Abstract
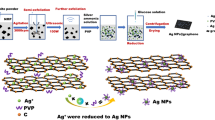
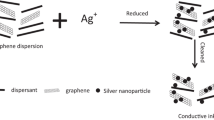
Finer nanoplates of silver are prepared by self-assembly on the surface of graphene, and the low-temperature sintered high conductivity ink containing the silver nanoplates is prepared. Most importantly, graphene is added to the solution before the chemical reduction reaction occurs. Firstly, it is found that silver nanoplates have self-assembly phenomenon on the surface of graphene. Secondly, the Ag nano hexagonal platelets (AgNHPs) with small particle sizes (10 nm), narrow distribution and good dispersion are prepared. Especially, smaller sizes (10 nm) and narrower particle size distribution of AgNHPs particles can be easily controlled by using this process. Finally, the conductivity of the ink is excellent. For example, when the printed patterns were sintering at 150 °C, the resistivity of the ink(GE: 0.15 g/L) reached the minimum value of 2.2×10−6 Ω·cm. And the resistivity value was 3.7×10−6 Qcm, when it was sintered at 100 °C for 30 min. The conductive ink prepared can be used for the field of printing electronics as ink-jet printing ink.
摘要
采用石墨烯诱导, 制备出六角片状纳米银, 并最终制备出在低烧结温度下导电性能好的纳米银 墨水. 其中核心之处是将石墨烯以原料的方式提前加入反应液中制备纳米银颗粒. 研究发现, 制备过 程中纳米银颗粒在石墨烯表面具有自组装现象. 其次, 制备出的六角片状纳米银颗粒粒径小(10 nm)、 分布窄、分散性好. 特别是, 本文采用的制备粒径小、分布窄的纳米银片的工艺更容易控制, 更有利 于批量生产. 最后, 制备的石墨烯-纳米银混合导电墨水具有优良的导电性. 如, 当石墨烯含量为 0.15 g/L, 在150 °C 烧结后, 印刷制成的导电线路的电阻率达到最小值为2.2×10−6 Ω·cm. 在100 °C 烧 结30 min 后, 电阻率为3.7×10−6 Ω·cm. 制备的混合导电墨水可用于印刷电子喷墨打印领域.
Similar content being viewed by others
References
NGE T T, NOGI M, SUGNUMA K. Electrical functionality of inkjet-printed silver nanoparticle conductive tracks on nanostructured paper compared with those on plastic substrates [J]. Journal of Materials Chemistry C, 2013, 1(34): 5235–5243.
XU L, LI H, XIA J, WANG L, XU H. Graphitic carbon nitride nanosheet supported high loading silver nanoparticle catalysts for the oxygen reduction reaction [J]. Materials Letters, 2014, 128(6): 349–353. DOI: https://doi.org/10.1016/j.matlet.2014.04.110.
PANDEY P A, BELL G R, ROURKE J P, WILSON N. Physical vapor deposition of metal nanoparticles on chemically modified graphene: Observations on metal-graphene interactions [J]. Small, 2011, 7(22): 3202–3210. DOI: https://doi.org/10.1002/smll.201101430.
AN B, CAI X H, WU F S, WU Y P. Preparation of micro-sized and uniform spherical Ag powders by novel wet-chemical method [J]. Transactions of Nonferrous Metals Society of China, 2010, 20: 1550–1554. DOI: https://doi.org/10.1016/S1003-6326(09)60337-X.
JOHNSON H, AROCKIASAMY A, GANESAN S, KANNUSAMY M. A new approach for deposition of silver film from AgCl through successive ionic layer adsorption and reaction technique [J]. Journal of Central South University, 2017, 24: 2793–2798. DOI: https://doi.org/10.1007/s11771-017-3693-4.
LIU P, MA J, DENG S, ZENG K, DENG D Y, XIE W, LU A X. Graphene-Ag nanohexagonal platelets-based ink with high electrical properties at low sintering temperatures [J]. Nanotechnology, 2016, 27: 385603. DOI: https://doi.org/10.1088/0957-4484/27/38/385603.
