Abstract
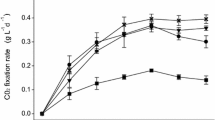
The easy acidification and high hydrogen sulfide (H2S) production during anaerobic digestion of macroalgae limited its application in biomethane production. In order to investigate the effects of ceramsite on methane and H2S productions during anaerobic digestion of macroalgae, batch experiments of Macrocystis pyrifera were carried out. Four groups named C0, C1, C2 and C3 added with 0, 1.5, 3.0 and 4.5 g/gsubstrate of ceramsite, respectively, were studied and compared. The highest cumulative methane yield of 286.3 mL/gsubstrate is obtained in C2, which is 40.11% higher than that of C0. The cumulative H2S yields of C1, C2 and C3 are 32.67%, 44.66% and 53.21% lower than that of C0, respectively. Results indicate that ceramsite addition permits higher methane yields, shorter lag-phase time and lower H2S yields during anaerobic digestion of Macrocystis pyrifera.
摘要
巨藻厌氧发酵过程存在易酸化、硫化氢产量高的问题,大大限制了其在生物制甲烷方面的应用。 为了研究陶粒对巨藻厌氧发酵的甲烷和硫化氢生产的影响,开展了以巨藻Macrocystis pyrifera 生物质 为底物的批次发酵试验。实验分4 组,C0、C1、C2 和C3,分别添加了0、1.5、3.0、4.5 g/gsubstrate 的 陶粒。其中,C2 组得到了最高的甲烷累积产量,为286.3 mL/gsubstrate,比C0 组提高了40.11%。C1、 C2、C3 组的硫化氢累积产量分别比C0 降低了32.67%、44.66%、53.21%。结果显示,添加陶粒的巨 藻发酵体系甲烷产量提高、延滞期缩短、硫化氢产量降低。
Similar content being viewed by others
References
GOYAL H B, SEAL D, SAXENA R C. Bio-fuels from thermochemical conversion of renewable resources: A review [J]. Renewable and Sustainable Energy Reviews, 2008, 12(2): 504–517.
DEMIRBAS A, DEMIRBAS M F. Importance of algae oil as a source of biodiesel [J]. Energy Conversion and Management, 2011, 52(1): 163–170.
KONDA N V S N M, SINGH S, SIMMONS B A, KLEINMARCUSCHAMER D. An investigation on economic feasibility of macroalgae as a potential feedstock for biorefineries [J]. Bioenergy Research, 2015, 8(3): 1046–1056.
GHPSH S, KLASS D L, CHYNOWETH D P. Bioconversion of macrocystis pyrifera to methane [J]. Journal of Chemical Technology and Biotechnology, 1981, 31(12): 791–807.
CHEN Hui-hui, ZHOU Dong, LUO Gang, ZHANG Shi-cheng, CHEN Jian-min. Macroalgae for biofuels production: Progress and perspectives [J]. Renewable and Sustainable Energy Reviews, 2015, 47: 427–437.
DEBOWSKI M, ZIELINSKI M, GRALA A, DUDEK M. Algae biomass as an alternative substrate in biogas production technologies—Review [J]. Renewable and Sustainable Energy Reviews, 2013, 27: 596–604.
JARD G, JACKOWIAK D, CARRERE H, DELGENES J P, TORRIJOS M, STEYER J P, DUMAS C. Batch and semi-continuous anaerobic digestion of Palmaria palmata: Comparison with Saccharina latissima and inhibition studies [J]. Chemical Engineering Journal, 2012, 209: 513–519.
FAN Xiao-lei, GUN Rong-bo, YUAN Xian-zheng, QIU Yan-ling, YANG Zhi-man, WANG Fei, SUN Meng-ting, ZHAO Xiao-xian. Biogas production from Macrocystis pyrifera biomass in seawater system [J]. Bioresource Technology, 2015, 197: 339–347.
YAZDANI P, ZAMANI A, KARIMI K, TAHERZADEH M J. Characterization of Nizimuddinia zanardini macroalgae biomass composition and its potential for biofuel production [J]. Bioresource Technology, 2015, 176: 196–202.
