Abstract
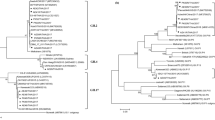
We isolated 4 Norwalk-like viruses (NLVs) contaminated oysters from 33 Chinese oysters collected from local commercial sources of Shandong Province. After amplification of the RNA-dependent RNA polymerase (RdRp) region of NLVs genomes with RT-PCR, the open reading frame 1 (ORF1) of the RdRp was sequenced and subjected to multiple-sequence alignment. The results showed that NLVs in the four isolates belong to genogroup II. The sequence comparison showed that the similarity between four Chinese oyster isolates were higher than 99.0%, which indicated that NLVs prevalent in close areas have high homogeneity in genome sequences. In addition, the most conserved sequences between diverse NLVs were used to design primers and TaqMan probes, then the real-time quantitative PCR assay was performed. According to the standard curve of GII NLVs, the original amounts (copies) of NLVs in positive patient’s fecal isolate, positive Japanese oyster isolate, and the Chinese oyster isolate were 8.9×108, 1.25×108 and 4.7×101 respectively. The detecting limit of NLVs was 1×101 copies. This study will be helpful for routine diagnosis of NLVs pathogens in foods and thus for avoiding food poisoning in the future.
Similar content being viewed by others
References
Ando, T., M. N. Mulders, D. C. Lewis, M. K. Estes, S. S. Monroe, and R. I. Glass, 1994. Comparison of the polymerase region of small round structured virus strains previously classified in three antigenic types by solid-phase immune electron microscopy. Arch. Virol., 135: 217–226.
Atomer, R. L., and M. K. Estes, 2001. Diagnosis of noncultivatable gastroenteritis viruses, the human caliciviruses. Clin. Microbiol. Rev., 14: 15–37.
Blacklow, N. R., G. Cukor, M. K. Bedigian, P. Echeverria, H. B. Greenberg, D. S. Schreiber, et al., 1979. Immune response and prevalence of antibody to Norwalk enteritis virus as determined by radioimmunoassay. J. Clin. Microbiol., 10: 903–909.
Chen, Z. Q., X. S. Chen, L. Luo, and B. Y. Ma, 2003. An epidemiological investigation and analysis for population viral incident caused by Norwalk-like virus. J. Trop. Med., 4: 190–192 (in Chinese with English abstract).
Dai, Y. C., J. Nie, Y. Liu, Z. F. Li, Y. Bai, J. D. Li, et al., 2004. Identification of two new strains of NLVs in Guangzhou. J. First Mil. Med. Univ., 24: 1016–1022 (in Chinese with English abstract).
Dolin, R., K. D. Roessner, J. J. Treanor, R. C. Reichman, M. Phillips, and H. P. Madore, 1986. Radioimmunoassay for detection of the Snow Mountain agent of viral gastroenteritis. J. Med. Virol., 19:11–18.
Fang, Z. Y., L. Y. Wen, S. J. Jin, and Z. H. Zhao, 1995. Detection of Norwalk-like viruses in stool specimens from children with diarrhea in China. Clin. J. Virol., 11: 215–219 (in Chinese with English abstract).
Green, S. M., P. R. Lambden, E. O. Caul, and I. N. Clarke, 1997. Capsid sequence diversity in small round structured viruses from recent UK outbreaks of gastroenteritis. J. Med. Virol., 52: 14–19.
Greenberg, H. B., and A. Z. Kapikian, 1978. Detection of Norwalk agent antibody and antigen by solid-phase radioimmunoassay and immune adherence hemagglutination assay. J. Am. Vet. Med. Assoc., 173: 620–623.
Hafliger, D., M. Gilgen, J. Luthy, and P. Hubner, 1997. Seminested RT-PCR systems for small round structured viruses and detection of enteric viruses in seafood. Intl. J. Food Microbiol., 37: 27–36.
Jiang, X., D. W. Cubitt, T. Berke, W. M. Zhong, X. M. Dai, L. K. Pickering, et al., 1997. Sapporo-like human caliciviruses are genetically and antigenically diverse. Arch. Virol., 142: 1813–1827.