CHI K, ZHANG Z, XI J, HUANG Y, XIAO F, WANG S, LIU Y. Freestanding graphene paper supported three-dimensional porous graphene-polyaniline nanocomposite synthesized by inkjet printing and in flexible all-solid-state supercapacitor [J]. ACS Applied Materials & Interfaces, 2014, 6: 16312. DOI: https://doi.org/10.1039/C4TA02721C.
XIAO F, YANG S X, ZHANG Z Y, LIU H F, XIAO J W, WAN L, LUO J, WANG S, LIU Y Q. Scalable synthesis of freestanding sandwich-structured graphene/polyaniline/graphene nanocomposite paper for flexible all-solid-state supercapacitor [J]. Scientific Reports, 2015, 5: 9359. DOI: https://doi.org/10.1038/srep09359.
SUN Y G, XIA Y N. Shape-controlled synthesis of gold and silver nanoparticles [J]. Science, 2002, 298(5601): 2176–2179. DOI: https://doi.org/10.1126/science.1077229.
GAO Y, JIANG P, SONG L, WANG J X, LIU L F, LIU D F, XIANG Y J, ZHANG Z X, ZHAO X W, DOU X Y, LUO S D, ZHOU W Y, XIE S S. Studies on silver nanodecadrons synthesized by PVP-assisted N,N-dimethylformamide (DMF) reduction [J]. Journal of Crystal Growth, 2006, 289: 376–380. DOI: https://doi.org/10.1016/j.jcrysgro.2005.11.123.
WILEY B J, XIONG Y J, LI Z Y, Y Y D, XIA Y N. Right bipyramids of silver: A new shape derived from single twinned seeds [J]. Nano Lett, 2006, 6: 765–768. DOI: https://doi.org/10.1021/nl060069q.
GILBERTSON L M, ZIMMERMAN J B, PLATA D L, HUTCHISON J E, ANASTAS P T. Designing nanomaterials to maximize performance and minimize undesirable implications guided by the principles of green chemistry [J]. Journal of Materials Chemistry C, 2015, 44: 5758–5777. DOI: https://doi.org/10.1039/c4cs00445k.
ISABEL P S, LUIS M L M. Synthesis of silver nanoprisms in DMF [J]. Nano Letters, 2002, 2(8): 903–905. DOI: https://doi.org/10.1021/nl025638i.
BAI L, MA X J, LIU J F, SUN X M, ZHAO D Y, EVANS D G. Rapid separation and purification of nanoparticles in organic density gradients [J]. Journal of the American Chemical Society, 2010, 132(7): 2333–2340. DOI: https://doi.org/10.1021/ja908971d.
LIU P, TANG Q Z, LIU H, LU A X. Low electrical resistivity of graphene-AgNHPs based ink with a new processing method [J]. RSC Advances, 2017, 7: 15228–15235. DOI: https://doi.org/10.1039/c7ra00309a.
XU Z, GAO H, HU G. Solution-based synthesis and characterization of a silver nanoparticle-graphene hybrid film [J]. Carbon, 2011, 49: 4731–4738. DOI: https://doi.org/10.1016/j.jcis.2011.05.009.
LIU Y, CHANG Q, HUANG L. Transparent, flexible conducting graphene hybrid films with a subpercolating network of silver nanowires [J]. Journal of Materials Chemistry C, 2013, 1: 2970–2974. DOI: https://doi.org/10.1039/C3TC30178H.
NERSISYAN H H, LEE J H, SON H T, WON C W, MAENG D Y. A new and effective chemical reduction method for preparation of nanosized silver powder and colloid dispersion [J]. Materials Research Bulletin, 2003, 38(6): 949–956. DOI: https://doi.org/10.1016/S0025-5408(03)0078-3.
CHUN K Y, OH Y, RHO J, AHN J H, KIM Y J, CHOI H R, BAIK S. Highly conductive, printable and stretchable composite films of carbon nanotubes and silver [J]. Nature Nanotechnology, 2010, 5: 853–857. DOI: https://doi.org/10.1038/NNANO.2010.232.
