Abstract
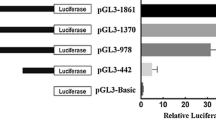
GATA4, as a member of the GATA transcription factor family, plays a significant role in lineage specification and trans-differentiation of cells by regulating target gene expression. In this study, the potential transcription factor binding sites in the promoter of Paralichthys olivaceus sox2 gene were analyzed, and a GATA4-binding motif was identified. A cleaved amplified polymorphic sequence-based binding assay experiment was performed, and the results showed a significant binding process between the Homo sapiens GATA4 recombinant protein and the DNA fragment of P. olivaceus sox2 promoter. Luciferase reporter assay revealed that GATA4 could improve the activity of the P. olivaceus sox2 promoter. Overexpression of P. olivaceus GATA4 could also increase the sox2 mRNA expression level in flounder brain cells. Overall, our results indicate that sox2 is a target gene of GATA4 in P. olivaceus. Sox2 is implicated in the maintenance of stem cell pluripotency and can reprogram differentiated cells to iPS cells. In addition, sox2 is involved in the development of the central nervous system (CNS). Therefore, this study may provide a reference for exploring the function of P. olivaceus GATA4 in the regulation of pluripotent stem cells and the development of the CNS.
Similar content being viewed by others
References
Adachi, K., Suemori, H., Yasuda, S. Y., Nakatsuji, N., and Kawase, E., 2010. Role of SOX2 in maintaining pluripotency of human embryonic stem cells. Genes to Cells, 15(5): 455–470, https://doi.org/10.1111/j.1365-2443.2010.01400.x.
Agnihotri, S., Wolf, A., Munoz, D. M., Smith, C. J., Gajadhar, A., Restrepo, A., et al., 2011. A GATA4-regulated tumor suppressor network represses formation of malignant human astrocytomas. The Journal of Experimental Medicine, 208(4): 689–702, https://doi.org/10.1084/jem.20102099.
Agnihotri, S., Wolf, A., Picard, D., Hawkins, C., and Guha, A., 2009. GATA4 is a regulator of astrocyte cell proliferation and apoptosis in the human and murine central nervous system. Oncogene, 28(34): 3033–3046, https://doi.org/10.1038/onc.2009.159.
Avilion, A. A., Nicolis, S. K., Pevny, L. H., Perez, L., Vivian, N., and Lovellbadge, R., 2003. Multipotent cell lineages in early mouse development depend on SOX2 function. Genes & Development, 17(1): 126–140, https://doi.org/10.1101/gad.224503.
Bertolini, J. A., Favaro, R., Zhu, Y., Pagin, M., Ngan, C. Y., Wong, C. H., et al., 2019. Mapping the global chromatin connectivity network for Sox2 function in neural stem cell maintenance. Cell Stem Cell, 24(3): 462–476.e6, https://doi.org/10.1016/j.stem.2019.02.004.
Bowles, J., Schepers, G., and Koopman, P., 2000. Phylogeny of the SOX family of developmental transcription factors based on sequence and structural indicators. Developmental Biology, 227(2): 239–255, https://doi.org/10.1006/dbio.2000.9883.
Cartharius, K., Frech, K., Grote, K., Klocke, B., Haltmeier, M., Klingenhoff, A., et al., 2005. Matlnspector and beyond: Promoter analysis based on transcription factor binding sites. Bioinformatics, 21(13): 2933–2942, https://doi.org/10.1093/bioinformatics/bti473.
Cavallaro, M., Mariani, J., Lancini, C., Latorre, E., Caccia, R., Gullo, F., et al., 2008. Impaired generation of mature neurons by neural stem cells from hypomorphic Sox2 mutants. Development, 135(3): 541, https://doi.org/10.1242/dev.010801.
Davis, R. L., Weintraub, H., and Lassar, A. B., 1987. Expression of a single transfected cDNA converts fibroblasts to myoblasts. Cell, 51(6): 987–1000, https://doi.org/10.1016/0092-8674(87)90585-X.
Fan, L., Jiang, J., Gao, J., Song, H., Liu, J., Yang, L., et al., 2015. Identification and characterization of a PRDM14 homolog in Japanese flounder (Paralichthys olivaceus). International Journal of Molecular Sciences, 16(5): 9097–9118, https://doi.org/10.3390/ijms16059097.
