Abstract
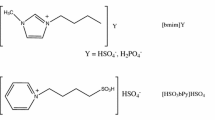
Several water-stable ionic liquids with different acidity and affinity were synthesized and applied as both solvents and acid catalysts for Fischer esterification of ethanol reacting with four aliphatic carboxylic acids (acetic acid, n-hexanoic acid, lauric acid, and stearic acid). Among the studied ionic liquids, [(n-bu-SO3H) MIM][HSO4] (3-butyl-1-(butyl-4-sulfonyl) imidazolium sulfate) and [(n-bu-SO3H) MIM][p-TSO] (3-butyl-1-(butyl-4-sulfonyl) imidazolium toluenesulfonate) show higher reactivity for the production of ethyl esters. The catalytic activities of these ionic liquids are strongly dependent on the acidity of their anions and cations, as well as their hydrophilicity and affinity with the reactants. Water refluxing through the condenser may be another important reason for obtaining high conversion of esterification, indicating a water-sequester process is still needed in order to obtain a higher yield of ester in the ionic liquid catalyzed esterification system. Kinetics studies show the conversions of the acids increase with reaction temperature and time, and reach equilibrium within about two hours. The apparent activation energies are 39.1±2.0, 49.7±2.5, 51.4±2.5 and 59.3±3.0 kJ·mol−1 for the formation of ethyl acetate, ethyl n-hexanoate, ethyl laurate and ethyl stearate, respectively.
Similar content being viewed by others
References
N. N. Gandhi, N. S. Patil, S. B. Sawant, J. B. Joshi, P. P. Wangikar and D. Mukesh, Cat. Rev. Sci. Eng., 42, 439 (2000).
H. E. Hoydonckx, D. E. De Vos, S. A. Chavan and P. A. Jacobs, Top. Catal., 27, 83 (2004).
J. Otera, Angew. Chem. Int. Ed., 40, 2044 (2001).
B. Dragoi and E. Dumitriu, Prog. Catal., 12, 1 (2003).
K. Kulesza and P. Bartoszewicz, In modern polymeric materials for environmental applications, 2 ed., Krakow, Poland, 93 (2006).
H. E. Lanman, R.V. Nguyen, X. Q. Yao, T. H. Chan and C. J. Li, J. Mol. Catal. A: Chem., 279, 218 (2008).
H. Y. Ju, J. Y. Ahn, M. D. Manju, K. H. Kim and D. W. Park, Korean J. Chem. Eng., 25, 471 (2008).
Z. Y. Duan, Y. L. Gu and Y. Q. Deng, J. Mol. Catal. A: Chem., 246, 70 (2006).
R. Sugimura, K. Qiao, D. Tomida and C. Yokoyama, Catal. Commun., 8, 770 (2007).
Y. Q. Deng, F. Shi, J. J. Beng and K. Qiao, J. Mol. Catal. A: Chem., 165, 33 (2001).
H. P. Zhu, F. Yang, J. Tang and M. Y. He, Green Chemistry, 5, 38 (2003).
T. Welton, Chem. Rev., 99, 2071 (1999).
J. Fraga-Dubreuil, K. Bourahla, M. Rahmouni, J. P. Bazureau and J. Hamelin, Catal. Commun., 3, 185 (2002).
P. Wasserscheid, M. Sesing and W. Korth, Green Chemistry, 4, 134 (2002).
P. Wasserscheid, R. van Hal and A. Bosmann, Green Chemistry, 4, 400 (2002).
M. T. Garcia, N. Gathergood and P. J. Scammells, Green Chemistry, 7, 9 (2005).
A. C. Cole, J. L. Jensen, I. Ntai, K. L. T. Tran, K. J. Weaver, D. C. Forbes and J. J. H. Davis, J. Am. Chem. Soc., 124, 5962 (2002).
D. C. Forbes, J. H. Davis, D. W. Morrison, K. L. T. Tran and K. J. Weaver, Abstr. Pap. Am. Chem. Soc., 225, U416 (2003).
D. C. Forbes and K. J. Weaver, J. Mol. Catal. A: Chem., 214, 129 (2004).
H. B. Xing, T. Wang, Z. H. Zhou and Y. Y. Dai, Ind. Eng. Chem. Res., 44, 4147 (2005).
H. B. Xing, T. Wang, Z. H. Zhou and Y. Y. Dai, J. Mol. Catal. A: Chem., 264, 53 (2007).
P. A. Ganeshpure and J. Das, React. Kinet. Catal. Lett., 92, 69 (2007).
D. Fang, X. L. Zhou, Z. W. Ye and Z. L. Liu, Ind. Eng. Chem. Res., 45, 7982 (2006).
Y. L. Gu, F. Shi and Y. Q. Deng, J. Mol. Catal. A: Chem., 212, 71 (2004).
P. A. Ganeshpure, G. George and J. Das, ARKIVOC, 273 (2007).
P. A. Ganeshpure, G. George and J. Das, J. Mol. Catal. A: Chem., 279, 182 (2008).
H. B. Zhang, F. Xu, X. H. Zhou, G. Y. Zhang and C X. Wang, Green Chemistry, 9, 1208 (2007).
Z. J. Wei, Y. Huang, S. G. Deng, D. Saha, Y. X. Liu and Q. L. Ren, React. Kinet. Catal. Lett., 95, 257 (2008).
Q. W. Yang, Z. J. Wei, H. B. Xing and Q. L. Ren, Catal. Commun., 9, 1307 (2008).
C. M. Wang, L. P. Guo, H. R. Li, Y. Wang, J. Y. Weng and L. H. Wu, Green Chemistry, 8, 603 (2006).
Animal and vegetable fats and oils-analysis by gas chromatography of methyl ester of fatty acids. In National Standard of China, GB/T 17377 (1998).
H. P. Nguyen, S. Znifeche and M. Baboulene, Synth. Commun., 34, 2085 (2004).
T. Joseph, S. Sahoo and S. B. Halligudi, J. Mol. Catal. A: Chem., 234, 107 (2005).
P. Izak, N. M. M. Mateus, C. A. M. Afonso and J. G. Crespo, Sep. Purif. Technol., 41, 141 (2005).
K. Belafi-Bako, N. Dormo, O. Ulbert and L. Gubicza, Desalination, 149, 267 (2002).
M. Pera-Titus, C. Fite, V. Sebastian, E. Lorente, J. Llorens and F. Cunill, Ind. Eng. Chem. Res., 47, 3213 (2008).
S. Al-Asheh, F. Banat and A. L. Al-Lagtah, Trans. IChemE, Part A, 82, 855 (2004).
T. Jiang, Y. H. Chang, G. Y. Zhao, B. X. Han and G. Y. Yang, Synth. Commun., 34, 225 (2004).
X. Z. Li and W. J. Eli, J. Mol. Catal. A: Chem., 279, 159 (2008).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wei, Z., Li, F., Xing, H. et al. Reactivity of Brönsted acid ionic liquids as dual solvent and catalyst for Fischer esterifications. Korean J. Chem. Eng. 26, 666–672 (2009). https://doi.org/10.1007/s11814-009-0111-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-009-0111-0