CHARCOSSET C, KIEFFER R, MANGIN D, PUEL F. Coupling between membrane processes and crystallization operations [J]. Industrial & Engineering Chemistry Research, 2010, 49(12): 5489–5495. DOI: https://doi.org/10.1021/ie901824x.
KIM T Y, KWON S W, PARK S J, YOON D H, SUH K S, YANG W S. Self-organized graphene patterns [J]. Advanced Materials, 2011, 23: 2734–2738. DOI: https://doi.org/10.1002/adma.201100329.
KIM Y H, YOO B, ANTHONY J E, PARK S K. Controlled deposition of a high performance small molecule organic single crystal transistor array by direct ink jet printing [J]. Advanced Materials, 2012, 24(4): 497–502. DOI: https://doi.org/10.1002/adma.201103032.
HE Yuan, LIU Yun-guo. Direct fabrication of highly porous grapheme/TiO2 composite nanofibers by electrospinning for photocatalytic application [J]. Journal of Central South University, 2018, 25: 2182–2189. DOI: https://doi.org/10.1007/s11771-018-3906-5.
ZHANG L, LIU H, ZHAO Y, SUN X, WEN Y, GUO Y, GAO X, DI C, YU G, LIU Y. Inkjet printing high-resolution, large-area graphene patterns by coffee-ring lithography [J]. Advanced Materials, 2012, 24(3): 436–440. DOI: https://doi.org/10.1002/adma.201103620.
LIU J H, HU Y, ZHANG Y W, CHEN W. Graphenestabilized silver nanoparticle electrochemical electrode for actuator design [J]. Advanced Materials, 2013, 25: 1270–1274. DOI: https://doi.org/10.1002/adma.201203655.
BADAWY A M E, LUXTON T P, SILVA R G, SCHECKEL K G, SUIDAN M T, TOLAYMAT T M. Impact of environmental conditions (pH, ionic strength, and electrolyte type) on the surface charge and aggregation of silver nanoparticles suspensions [J]. Environmental Science & Technology, 2010, 44(4): 1260–1266. DOI: https://doi.org/10.1021/es902240k.
DONG X Y, GAO Z W, YANG K F, ZHANG W Q, XU L W. Nanosilver as a new generation of silver catalysts in organic transformations for efficient synthesis of fine chemicals [J]. Journal of Materials Chemistry, 2015, 5: 2554–2574. DOI: https://doi.org/10.1039/C5CY00285K.
LU G, LI H, LIUSMAN C, YIN Z, WU S, ZHANG H. Surface enhanced Raman scattering of Ag or Au nanoparticle-decorated reduced graphene oxide for detection of aromatic molecules [J]. Chemical Science, 2011, 2: 1817–1821. DOI: https://doi.org/10.1039/C1SC00254F.
LEE J, SHIM S, KIM B, SHIN H S. Surface-enhanced Raman scattering of single- and few-layer graphene by the deposition of gold nanoparticles [J]. Chemistry-A European Journal, 2011, 17: 2381–2387. DOI: https://doi.org/10.1002/chem.201002027.
ISRAELACHVILI J. Intermolecular & surface force [M]. London: Academic Press, 1997.
XU Y X, WU Q, SUN Y, BAI H, SHI G. Three-dimensional self-assembly of graphene oxide and DNA into multifunctional hydrogels [J]. ACS Nano, 2010, 4(10): 7358–7362. DOI: https://doi.org/10.1021/nn1027104.
SCHWEINSBERG D P, HOPE G A, TRUEMAN A, OTIENO-ALEGO V. An electrochemical and SERS study of the action of polyvinylpyrrolidone and polyethylenimine as inhibitors for copper in aerated H2SO4 [J]. Corrosion Science, 1996, 38(4): 587–599.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, P., He, Wq. & Lu, Ax. Preparation of low-temperature sintered high conductivity inks based on nanosilver self-assembled on surface of graphene. J. Cent. South Univ. 26, 2953–2960 (2019). https://doi.org/10.1007/s11771-019-4227-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11771-019-4227-z