Abstract
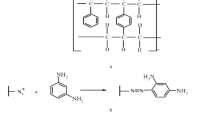
A new chelating resin was prepared by coupling Amberlite XAD-4 with phenol through an azo spacer, then modified by allyl bromide and characterized (by elemental analysis and IR) and studied for preconcentration of Cu(II) using flame atomic absorption spectrometry (FAAS) for metal monitoring. The optimum pH value for sorption of the above mentioned metal ion was 4.5. The resin was subjected to chemical evaluation through batch binding and column chromatography of Cu(II). The chelating resin can be reused for 15 cycles of sorption-desorption without any significant change in sorption capacity. A recovery of 98% was obtained for the metal ion with 0.5 M HNO3 as eluting agent. The equilibrium adsorption data of Cu(II) on modified resin were analyzed by Langmuir, Freundlich and Temkin models. Based on equilibrium adsorption data the Langmuir, Freundlich and Temkin constants were determined to be 0.061, 0.193 and 0.045 at pH 4.5 and 25 °C. The method was applied for the copper determination from industrial waste water sample.
Similar content being viewed by others
References
B. S. Garg, R.K. Sharma, N. Bhojak and S. Mittal, Microchem. J., 61, 94 (1999).
V. Camel, Spectrochim. Acta Part B, 58, 1177 (2003).
S. D. D. Cekic, H. Filik and R. Apak, Anal. Chim. Acta, 505, 15 (2004).
S.G. Dmitrienko, O.A. Sviridova, L.N. Pyatkova and V. M. Senyavin, Anal. Bioanal. Chem., 374, 361 (2002).
P. K. Gal, S. Patel and B. K. Mishra, Talanta, 62, 1005 (2004).
V. A. Lemos and P. X. Baliza, Talanta, 67, 564 (2005a).
V.A. Lemos, P.X. Baliza and J. S. Santos, Sep. Sci. Technol., 39, 3317 (2004).
V.A. Lemos, P.X. Baliza and R. T. Yamaki, Talanta, 61, 675 (2003).
M. Kumar, D. P. S. Rathore and A. K. Singh, Talanta, 51, 1187 (2000).
I. Narin, M. Soylak, K. Kayakirilmaz, L. Elci and M. Dogan, Anal. Lett., 36, 641 (2003).
D. Prabhakaran and M. S. Subramanian, Talanta, 59, 1227 (2003).
P. K. Tewari and A. K. Singh, Analyst, 125, 2350 (2000).
P. K. Tewari and A. K. Singh, Talanta, 56, 735 (2002).
N. J.K. Simpson, Solid Phase Extraction, Principles, Techniques and Applications, 1th ed., Marcel Dekker Inc., New York (2000).
R. Saxena, A. K. Singh and D. P. S. Rathore, Analyst, 120, 403 (1995).
P. K. Tewari and A. K. Singh, Analyst, 124, 1847 (1999).
P. K. Tewari and A. K. Singh, Talanta, 53, 823 (2001).
Y. Guo, B. Din, Y. Liu, X. Chang, S. Meng and M. Tian, Anal. Chim. Acta, 504, 319 (2004).
P. Metilda, K. Sanghamitra, J. Mary Gladis, G. R.K. Naidu and T. Prasada Rao, Talanta, 65, 192 (2005).
R. Saxena, A. K. Singh and S. S. Sambi, Anal. Chim. Acta, 295, 199 (1994).
V.A. Lemos, P. X. Baliza, J. S. Santos, L. S. Nunes, A.A. Sesus and M. E. Rocha, Talanta, 66, 174 (2005b).
K. Dev, R. Pathak and G. N. Rao, Talanta, 48, 579 (1999).
L. Langmuir, J. Am. Chem. Soc., 40, 1361 (1918).
K. L. Hall, L. C. Eagleton, A. Acrivos and T. Vermeulen, Ind. Eng. Chem. Fundam., 5, 212 (1966).
H. M.A. Freundlich, J. Phys. Chem., 57, 385 (1906).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nezhati, M.N., Panahi, H.A., Moniri, E. et al. Synthesis, characterization and application of allyl phenol modified amberlite XAD-4 resin for preconcentration and determination of copper in water samples. Korean J. Chem. Eng. 27, 1269–1274 (2010). https://doi.org/10.1007/s11814-010-0174-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-010-0174-y