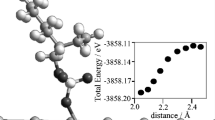
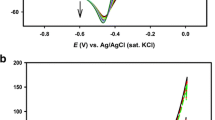
Abstract
The electrode process of diethyldithiocarbamate on the surface of pyrrhotite was studied using systematic electrochemical analysis, including cyclic voltammetry, chronopotentiometry and galvanostatic. Experimental results show that tetraethylthioram disulphide (TETD) is electrodeposited on pyrrhotite electrode surface in the presence of 1.0×10−4 mol/L diethyldithiocarbamate when the electrode potential is higher than 0.25 V. The electrochemical kinetics parameters of the electrode process of diethyldithiocarbamate on surface of pyrrhotite are calculated as follows: the exchange current density is 2.48 µA/cm2, and the transmission coefficient is 0.46. The electrodeposition includes two steps electrochemical reaction. The first reaction is electrochemical adsorption of diethyldithiocarbamate ion, then the adsorbed ion associates with a diethyldithiocarbamate ion from the solution and forms tetraethylthioram disulphide on the surface of pyrrhotite.
Similar content being viewed by others
References
Song S, Lopez-Valdivieso A, Ojeda-Escamilla M C. Electrophoretic mobility study of the adsorption of alkyl xanthate ions on galena and sphalerite[J]. Journal of Colloid and Interface Science, 2001, 237(1): 70–75.
Grano S R, Prestidge C A, Ralston J. Sulphite modification of galena surfaces and its effect on flotation and xanthate adsorption[J]. International Journal of Mineral Processing, 1997, 52(1): 1–29.
Chmielewski T, Lekki J. Electrochemical investigation on adsorption of potassium ethyl xanthate on galena [J]. Minerals Engineering, 1989, 2(3): 387–391.
Herrera-Urbina R, Sotillo F J, Fuerstenau D W. Effect of sodium of sodium sulfide additions on the pulp potential and amyl xanthate flotation of cerussite and galena[J]. International Journal of Mineral Processing, 1999, 54(3): 157–170.
Ndzebet E, Schuhmann D, Vanel P. Study of the impedance of a galena electrode under conditions similar to those used in sulphide mineral flotation—I. Electrode oxidation and xanthate adsorption[J]. Electrochimica Acta, 1994, 39 (5): 745–753.
Chermyshova I. Anodic processes on a galena (PbS) electrode in the presence of n-butyl xanthate studied FTIR-spectroelectrochemically[J]. Journal of Physical Chemistry B, 2001, 105(34): 8185–8191.
HU Qing-chun, WANG Dian-zuo, LI Bai-dan. Electrochemical investigation of the diethyldithiocarbamategalena flotation system[J]. International Journal of Mineral Processing, 1992, 47(4): 289–305.
WANG Dian-zuo, GU Guo-hua, LIU Ru-yi. Potential adjustment flotation of galena-lime-diethyldithiocarbamate system [J]. Chinese Journal of Nonferrous Metals, 1998, 8(2): 322–326. (in Chinese)
LI Quan, QIU Guan-zhou, QIN Wen-qing, et al. Kinetics of electrochemical process of pyrite electrode in diethyldithiocarbamate solution[J]. Mining and Metallurgical Engineering, 2001, 21(2): 30–33. (in Chinese)
Bhaskar R G, Forsling W. Adsorption mechanism of diethyldithiocarbamate on covellite, cuprite and tenorite[J]. Colloids and Surfaces, 1991, 60(26): 53–69.
QIN Wen-qing, QIU Guan-zhou, HU Yue-hua, et al. Dynamics of electrodeposition of tetraethylthioram disulphide (TETD) on pyrite surface[J]. Journal of Central South University of Technology(English Edition), 2001, 8(3): 164–168.
Hamilton C, Woods R. An investigate of surface oxidation of pyrite and pyrrhotite by linear potential sweep voltammetry[J]. Journal of Electroanalytical Chemistry, 1981,118:327–343.
Valli M, Persson I. A vibration and X-ray photoelectron spectroscopic study of the interaction between chalcopyrite, marcasite, pentlandite, pyrrhotite and troilite, and ethylxanthate and decylxanthate ions in aqueous solution[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 1994, 83(3): 207–217.
Author information
Authors and Affiliations
Corresponding author
Additional information
Foundation item: Project (50204013) supported by the National Natural Science Foundation of China
Rights and permissions
About this article
Cite this article
Li, Wz., Qin, Wq., Qiu, Gz. et al. Electrode process of diethyldithiocarbamate on surface of pyrrhotite. J Cent. South Univ. Technol. 12, 416–419 (2005). https://doi.org/10.1007/s11771-005-0174-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11771-005-0174-y