Abstract
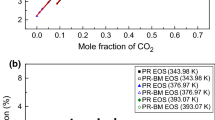
The vapor+liquid equilibrium (VLE) for the 2-components of propylene glycol monomethyl ether (PGME) and propylene glycol monomethyl ether acetate (PGMEA) in high pressure (HP) supercritical carbon dioxide (S-CO2) was evaluated. The solubility data determination was performed by the synthetic method at T=(313.2 to 393.2) K and p=(1.92 to 16.5) MPa. The obtained results indicated that the solubility of S-CO2 was found to increase monotonically with the increase of system temperature and mole fraction of PGME and PGMEA in binary (solute+solvent) mixtures. The solubility curve of PGME and PGMEA in the PGME+S-CO2 and PGMEA+S-CO2 models increases in connection with the increasing T at a steady pressure. The PGME+S-CO2 and PGMEA+S-CO2 models reveal type-I phase behavior (PB). The critical properties were achieved by Joback and Aspen plus method. Moreover, the experimental result adequately correlated with the Peng-Robinson equation of state (P-R E-O-S). Root mean square deviation (RMSD) for the PGME+S-CO2 [Joback: kij=0.0, hij=−0.060, Aspen: kij=0.0, hij=−0.065] and PGMEA+S-CO2 [Joback: kij=0.0, hij=0.0, Aspen: kij=0.0, hij=0.0] systems using two factors determined at 353.2 K was 9.07% (Joback), 10.98% (Aspen) and 4.03% (Joback), 4.78% (Aspen), respectively.
Similar content being viewed by others
Abbreviations
- a:
-
fascination factor [Pa-m6/mol2]
- b:
-
Co-volume factor [m3/mol]
- BP:
-
bubble point [MPa]
- CP:
-
critical point
- DP:
-
dew point
- N:
-
number of the data points
- P:
-
pressure [Pa]
- R:
-
universal gas constant [8.314462 J/(mol·K)]
- T:
-
temperature [K]
- v:
-
molar volume [m3·mol−1]
- x:
-
liquid phase mole fractions
- α :
-
alpha function
- ω :
-
acentric factor
- E-O-S :
-
equation of state
- OF :
-
objective function
- P-R :
-
Peng-Robinson
References
K. Zelijko, D. Cor and M. K. Hrncic, J. Chem. Eng. Data, 63, 860 (2018).
G. J. Philip and L. Walter, Chemical synthesis using supercritical fluids, Weinheim, Wiley-VCH (1999).
G. Brunner, Annu. Rev. Chem. Biomol. Eng., 1, 321 (2010).
L.-S. Anne, C. Aymonier and F. Cansell, J. Chem. Technol. Biot., 85, 583 (2010).
H.-S. Byun and H.-Y. Lee, J. Chem. Eng. Data, 51, 1436 (2006).
H.-S. Byun, M.-Y. Choi and J.-S. Lim, J. Supercrit. Fluids, 37, 323 (2006).
Y.-S. Jang, H.-H. Jeong and H.-S. Byun, J. Ind. Eng. Chem., 18, 414 (2012).
S.-H. Kim, M.-H. Park, J.-S. Lim and H.-S. Byun, J. Ind. Eng. Chem., 16, 962 (2010).
S.-H. Cho, D.-S. Yang and H.-S. Byun, Fluid Phase Equilib., 351, 18 (2013).
C.-R. Kim and H.-S. Byun, Fluid Phase Equilib., 381, 51 (2014).
S.-H. Cho, C.-R. Kim, S.-D. Yoon and H.-S. Byun, Fluid Phase Equilib., 396, 74 (2015).
H.-S. Byun and D.-H. Lee, Ind. Eng. Chem. Res., 45, 3354 (2006).
H.-S. Byun, B. M. Hasch and M. A. McHugh, Fluid Phase Equilib., 115, 179 (1996).
H.-S. Byun and C. Park, Korean J. Chem. Eng., 19, 126 (2002).
H.-S. Byun and J.-S. Shin, J. Chem. Eng. Data, 48, 97 (2003).
L. Michele and M. A. McHugh, Fluid Phase Equilib., 157, 285 (1999).
Y. Wu, M. S. Newkirk, S. T. Dudek, K. Williams, V. Krukonis and M. A. McHugh, Ind. Eng. Chem. Res., 53, 10133 (2014).
J. Liu, D. Li, H.-S. Byun and M. A. McHugh, Fluid Phase Equilibria, 267, 39 (2008).
J. M. G. Cowie and I. J. McEwen, Polymer, 24, 1453 (1983).
J. M. G. Cowie and I. J. McEwen, J. Chem. Soc., Faraday Trans. 1: Phys. Chem. Condensed Phases, 70, 171 (1974).
J. M. G. Cowie and I. J. McEwen, British Polymer J., 7, 459 (1975).
H. Matsukawa, A. Fujii, T.-A. Hoshina and K. Otake, J. Chem. Eng. Data, 548, 113172 (2021).
N. V. K. A. Sudhir, B. R. Mellein, E. M. Saurer and J. F. Brennecke, J. Phys. Chem. B, 108, 20355 (2004).
M. A. McHugh and V. J. Krukonis, Supercritical fluid extraction, 2nd ed., Butterworth-Heinemann, Stoneham (1994).
M. A. McHugh, F. Rindfleisch, P. T. Kuntz, C. Schmaltz and M. Buback, Polymer, 39, 6049 (1998).
S.-H. Lee, M. A. LoStracco, B. M. Hasch and M. A. McHugh, J. Phys. Chem., 98, 4055 (1994).
P. N. P. Ghoderao, D. Dhamodharan and H.-S. Byun, J. Chem. Thermodyn., 168, 106746 (2022).
S.-H. Cho, C.-R. Kim, S.-D. Yoon and H.-S. Byun, Fluid Phase Equilib., 396, 74 (2015).
R. R. Mallepally, V. S. Gadepalli, B. A. Bamgbade, N. Cain and M. A. McHugh, J. Chem. Eng. Data, 61, 2818 (2016).
Y. Wu, M. S. Newkirk, S. T. Dudek, K. Williams, V. Krukonis and M. A. McHugh, Ind. Eng. Chem. Res., 53, 10133 (2014).
K. L. Albrecht, F. P. Stein, S. J. Han, C. J. Gregg and M. Radosz, Fluid Phase Equilib., 117, 84 (1996).
B. Folie, C. Gregg, G. Luft and M. Radosz, Fluid Phase Equilib., 120, 11 (1996).
E. Kiran, J. Supercrit. Fluids, 110, 126 (2016).
B. E. Poling, J. M. Prausnitz and J. P. O’connell, The properties of gases and liquids, Vol. 5, McGraw-Hill, New York (2001).
D. Y. Peng and D. B. Robinson, Ind. Eng. Chem. Fundam., 15, 59 (1976).
C.-T. Hsieh, M.-J. Lee and H.-M. Lin, Ind. Eng. Chem. Res., 45, 2123 (2006).
L. S. Robert and P. H. van Konynenburg, Discuss. Faraday Soc., 49, 87 (1970).
Acknowledgements
This research was supported by Ministry of Trade, Industry & Energy (MOTIE), Korea Institute for Advancement of Technology (KIAT) through “A supporting program for the middle market enterprises in each region” (No. P0017536).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kwon, YT., Dhamodharan, D., Choi, H. et al. Experimental and computational phase behavior analysis of the PGME+CO2 and PGMEA+CO2 mixture at high pressures. Korean J. Chem. Eng. 39, 2783–2791 (2022). https://doi.org/10.1007/s11814-022-1110-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-022-1110-7