BRUHN A, DAHL J, NIELSEN H B, NIKOLAISEN L, RASMUSSEN M B, MARKAGER S, OLESEN B, ARIAS C, JENSEN P D. Bioenergy potential of Ulva lactuca: Biomass yield, methane production and combustion [J]. Bioresource Technology, 2011, 102(3): 2595–2604.
MONTINGELLIA M E, TEDESCO S, OLABI A G. Biogas production from algal biomass: A review [J]. Renewable and Sustainable Energy Reviews, 2015, 43: 961–972.
GHADIRYANFAR M, ROSENTRATER K A, KEYHANI A, OMID M. A review of macroalgae production, with potential applications in biofuels and bioenergy [J]. Renewable and Sustainable Energy Reviews, 2016, 4: 473–481.
HIGGINS M J, CHEN Y C, YAROSZ D P, MURTHY S N, MAAS N A, GLINDEMANN D, NOVAK J T. Cycling of volatile organic sulfur compounds in anaerobically digested biosolids and its implications for odors [J]. Water Environment Research, 2006, 78(3): 243–252.
DU Wei-wei, PARKER W J. Behavior of volatile sulfur compounds in mesophilic anaerobic digestion [J]. Water Environment Federation, 2009, 12(1): 3963–3979.
BELLE A J, LANSING S, MULBRY W, WEIL R R. Methane and hydrogen sulfide production during co-digestion of forage radish and dairy manure [J]. Biomass and Bioenergy, 2015, 80: 44–51.
ABBOTT T, ESKICIOGLU C. Effects of metal salt addition on odor and process stability during the anaerobic digestion of municipal waste sludge [J]. Waste Management, 2015, 46: 449–458.
CHEN Ye, CHENG J J, CREAMER K S. Inhibition of anaerobic digestion process: A review [J]. Bioresource Technology, 2008, 99(10): 4044–4064.
HAO Xiao-yan, HOU Guang-ying, ZHENG Pei-xue, LIU Ru-tao, LIU Chun-guang. H2S in-situ removal from biogas using a tubular zeolite/TiO2 photocatalytic reactor and the improvement on methane production [J]. Chemical Engineering Journal, 2016, 294: 105–110.
JIA Li-juan, NING Ping, WANG Xiang-yu, QU Guang-fei, XIONG Xiang-feng. Dynamics of liquid-phase catalytic oxidation of hydrogen sulfide removal in rural biogas [J]. Journal of Central South University, 2014, 21(7): 2843–2847.
MILAN Z, SANCHEZ E, WEILAND P, BORJA R, MARTIN A, ILANGOVAN K. Influence of different natural zeolite concentrations on the anaerobic digestion of piggery waste [J]. Bioresource Technology, 2001, 80(1): 37–43.
WATANABE R, TADA C, BABA Y, FUKUDA Y, NAKAI Y. Enhancing methane production during the anaerobic digestion of crude glycerol using Japanese cedar charcoal [J]. Bioresource Technology, 2013, 150: 387–392.
COSOLI P, FERRONE M, PRICL S, FERMEGLIA M. Hydrogen sulphide removal from biogas by zeolite adsorption, Part I. GCMC molecular simulations [J]. Chemical Engineering Journal, 2008, 145(1): 86–92.
CHEN Yi-qing, FAN Zhi-dong, MA Li-xia, YIN Juan, LUO Man, CAI Wang-feng. Performance of three pilot-scale immobilized-cell biotrickling filters for removal of hydrogen sulfide from a contaminated air steam [J]. Saudi Journal of Biological Sciences, 2014, 21(5): 450–456.
JIANG An-xi, ZHAO Yu-xin, XU Gui-qin, LI Li, MA Li. Evolution of air biofilter for H2S and NH3 biodegradation [J]. Journal of Science of Heilongjiang University, 2003, 20(1): 92–95. (in Chinese)
YUAN Xian-zheng, SHI Xiao-shuang, ZHANG Pei-song, WEI Yue-li, GUO Rong-bo, WANG Li-sheng. Anaerobic biohydrogen production from wheat stalk by mixed microflora: Kinetic model and particle size influence [J]. Bioresource Technology, 2011, 102(19): 9007–9012.
