Abstract
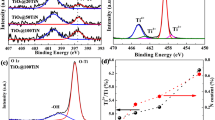
Titanium dioxide is coated on the surface of MCM-41 wafer through the plasma enhanced chemical vapor deposition (PECVD) method using titanium isopropoxide (TTIP) as a precursor. Annealing temperature is a key factor affecting crystal phase of titanium dioxide. It will transform an amorphous structure to a polycrystalline structure by increasing temperature. The optimum anatase phase of TiO2 which can acquire the best methanol conversion under UV-light irradiation is obtained under an annealing temperature of 700°C for 2 h, substrate temperature of 500°C, 70 mL·min−1 of oxygen flow rate, and 100W of plasma power. In addition, the films are composed of an anatase-rutile mixed phase, and the ratio of anatase to rutile varies with substrate temperature and oxygen flow rate. The particle sizes of titanium dioxide are between 30.3 nm and 59.9 nm by the calculation of Scherrer equation. Under the reaction conditions of 116.8 mg·L-1 methanol, 2.9 mg·L−1 moisture, and 75°C of reaction temperature, the best conversion of methanol with UV-light is 48.2% by using the anatase-rutile (91.3/8.7) mixed phase TiO2 in a batch reactor for 60 min. While under fluorescent light irradiation, the best photoactivity appears by using the anatase-rutile (55.4/44.6) mixed phase TiO2 with a conversion of 40.0%.
Similar content being viewed by others
References
Euvananont C, Junin C, Inpor K, Limthongkul P, Thanachayanont C. TiO2 optical coating layers for self-cleaning applications. Ceramics International, 2008, 34(4): 1067–1071
Dholam R, Patel N, Adami M, Miotello A. Physically and chemically synthesized TiO2 composite thin films for hydrogen production by photocatalytic water splitting. International Journal of Hydrogen Energy, 2008, 33(23): 6896–6903
Ghezzar M R, Abdelmalek F, Belhadj M, Benderdouche N, Addou A. Enhancement of the bleaching and degradation of textile wastewaters by Gliding arc discharge plasma in the presence of TiO2 catalyst. Journal of Hazardous Materials, 2009, 164(2–3): 1266–1274
Suhail M H, Rao G M, Mohan S. DC reactive magnetron sputtering of titanium-structural and optical characterization of TiO2 films. Journal of Applied Physics, 1992, 71(3): 1421–1427
Takahashi Y, Suzuki H, Nasu M. Rutile growth at the surface of TiO2 films deposited by vapour-phase decomposition of isopropyl titanate. Journal of the Chemical Society, Faraday Transactions 1: Physical Chemistry in Condensed Phases, 1985, 81(12): 3117–3125
Lee W G, Woo S I, Kim J C, Choi S H, Oh K H. Preparation and properties of amorphous TiO2 thin films by plasma enhanced chemical vapor deposition. Thin Solid Films, 1994, 237(1–2): 105–111
Lee Y H. A role of energetic ions in RF-biased PECVD of TiO2. Vacuum, 1999, 51(4): 503509
Battiston G A, Gerbasi R, Gregori A, Porchia M, Cattarin S, Rizzi G A. PECVD of amorphous TiO2 thin films: effect of growth temperature and plasma gas composition. Thin Solid Films, 2000, 371(1–2): 126–131
Hsiao L C. Develop a extraction method of novel materials which are applied on Volatile Organic Compounds in air. Dissertation for the Master Degree. Taoyuan: Central University, 2005 (in Chinese)
Sadjadi M S, Farhadyar N, Zare K. Synthesis of nanosize MCM-41 loaded with TiO2 and study of its photocatalytic activity. Superlattices and Microstructures, 2009, 46(1–2): 266–271
Signoretto M, Ghedini E, Trevisan V, Bianchi C L, Ongaro M, Cruciani G. TiO2-MCM-41 for the photocatalytic abatement of NOx in gas phase. Applied Catalysis B: Environmental, 2010, 95(1–2): 130–136
Grill A. Cold Plasma in Materials Fabrication. 1st ed. Wiley-IEEE Press, 1994, 1–23
Chou H P. Structure and photocatalysis of undoped and nitrogendoped titanium oxide films by reactive sputtering. Dissertation for the Master Degree. Taitung: Dong Hwa University, 2004 (in Chinese)
Mwabora J M, Lindgren T, Avendaño E, Jaramillo T F, Lu J, Lindquist S E, Granqvist C G. Structure, composition, and morphology of photoelectrochemically active TiO2 − xNx thin films deposited by reactive DC magnetron sputtering. Journal of Physical Chemistry B, 2004, 108(52): 20193–20198
Zhang Q H, Gao L, Guo J K. Effects of sulfate ions and hydrolytic temperature on the properties of TiCl4-derived nanostructured TiO2. Journal of Inorganic Materials, 2000, 15(6): 992–998 (in Chinese)
Liqiang J, Xiaojun S, Weimin C, Zili X, Yaoguo D, Honggang F. The preparation and characterization of nanoparticle TiO2/Ti films and their photocatalytic activity. Journal of Physics and Chemistry of Solids, 2003, 64(4): 615–623
McCurdy P R, Sturgess L J, Kohli S, Fisher E R. Investigation of the PECVD TiO2-Si(100) interface. Applied Surface Science, 2004, 233(1–4): 69–79
Herman G S, Dohnalek Z, Ruzycki N, Diebold U. Experimental investigation of the interaction of water and methanol with anatase-TiO2(101). Journal of Physical Chemistry B, 2003, 107(12): 2788–2795
Bickley R I, Gonzalez-Carreno T, Lees J S, Palmisano L, Tilley R J D. A structural investigation of titanium dioxide photocatalysts. Journal of Solid State Chemistry, 1991, 92(1): 178–190
Serpone N, Maruthamuthu P, Pichat P, Pelizzetti E, Hidaka H. Exploiting the interparticle electron transfer process in the photocatalysed oxidation of phenol, 2-chlorophenol and pentachlorophenol: chemical evidence for electron and hole transfer between coupled semiconductors. Journal of Photochemistry and Photobiology A Chemistry, 1995, 85(3): 247–255
Yan Q Z, Su X T, Zhou Y P, Ge C C. Controlled synthesis of TiO2 nanometer powders by sol-gel auto-igniting process and their structural property. Acta Physico-Chimica Sinica, 2005, 21(1): 57–62 (in Chinese)
Lee K B. Photocatalytic degradation of methanol gas with TiO2 thin films by plasma enhanced CVD. Dissertation for the Master Degree. Taichung: Chung Hsing University, 2006 (in Chinese)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, S., Wang, K., Jehng, J. et al. Preparation of TiO2/MCM-41 by plasma enhanced chemical vapor deposition method and its photocatalytic activity. Front. Environ. Sci. Eng. 6, 304–312 (2012). https://doi.org/10.1007/s11783-010-0297-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11783-010-0297-8