thuy, L.T., Mallick, S., Kim, S. et al. Synthesis and characterization of oligo aminoglycosides and polyethylenimine conjugates as polymeric gene carriers. Korean J. Chem. Eng. 40, 325–336 (2023). https://doi.org/10.1007/s11814-022-1296-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-022-1296-8