Abstract
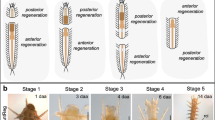
To understand the mechanisms of starfish regeneration, the arms of adult starfish Asterias rollestoni Bell were amputated and their regeneration patterns and cellular mechanisms were studied. It was found that cells in the outer epidermis and inner parietal peritoneum near the end of the stump began to dedifferentiate 4 d after amputation. The dedifferentiated cells in the outer epidermis proliferated, migrated to the wound site and formed a thickened pre-epidermis which would then re-differentiate gradually into mature epidermis. The new parietal peritoneum formed on the coelomic side of wound might be from the curvely elongated parietal peritoneum, resulting from the dedifferentiated and proliferated cells by extension. Afterwards, the proliferated cells made the outer epidermis and inner parietal peritoneum invaginate into the interior dermis and formed blastema-like structures together with induced dedifferentiated dermal cells. Most interestingly, the arm regeneration in A. rollestoni was achieved synchronously by de novo arm-bud formation and growth, and arm-stump elongation. The crucial aspects of arm-bud formation included cell dedifferentiation, proliferation and migration, while those of arm-stump elongation included cell dedifferentiation, proliferation, invagination, and arm-wall-across blastema-like structure formation. The unique pattern and cellular mechanisms of amputated arm regeneration make it easier to understand the rapid regeneration process of adult starfish. This study may lay solid foundations for the research into molecular mechanisms of echinoderm regeneration.
Similar content being viewed by others
References
Alibardi, L., 1996. Observations on proliferating sheath cells in the regenerating nerves of lizard. Histology and Histopathology, 11(4): 937–942.
Belshner, K., Oviedo, N., Newmark, P., and Sánchez Alvarado, A., 2001. The regeneration gap. Nature, 414(6862): 388–390.
Candia Carnevali, M. D., and Bonasoro, F., 2001. Microscopic overview of crinoid regeneration. Microscopic Research and Technique, 55(6): 403–426.
Candia Carnevali, M. D., Bonasoro, F., Patruno, M., and Thorndyke, M. C., 1998. Cellular and molecular mechanisms of arm regeneration in crinoid echinoderms: the potential of arm explants. Development Genes and Evolution, 208(8), 421–430.
Chalkley, D.T., 1954. A quantitative histological analysis of forelimb regeneration in Triturus viridescens. Journal of Morphology, 94(1): 21–70.
Emson, R.H., Wilkie, J.C., 1980. Fission and autotomy in echinoderms. Oceanography and Marine Biology: an Annual Review, 18(1): 155–250
Fan, T. J., Du, Y. T., Cong, R. S., Sun, W. J., Tang, Z. H., Yuan, W. P., Shi, Y., Geng, X. F., and Li, M. Y., 2005. Histological examination of arm regeneration process in starfish Asterias rollestoni. Periodical of Ocean University of China, 35(4): 559–563 (in Chinese, with English abstract).
Feder, M. E., 1999. Engineering candidate genes in studies of adaptation: the heat-shock protein Hsp70 in Drosophila melanogaster. American Naturalist, 154(1): 55–66.
Garcia-Arraras, J. E., Diaz-Miranda, L., Torres, I. I., File, S., Jimenez, L. B., Rivera-Bermudez, K., Arroyo, E. J., and Cruz, W., 1999. Regeneration of the enteric nervous system in the sea cucumber Holothuria glaberrina. Journal of Comparative Neurology, 406(4): 461–475.
Garcia-Arraras, J. E., Estrada-Rodgers, L., Santiago, R., Torres, I., Diaz-Miranda, L., and Torres-Avillan, I., 1998. Cellular mechanisms of intestine regeneration in the sea-cucumber Holothuria glaberrina Selenka (Holothuroidea: Echinodermata). Journal of Experimental Zoology, 281(4): 288–304.
Goss, R. J., 1969. Principles of Regeneration. Academic Press, New York, 292pp.
Hill, S. D., 1970. Origin of the regeneration blastema in polychaete annelids. American zoologist, 10(2): 101–112.
Hotchkiss, F. H. C., 2009. Arm stumps and regeneration models in Asteroidea (Echinodermata). Proceedings of the Biological Society of Washington, 122(3): 342–354.