Fujiwara, Y., Browne, C. P., Cunniff, K., Goff, S. C., and Orkin, S. H., 1996. Arrested development of embryonic red cell precursors in mouse embryos lacking transcription factor GATA-1. Proceedings of the National Academy of Sciences of the United States of America, 93(22): 12355, https://doi.org/10.1073/pnas.93.22.12355.
Gao, J., Wang, Z., Shao, K., Fan, L., Yang, L., Song, H., et al., 2014. Identification and characterization of a Sox2 homolog in the Japanese flounder Paralichthys olivaceus. Gene, 544(2): 165–176, https://doi.org/10.1016/j.gene.2014.04.062.
Holtzinger, A., 2005. Gata4 regulates the formation of multiple organs. Development, 132(17): 4005–4014, https://doi.org/10.1242/dev.01978.
Huang, H. N., Chen, S. Y., Hwang, S. M., Yu, C. C., Su, M. W., Mai, W., et al, 2014. miR-200c and GATA binding protein 4 regulate human embryonic stem cell renewal and differentiation. Stem Cell Research, 12(2): 338–353, https://doi.org/10.1016/j.scr.2013.11.009.
Huang, P. Y., He, Z. Y., Ji, S. Y., Sun, H. W., Xiang, D., Liu, C. C., et al., 2011. Induction of functional hepatocyte-like cells from mouse fibroblasts by defined factors. Nature, 475(7356): 386, https://doi.org/10.1038/nature10116.
Ieda, M., Fu, J. D., Delgado-Olguin, P., Vedantham, V., Hayashi, Y., Bruneau, B. G., et al., 2010. Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell, 142(3): 375–386, https://doi.org/10.1016/j.cell.2010.07.002.
Jiang, Y., Tarzami, S., Burch, J. B. E., and Evans, T., 1998. Common role for each of the cGATA-4/5/6 genes in the regulation of cardiac morphogenesis. Developmental Genetics, 22(3): 263, https://doi.org/10.1002/(SICI)1520-6408(1998)22:3<263::AID-DVG8>3.0.CO;2-4.
Kulessa, H., Frampton, J., and Graf, T., 1995. GATA-1 reprograms avian myelomonocytic cell lines into eosinophils, thromboblasts, and erythroblasts. Genes & Development, 9(10): 1250, https://doi.org/10.1101/gad.9.10.1250.
Kuo, C. T., Morrisey, E. E., Anandappa, R., Sigrist, K., Lu, M. M., Parmacek, M. S., et al., 1997. GATA4 transcription factor is required for ventral morphogenesis and heart tube formation. Genes & Development, 11(8): 1048–1060, https://doi.org/10.1101/gad.11.8.1048.
Laforest, B., and Nemer, M., 2011. GATA5 interacts with GATA4 and GATA6 in outflow tract development. Developmental Biology, 358(2): 368, https://doi.org/10.1016/j.ydbio.2011.07.037.
Lentjes, M. H., Niessen, H. E., Akiyama, Y., de Bruine, A. P., Melotte, V., and van Engeland, M., 2016. The emerging role of GATA transcription factors in development and disease. Expert Reviews in Molecular Medicine, 18: e3, https://doi.org/10.1017/erm.2016.2.
Liu, P., Chen, M., Liu, Y., Qi, L. S., and Ding, S., 2018a. CRISPR-based chromatin remodeling of the endogenous Oct4 or Sox2 locus enables reprogramming to pluripotency. Cell Stem Cell, 22(2): 252–261, https://doi.org/10.1016/j.stem.2017.12.001.
Liu, W., Song, H., Li, A., Du, X., Liu, Y., He, Y., et al., 2016. Functional characterization of the Japanese flounder (Paralichthys olivaceus) Sox2 gene promoter. Fish Physiology and Biochemistry, 42(5): 1275–1285, https://doi.org/10.1007/s10695-016-0216-4.
Liu, X. M., Li, Z., Wang, B., Zhu, H., Liu, Y. Z., Qi, J., et al., 2018b. GATA4 is a transcriptional regulator of R-spondin1 in Japanese flounder (Paralichthys olivaceus). Gene, 648: 68–75, https://doi.org/10.1016/j.gene.2018.01.041.
Loh, K. M., and Lim, B., 2011. A precarious balance: Pluripotency factors as lineage specifiers. Cell Stem Cell, 8(4): 363–369, https://doi.org/10.1016/j.stem.2011.03.013.
