Abstract
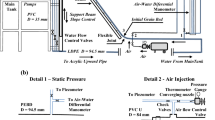
Molten salt oxidation process is one of the most promising alternatives to incineration that can be used to effectively destroy the organic components of mixed and hazardous wastes. To detect the flow characteristics of the molten salt oxidation process (air-molten carbonate salt two-phase flow), differential pressure fluctuation signals from a molten salt oxidation process have been analyzed by adopting the stochastic methods. Effects of the input air flow rate (0.05–0.22 m/sec) and the molten salt temperature (870–970 °C) on the phase holdup and flow characteristics are studied. The gas holdup increases with an increasing molten salt temperature due to the decrease of the viscosity and surface tension of the molten carbonate salt. It is found that a stochastic analysis of the differential pressure signals enables us to obtain the flow characteristics in the molten salt oxidation process. The experimentally obtained gas holdup data in the molten salt reactor were well described and characterized by means of the drift-flux model.
Similar content being viewed by others
References
W. D. Deckwer, R. Burckhart and G. Zoll, Chem. Eng. Sci., 29, 2177 (1974).
Y. Kang, J. S. Shim, M. H. Ko and S. D. Kim, Korean J. Chem. Eng., 13, 317 (1996).
S. J. Kim, Y. J. Cho, C. G. Lee, Y. Kang and S. D. Kim, Korean J. Chem. Eng., 19, 175 (2002).
J. H. Bell, P. A. Hass and J. C. Rudolph, Sep. Sci. Technol., 30, 1755 (1995).
P. C. Hsu, K. G. Foster, T. D. Ford, P. H. Wallman, E. Watkins, C. O. Pruneda and M.G. Adamson, Waste Management, 20, 363 (2000).
M. G. Adamson, P. C. Hsu, D. L. Hipple, K.G. Foster, R. W. Hopper and T. D. Ford, in G. E. Marcelle (Ed.), France, 1 (1998).
C. O. Pruned, B. E. Watkins and R. S. Upadhye, JANNAF Propulsion and Subcommittee Joint Meeting, December, FL (1995).
M. Alam and S. Kamath, Environ. Sci. Technol., 32, 3986 (1998).
C. Vial, S. Poncin, G. Wild and N. Midoux, Chem. Eng. Proc., 40, 135 (2001).
K. J. Woo, Y. J. Cho, K. I. Kim, Y. Kang and S. D. Kim, HWAHAK KONGHAK, 36, 937 (1998).
T. J. Lin, K. Tsuchiya and L. S. Fan, AIChE J., 44, 545 (1998).
R. Pohorecki, W. Moniuk, A. Zdrojkowski and P. Bielski, Chem. Eng. Sci., 56, 1167 (2001).
Y. J. Cho, P. S. Song, S. H. Kim, Y. Kang and S. D. Kim, J. Chem. Eng. of Japan, 34, 254 (2001).
Y. J. Cho, K. J. Woo, Y. Kang and S. D. Kim, Chem. Eng. Pro., 41, 699 (2002).
L. T. Fan, Y. Kang, M. Yashima and D. Neogi, Chem. Eng. Comm., 135, 147 (1995).
Y. Kang, K. J. Woo, M. H. Ko, Y. J. Cho and S. D. Kim, Korean J. Chem. Eng., 16, 784 (1999).
L. S. Fan, Butterworths, MA (1989).
Y. Kang and S.D. Kim, Ind. Eng. Chem. Process Des. Dev., 25, 717 (1986).
J. Zahradnik, M. Fialova, M. Ruzicka, J. Drahos, F. Kastanek and N. H. Thomas, Chem. Eng. Sci., 52, 3811 (1997).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cho, YZ., Yang, HC. & Kang, Y. Two phase (air-molten carbonate salt) flow characteristics in a molten salt oxidation reactor. Korean J. Chem. Eng. 26, 828–832 (2009). https://doi.org/10.1007/s11814-009-0138-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-009-0138-2