Huet, M., 1972. Ultrastructural study and development of the neuroepithelial cells of regenerating epidermic arm cells in Asterina gibbosa Penn. Zeitschrift fuer Zellforschung und Mikroskopische Anatomie, 126(1): 75–89 (in French).
Kobayashi, C., Watanabe, K., and Agata, K., 1999. The process of pharynx regeneration in planarians. Developmental Biology, 211(1): 27–38.
Kozlova, A. B., Petukhova, O. A., and Pinaev, G. P., 2006. The analysis of cellular elements in coelomic fluid during early regeneration of the starfish Asterias rubens L. Tsitologiia, 48(3): 175–183.
Lawrence, J. M., 1987. A Functional Biology of Echinoderms. Croom Helm, London, 340pp.
Mattson, P., 1976. Regeneration. Bobbs-Merrill, Indianapolis, 178pp.
Mladenov, P. V., Bisgrove, B., Asotra, S., and Burke, R. D., 1989. Mechanisms of arm-tip regeneration in the sea star, Leptasterias hexactis. Roux’s Archives of Developmental Biology, 198(1): 19–28.
Piscopo, S., De Stefano, R., Thorndyke, M. C., and Brown, E. R., 2005. Alteration and recovery of appetitive behaviour following nerve section in the starfish Asterias rubens. Behavioural Brain Research, 164(1): 36–41.
Price, J., and Allen, S., 2004. Exploring the mechanisms regulating regeneration of deer antlers. Philosophical Transactions of the Royal Society of London — Series B: Biological Sciences, 359(1445): 809–822.
Rajnoch, C., Ferguson, S., Metcalfe, A. D., Herrick, S. E., Willis, H. S., and Ferguson, M. W., 2003. Regeneration of the ear after wounding in different mouse strains is dependent on the severity of wound trauma. Developmental Dynamics, 226(2): 388–397.
Repesh, L. A., and Oberpriller, J. C., 1978. Scanning electron microscopy of epidermal cell migration in wound healing during limb regeneration in the adult newt, Notophthalmus viridescens. American Journal of Anatomy, 151(4): 539–555.
Santos-Ruiz, L., Santamaría, J. A., Ruiz-Sánchez, J., and Becerra, J., 2002. Cell proliferation during blastema formation in the regenerating teleost fin. Developmental Dynamics, 223(2): 262–272.
Sköld, M., and Rosenberg, R., 1996. Arm regeneration frequency in eight species of Ophiuroidea (Echinodermata) from European sea areas. Journal of Sea Research, 35: 353–362.
Stocum, D. L., 2004. Amphibian regeneration and stem cells. Current Topics in Microbiology and Immunology, 280: 1–70.
Thorndyke, M. C., Chen, W. C., Beesley, P. W., and Patruno, M., 2001. Molecular approach to echinoderm regeneration. Microscopy Research and Technique, 55(6): 474–485.
Thorndyke, M. C., Chen, W. C., Moss, C., Candia Carnevali, M. D., and Bonasoro, F., 1999. Regeneration in echinoderms: cellular and molecular aspects. In: Echinoderm Research. Candia Carnevali, M. D., and Bonasoro, F., eds., Balkema, Rotterdam, 159–164.
Vickery, M. C., Vickery, M. S., Amsler, C. D., and McClintock, J. B., 2001a. Regeneration in echinoderm larvae. Microscopy Research and Technique, 55(6): 464–473.
Vickery, M. C., Vickery, M. S., McClintock, J. B., and Amsler, C. D., 2001b. Utilization of a novel deuterostome model for the study of regeneration genetics: molecular cloning of genes that are differentially expressed during early stages of larval starfish regeneration. Gene, 262(1–2): 73–80.
Willmer, P., 1990. Invertebrate Relationships: Patterns in Animal Evolution. Cambridge University Press, Cambridge, 400pp.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Fan, T., Fan, X., Du, Y. et al. Patterns and cellular mechanisms of arm regeneration in adult starfish Asterias rollestoni bell. J. Ocean Univ. China 10, 255–262 (2011). https://doi.org/10.1007/s11802-011-1837-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11802-011-1837-y