Jiang, X., P. W. Huang, W. M. Zhong, T. Farkas, D. W. Cubitt, and D. O. Matson, 1999. Design and evaluation of a primer pair that detects both Norwalk-and Sapporo-like caliciviruses by RT-PCR. J. Virol. Methods, 83: 145–154.
Kageyama, T., S. Kojima, M. Shinohara, K. Uchida, S. Fukushi, F. B. Hoshino, et al., 2003. Broadly reactive and highly sensitive assay for Norwalk-like viruses based on realtime quantitative reverse transcription-PCR. J. Clin. Microbiol., 41: 1548–1557.
Katayama, K., H. Shirato-Horikoshi, S. Kojima, T. Kageyama, T. Oka, F. B. Hoshina, et al., 2002. Phylogenic analysis of the complete genome of 18 Norwalk-like viruses. Virology, 299: 225–239.
Kawamoto, H., K. Yamazaki, E. Utagawa, and T. Ohyama, 2001. Nucleatide sequence analysis and development of consensus primers of RT-PCR for detection of norwalk-like viruses prevailing in Japan. J. Med. Virol., 64: 569–576.
Lees, D. N., K. Henshilwood, and W. J. Dore, 1994. Development of a method for detection of enteroviruses in shellfish by PCR with poliovirus as a model. Appl. Environ. Microbiol., 60: 2999–3005.
Loisy, F., R. L. Atmar, P. Guillon, P. Le Cann, M. Pommepuy, and F. S. Le Guyader, 2005. Real-time RT-PCR for norovirus screening in shellfish. J. Virol. Methods, 123: 1–7.
Lü, H. X., Z. Y. Fang, H. P. Xie, J. Y. Tang, H. K. Hu, L. S. Zhang, et al., 2003. Epidemiological study of human caliciviruses among children with acute diarrhea in Lulong county, 1999–2001. Clin. J. Epidemiol., 24: 1118–1121 (in Chinese with English abstract).
McGoldrick, A., E. Bensaude, G. Ibata, G. Sharp, and D. J. Paton, 1999. Closed one-tube reverse transcription nested polymerase chain reaction for the detection of pestiviral RNA with fluorescent probes. J. Virol. Methods, 79: 85–95.
Moe, C. L., J. Gentsch, T. Ando, G. Grohmann, S. S. Monroe, X. Jiang, et al., 1994. Application of PCR to detect Norwalk virus in fecal specimens from outbreaks of gastroenteritis. J. Clin. Microbiol., 32: 642–648.
Saito, H., S. Saito, K. Kamada, S. Harata, H. Sato, M. Morita, et al., 1998. Application of RT-PCR designed from the sequence of the local SRSV strain to the screening in viral gastroenteritis outbreaks. Microbiol. Immunol., 42: 439–446.
Treanor, J., R. Dolin, and H. P. Madore, 1988. Production of a monoclonal antibody against the Snow Mountain agent of gastroenteritis by in vitro immunization of murine spleen cells. Proc. Natl. Acad. Sci. USA, 85: 3613–3617.
Vinje, J., R. A. Hamidjaja, and M. D. Sobsey, 2004. Development and application of a capsid VP1 (region D) based reverse transcription PCR assay for genotyping of genogroup I and II noroviruses. J. Virol. Methods, 116: 109–117.
Vinje, J., and M. P. Koopmans, 1996. Molecular detection and epidemiology of small round-structured viruses in outbreaks of gastroenteritis in The Netherlands. J. Infect. Dis., 174: 610–615.
Wang, J., X. Jiang, H. P. Madore, J. Gray, U. Desselberger, T. Ando, et al., 1994. Sequence diversity of small, round structured viruses in the Norwalk virus group. J. Virol., 68: 5982–5990.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, J., Tang, Q., Yue, Z. et al. Sequence analysis and quantitative detection of Norwalk-like viruses in cultured oysters of China. J. Ocean Univ. China 7, 223–227 (2008). https://doi.org/10.1007/s11802-008-0223-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11802-008-0223-x