Abstract
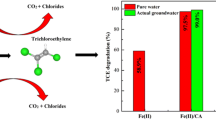
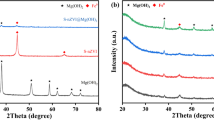
Zero-valent iron (ZVI) aerogels have been synthesized by sol-gel method and supercritical CO2 drying, followed by H2 reduction in the temperature range of 350–500 °C. When applied to trichloroethylene (TCE) dechlorination, the ZVI aerogel reduced at 370 °C showed the highest performance in the conditions employed in this study. Thus, the effect of reduction temperature in preparing ZVI aerogels has been investigated by several characterizations such as BET, XRD, TPR, and TEM analyses. As the reduction temperature decreased from 500 to 350 °C, the BET surface area of the resulting aerogels increased from 6 to 30 m2/g, whereas their Fe0 content decreased up to 64%. It was also found that H2 reduction at low temperatures such as 350 and 370 °C leads to the formation of ZVI aerogel particles consisting of both Fe0 and FeO x in the particle cores with a different amount ratio, where FeO x is a mixture of maghemite and magnetite. It is, therefore, suggested that reduction at 370 °C for ZVI aerogel preparation yielded particles homogeneously composed of Fe0 and FeO x in the amount ratio of 87/13, resulting in high TCE dechlorination rate. On the other hand, when Pd- and Ni-ZVI aerogels were prepared via cogellation and then applied for TCE dechlorination, we also observed a similar effect of reduction temperature. However, the reduction at 350 or 370 °C produced Pd- or Ni-ZVI aerogel particles in which Fe0 and Fe3O4 co-exist homogeneously. Since both Fe0 and Fe3O4 are advantageous in TCE dechlorination, the activities of Pd- and Ni-ZVI aerogels reduced at 350 °C were comparable to those of both aerogels reduced at 370 °C, although the former aerogels have less Fe0 content.
Similar content being viewed by others
References
C. M. Kao, S. C. Chen and J.K. Liu, Chemosphere, 43, 1071 (2001).
M.M. Scherer, S. Richter, R. L. Valentine and P. J. J. Alvarez, Critical Reviews in Environmental Science and Technology, 30(3), 363 (2000).
D. E. Meyer and D. Bhattacharyya, J. Phys. Chem. B, 111, 7142 (2007).
S. M. Ponder, J.G. Darab and T. E. Mallouk, Environ. Sci. Technol., 34, 2564 (2000).
S. M. Ponder, J.G. Darab, J. Bucher, D. Caulder, I. Craig, L. Davis, N. Edelstein, W. Lukens, H. Nitsche, L. Rao, D.K. Shuh and T. E. Mallouk, Chem. Mater., 13, 479 (2001).
B. Schrick, J. L. Blough, A. D. Jones and T. E. Mallouk, Chem. Mater., 14, 5140 (2002).
C. B. Wang and W. X. Zhang, Environ. Sci. Technol., 31, 2154 (1997).
J. T. Nurmi, P.G. Tratnyek, V. Sarathy, D. R. Baer, J. E. Amonette, K. Pecher, C. Wang, J. C. Linehan, D.W. Matson, R. L. Penn and M. D. Driessen, Environ. Sci. Technol., 39, 1221 (2005).
G. N. Glavee, K. J. Klabunde, C.M. Sorensen and G. C. Hadjipanayis, Inorg. Chem., 34, 28 (1995).
N. Duxin, O. Stephan, C. Petit, P. Bonville, C. Colliex and M. P. Pileni, Chem. Mater., 9, 2096 (1997).
N. Duxin, M. P. Pileni, W. Wernsdorfer, B. Barbara, A. Benoit and D. Mailly, Langmuir, 16, 11 (2000).
E. E. Carpenter, S. Calvin, R. M. Stroud and V. G. Harris, Chem. Mater., 15, 3245 (2003).
M. Uegami, J. Kawano, T. Okita, Y. Fujii, K. Okinaka, K. Kakuya and S. Yatagai, US Patent 7,022,256 (2006).
Y. Liu, H. Choi, D. Dionysiou and G.V. Lowry, Chem. Mater., 17, 5315 (2005).
P.D. Mackenzie, D. P. Horney and T.M. Sivavec, J. Hazard. Mater., 68, 1 (1999).
C.-C. Liu, D.-H. Tseng and C.-Y. Wang, J. Hazard. Mater. B, 136, 706 (2006).
T. L. Johnson, W. Fish, Y.A. Gorby and P.G. Tratnyek, J. Contam. Hydrol., 29, 379 (1998).
A.G. B. Williams and M. M. Scherer, Environ. Sci. Technol., 38, 4782 (2004).
W.X. Zhang, C. B. Wang and H. L. Lien, Cata. Today, 40(4), 387 (1998).
E.K. Nyer and D. B. Vance, Ground Water Monit. Rem., 21(2), 41 (2001).
A. E. Gash, T. M. Tillotson, J. H. Satcher Jr., J. F. Poco, L.W. Hrubesh and R. L. Simpson, Chem. Mater., 13, 999 (2001).
O. J. Wimmers, P. Arnoldy and J.A. Moulijn, J. Phys. Chem., 90, 1331 (1986).
M. J. Tiernan, P.A. Barnes and G. M. B. Parkes, J. Phys. Chem., 105, 220 (2001).
E. E. Carpenter, J.W. Long, D. R. Rolison, M. S. Logan, K. Pettigrew, R. M. Stroud, L. T. Kuhn, B.R. Hansen and S. Morup, J. Appl. Phys., 99, 08N711 (2006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ryu, J., Suh, D.J., Park, YK. et al. Effect of reduction temperature on the preparation of zero-valent iron aerogels for trichloroethylene dechlorination. Korean J. Chem. Eng. 25, 1377–1384 (2008). https://doi.org/10.1007/s11814-008-0226-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11814-008-0226-8