Lowry, J. A., and Atchley, W. R., 2000. Molecular evolution of the GATA family of transcription factors: Conservation within the DNA-binding domain. Journal of Molecular Evolution, 50(2): 103–115, https://doi.org/10.1109/MWC.2008.4454707.
Martin, G. R., 1981. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proceedings of the National Academy of Sciences of the United States of America, 78(12): 7634–7638, https://doi.org/10.1073/pnas.78.12.7634.
Merika, M. and Orkin, S. H., 1993. DNA-binding specificity of GATA family transcription factors. Molecular and Cellular Biology, 13(7): 3999–4010, https://doi.org/10.1128/MCB.13.7.3999.
Montserrat, N., Nivet, E., Martinez, I. S., Hishida, T., Kumar, S., Miquel, L., et al., 2013. Reprogramming of human fibroblasts to pluripotency with lineage specifiers. Cell Stem Cell, 13(3): 341, https://doi.org/10.1016/j.stem.2013.06.019.
Nemer, G., and Nemer, M., 2003. Transcriptional activation of BMP-4 and regulation of mammalian organogenesis by GATA-4 and -6. Developmental Biology, 254(1): 131, https://doi.org/10.1016/S0012-1606(02)00026-X.
Pandolfi, P. P., Roth, M. E., Karis, A., Leonard, M. W., Dzierzak, E., Grosveld, F. G., et al., 1995. Targeted disruption of the GATA3 gene causes severe abnormalities in the nervous system and in fetal liver haematopoiesis. Nature Genetics, 11(1): 40–44, https://doi.org/10.1038/ng0995-40.
Sambrook, J., Fritsch, E. F., and Maniatis, T., 1982. Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory Press, New York, 545pp.
Sarkar, A., and Hochedlinger, K., 2013. The Sox family of transcription factors: Versatile regulators of stem and progenitor cell fate. Cell Stem Cell, 12(1): 15–30, https://doi.org/10.1016/j.stem.2012.12.007.
Shu, J., Wu, C., Wu, Y., Li, Z., Shao, S., Zhao, W., et al., 2013. Induction of pluripotency in mouse somatic cells with lineage specifiers. Cell, 153(5): 963–975, https://doi.org/10.1016/j.cell.2015.05.020.
Shu, J., Zhang, K., Zhang, M. J., Yao, A. Z., Shao, S. D., Du, F. X., et al., 2015. GATA family members as inducers for cellular reprogramming to pluripotency. Cell Research, 25(2): 169–180, https://doi.org/10.1038/cr.2015.6.
Simon, M. C., 1995. Gotta have GATA. Nature Genetics, 11(1): 9–11, https://doi.org/10.1038/ng0995-9.
Takahashi, K. A. Y. S., 2006. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell, 126: 663–676, https://doi.org/10.1016/j.cell.2006.07.024.
Tsai, F. Y., Keller, G., Kuo, F. C., Weiss, M., Chen, J., Rosenblatt, M., et al., 1994. An early haematopoietic defect in mice lacking the transcription factor GATA-2. Nature, 371(6494): 221–226, https://doi.org/10.1038/371221a0.
Xie, Y., Zhang, Y., Zhao, X., Liu, Y., and Chen, L., 2016. A CAPS-based binding assay provides semi-quantitative validation of protein-DNA interactions. Scientific Reports, 6: 21030, https://doi.org/10.1038/srep21030.
Yang, Y., Li, Z., Wu, X., Chen, H., and Xu, W., 2016. Direct reprogramming of mouse fibroblasts toward Leydig-like cells by defined factors. Stem Cell Reports, 8(1): 39–53, https://doi.org/10.1016/j.stemcr.2016.11.010.
Acknowledgements
This research was supported by the National Natural Science Foundation of China (No. 31672646), the National Key R&D Program of China (No. 2018YFD0900101), the Natural Science Foundation of Shandong Province (No. ZR2018BC052), and the Science and Technology Project of Yantai University (No. 2219008). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, X., Li, Z., Yan, W. et al. GATA4 Is a Transcriptional Regulator of SOX2 in Japanese Flounder (Paralichthys olivaceus). J. Ocean Univ. China 21, 163–170 (2022). https://doi.org/10.1007/s11802-022-4806-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11802-022-4806-8