DUBOIS M, GILLES K A, HAMILTON JK, REBERS P A, Smith F. Colorimetric method for determination of sugars and related substances [J]. Analytical Chemistry, 1956, 28(3): 350–356.
SAFI C, CHARTON M, PIGNOLET O, SILVESTRE F, VACA-GARCIA C, PONTALIER P Y. Influence of microalgae cell wall characteristics on protein extractability and determination of nitrogen-to-protein conversion factors [J]. J Appl Phycol, 2013, 25(2): 523–529.
HE Shuai, FAN Xiao-lei, KATUKURI N R, YUAN Xian-zheng, WANG Fei, GUO Rong-bo. Enhanced methane production from microalgal biomass by anaerobic bio-pretreatment [J]. Bioresource Technology, 2016, 204: 145–151.
HUILINIR C, QUINTRIQUEO A, ANTILEO C, MONTALVO S. Methane production from secondary paper and pulp sludge: Effect of natural zeolite and modeling [J]. Chemical Engineering Journal, 2014, 257: 131–137.
GUERRERO L, MONTALVO S, HUILINIR C, CAMPOS J L, BARAHONA A, BORJA R. Advances in the biological removal of sulphides from aqueous phase in anaerobic processes: A review [J]. Environmental Reviews, 2016, 24(1): 84–100.
BORJA R, BANKS C J. Semicontinuous anaerobic digestion of soft drink wastewater in immobilised cell bioreactors [J]. Biotechnology Letters, 1993, 15(7): 767–772.
ZHAI Ning-ning, ZHANG Tong, YIN Dong-xue, YANG Gai-he, WANG Xiao-jiao, REN Guang-xin, FENG Yong-zhong. Effect of initial pH on anaerobic co-digestion of kitchen waste and cow manure [J]. Waste Management, 2015, 38: 126–131.
ZHANG Tong, MAO Chun-lan, ZHAI Ning-ning, WANG Xiao-jiao, YANG Gai-he. Influence of initial pH on thermophilic anaerobic co-digestion of swine manure and maize stalk [J]. Waste Management, 2015, 35: 119–126.
KIM M, GOMEC C Y, AHN Y, SPEECE R E. Hydrolysis and acidogenesis of particulate organic material in mesophilic and Thermophilic anaerobic digestion [J]. Environmental technology, 2003, 24(9): 1183–1190.
BABEL S, FUKUSHI K, SITANRASSAMEE B. Effect of acid speciation on solid waste liquefaction in an anaerobic acid digester [J]. Water Research, 2004, 38(9): 2417–2423.
DOGAN T, INCE O, OZ N A, INCE B K. Inhibition of volatile fatty acid production in granular sludge from a UASB reactor [J]. Journal of Environmental Science and Health, 2005, 40(3): 633–644.
ROMERO-GUIZA M S, VILA J, MATA-ALVAREZ J, CHIMENOS J M, ASTALS S. The role of additives on anaerobic digestion: A review [J]. Renewable and Sustainable Energy Reviews, 2016, 58: 1486–1499.
MONTALVO S, GUERRERO L, BORJA R, SANCHEZ E, MILAN Z, CORTES I, ANGELES DE LA LA RUBIA M. Application of natural zeolites in anaerobic digestion processes: A review [J]. Applied Clay Science, 2012, 58: 125–133.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: Project(2014BAC31B01) supported by the National Science & Technology Support during the 12th Five-Year Plan Period, China; Projects(2015GSF117016, 2015GSF115037) supported by the Key Research & Development Project of Shandong Province, China
Rights and permissions
About this article
Cite this article
Sun, Mt., Fu, Sf., He, S. et al. Effects of ceramsite on methane and hydrogen sulphide productions from macroalgae biomass. J. Cent. South Univ. 25, 1076–1083 (2018). https://doi.org/10.1007/s11771-018-3807-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11771-018-3807-7