Abstract
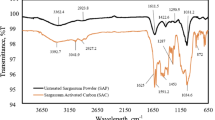
Waste crab shell, which has some functional groups like -NHCO or NO2 groups, was used as an adsorbent to remove arsenate ions (As (V)). The functional groups in crab shells were confirmed by FT-IR analysis. Waste crab shell had a high uptake capacity of 35.92 mg/g-dry mass for arsenate ion at pH 4, and the regression curve using the Langmuir isotherm equation fit well with the experimental data. The effects of pH, loading of crab shells, and time on uptake capacity of arsenate ions were also investigated. The adsorption capacity of arsenate ions was increased as the pH value was increased because the amount of negative arsenic species increased as the pH value was increased. Waste crab shells could remove arsenate ions of about 45% with 0.5 g of loading amount, and adsorption of arsenate ions was almost completed in 30 min when initial concentration of arsenate ions was 100 and 9.3 mg/L, respectively. Considering recycling of crab shell, it could be an economical and promising adsorbent.
Similar content being viewed by others
References
Shugik, and T. S. Singh, Ind. J. Environ. Health., 45 (2003).
D. A. Joseph, L. M. Denise, M. F. Sarah and W. R. Keith, Environ. Sci. Technol., 37 (2003).
M. F. Hughes, Toxicol. Lett., 133 (2002).
S. Chakravarty, V. Dureja, G. Bhattacharya, S. Maity and S. Bhattacharjee, Water Res., 36 (2001).
K. A. Matis, A. I. Zamboulis, D. Zamboulis and A.V. Valtadorou, Water Air Soil Pollut., 111 (1999).
A. Zouboulis and I. Katsoyiannis, Sep. Sci. Technol., 37 (2002).
L. G. Twidwell, R. G. Robins and T. Nishimura, EPD Congress, Arsenic Symposium, TMS, USA (2005).
P. B. Nagarnaik, A. G. Bhole and G. S. Natarajan, Ind. Environ. Health, 45 (2003).
Y. Madrid, M. E. Barrio-Cordoba and C. Camara, Analyst., 123, 1593 (1998).
M. C. Dos Santos and E. Lenzi, Environ. Technol., 21, 615 (2000).
W. Wafworo, C.W. Seo and W. E. Marshall, J. Chem. Technol. Biotechnol., 74, 1117 (1999).
T. Vaughan, C.W. Seo and W. E. Marshall, Bioresour. Technol., 78, 133 (2001).
C. Jeon and J.W. Kim, J. Ind. Eng. Chem., Submitted (2009).
K. Vijayaraghavan, K. Palanivelu and M. Velan, J. Hazard. Mater., B 119 (2005).
S. Pradhan, S. S. Shukla and K. L. Dorris, J. Hazard. Mater., B 125 (2005).
K. Vijayaraghavan, K. Palanivelu and M. Velan, Bioresour. Technol., 97 (2006).
S. H. Yeom and D. J. Jeon, Bioresour. Technol., 86, 32 (2009).
M. M. Srivasava, P. Kumari, P. Sharma and S. Srivastava, Int. J. Miner. Process., 78, 131 (2006).
E. Diamantopoulos, S. Ioannidis and G. P. Sakelaropoulos, Water Res., 27(12) (1993).
H. S. Altundogan, F. Tumen and M. Bildik, Waste Manage., 22 (2002).
L. Jun-Kun and W. U. Tsair-Fuh, Water Res., 35(8) (2001).
B. D. Honeyman and A. H. Santcchi, Environ. Sci. Technol., 22, 862 (1988).
C. Tien, Butter worth-Heinemann, Boston, pp. 29–40 (1994).
H. N. Catherine, B. Volesky and C. Daniel, Water Res., 41, 2473 (2007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jeon, C. Removal of As(V) from aqueous solutions by waste crab shells. Korean J. Chem. Eng. 28, 813–816 (2011). https://doi.org/10.1007/s11814-010-0432-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-010-0432-z