Abstract
Purpose
The risk of second malignancies among female breast cancer patients has been studied for decades. In contrast, very little is known about second primary tumors in men. Risk factors for breast cancer in men, including genetic, hormonal and environmental factors, provide parallels to the etiology of breast cancer in women. This review considers the literature related to the risk of developing a second cancer in patients with male breast cancer.
Materials and methods
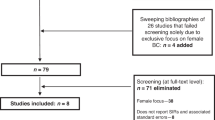
A systematic review of the literature between 1966 and 2007 was conducted and acceptable articles used for analysis. All retrieved articles were screened to identify any papers that had been missed. Studies were included if they discussed the risk of subsequent malignancy in patients with male breast cancer.
Results
Patients with history of male breast cancer have an increased risk of a second ipsilateral, or contralateral breast cancer (standardized incidence ratio 30–110). The risk of subsequent contralateral breast cancer was highest in men under 50 years of age at the time of the diagnosis of the initial cancer. The data on non-breast second primary cancers is diverse. One study has suggested an increased incidence of cancers of the small intestine, prostate, rectum and pancreas, and of non-melanoma skin cancer and myeloid leukaemia. Other investigators did not find an increase in the overall risk of subsequent cancer development in men diagnosed initially with primary breast cancer. Although sarcoma, lung and esophageal cancers are well recognized complications of radiation therapy for female breast cancer, there is no evidence for the association of these cancers following radiation therapy in male breast cancer.
Conclusions
Although the incidence of second primary cancer in patients with primary male breast cancer requires further study, male breast cancer survivors should probably undergo periodic screening for the early detection of second breast cancers and other adverse health effects.
Similar content being viewed by others
References
Fentiman IS, Fourquet A, Hortobagyi GN. Male breast cancer. Lancet 2006;367(9510):595–604.
Sasco AJ, Lowenfels AB, Pasker-de Jong J. Review article: epidemiology of male breast cancer. A meta-analysis of published case–control studies and discussion of selected aetiological factors. Int J Cancer 1993;53:538–49.
Giordano SH, Cohen DS, Buzdar AU, Perkins G, Hortobagyi GN. Breast carcinoma in men: a population-based study. Cancer 2004;101(1):51–7.
Anderson WF, Althuis MD, Brinton LA, Devesa SS. Is male breast cancer similar or different than female breast cancer? Breast Cancer Res Treat 2004;83(1):77–86.
Liede A, Karlan BY, Narod SA. Cancer risks for male carriers of germline mutations in BRCA1 or BRCA2: a review of the literature. J Clin Oncol 2004;22(4):735–42.
Struewing JP, Coriaty ZM, Ron E, et al. Founder BRCA1/2 mutations among male patients with breast cancer in Israel. Am J Hum Genet 1999;65:1800–2.
Thorlacius S, Sigurdsson S, Bjarnadottir H, et al. Study of a single BRCA2 mutation with high carrier frequency in a small population. Am J Hum Genet 1997;60:1079–84.
Haraldsson K, Loman N, Zhang QX, et al. BRCA2 germ-line mutations are frequent in male breast cancer patients without a family history of the disease. Cancer Res 1998;58:1367–71.
Csokay B, Udvarhelyi N, Sulyok Z, et al. High frequency of germ-line BRCA2 mutations among Hungarian male breast cancer patients without family history. Cancer Res 1999;59:995–8.
Thompson D, Easton D. Variation in cancer risks, by mutation position, in BRCA2 mutation carriers. Am J Hum Genet 2001;68:410–9.
The Breast Cancer Linkage Consortium. Cancer risks in BRCA2 mutation carriers. The Breast Cancer Linkage Consortium. J Natl Cancer Inst 1999;91(15):1310–6.
Moslehi R, Chu W, Karlan B, et al. BRCA1 and BRCA2 mutation analysis of 208 Ashkenazi Jewish women with ovarian cancer. Am J Hum Genet 2000;66:1259–72.
Weiss JR, Moysich KB, Swede H. Epidemiology of male breast cancer. Cancer Epidemiol Biomarkers Prev 2005;14(1):20–6.
D’Avanzo B, La Vecchia C. Risk factors for male breast cancer. Br J Cancer 1995;71:1359–62.
Calle EE, Thun MJ. Obesity and cancer. Oncogene 2004;23(38):6365–78.
Volm MD, Talamonti MS, Thangavelu M, Grtadishar WK. Pituitary adenoma and bilateral male breast cancer: an unusual association. J Surg Oncol 1997;64:74–8.
Forloni F, Giovilli M, Pecis C, et al. Pituitary prolactin-secreting macroadenoma combined with bilateral breast cancer in a 45-year old male. J Endocrinol Investig 2001;24:454.
Schell SR, Montague ED, Spanos WJ Jr, et al. Bilateral breast cancer in patients with initial stage I and II disease. Cancer 1982;50(6):1191–4.
Chaudary MA, Millis RR, Hoskins EO, Halder M, Bulbrook RD, Cuzick J, Hayward JL. Bilateral primary breast cancer: a prospective study of disease incidence. Br J Surg 1984;71(9):711–4.
Fisher B, Jeong JH, Anderson S, Bryant J, Fisher ER, Wolmark N. Twenty-five-year follow-up of a randomized trial comparing radical mastectomy, total mastectomy, and total mastectomy followed by irradiation. N Engl J Med 2002;347(8):567–75.
Hemminki K, Scelo G, Boffetta P, et al. Second primary malignancies in patients with male breast cancer. Br J Cancer 2005;92(7):1288–92.
Hemminki K, Granstrom C. Re: risk of subsequent cancer following breast cancer in men. J Natl Cancer Inst 2002;94(24):1892.
Cutuli BF, Lacroze M, Dilhuydy JM, et al. Breast cancer in men: incidence and types of associated previous synchronous and metachronous cancers. Bull Cancer 1992;79(7):689–96.
Auvinen A, Curtis RE, Ron E. Risk of subsequent cancer following breast cancer in men. J Natl Cancer Inst 2002;94(17):1330–2.
Broet P, de la Rochefordiere A, Scholl SM, et al. Contralateral breast cancer: annual incidence and risk parameters. J Clin Oncol 1995;13:1578–83.
Dong C, Hemminki K. Second primary breast cancer in men. Breast Cancer Res Treat 2001;66:171–2.
Sigurdsson S, Thorlacius S, Tomasson J, et al. BRCA2 mutation in Icelandic prostate cancer patients. J Mol Med 1997;75:758–61.
Anderson DE, Badzioch MD. Breast cancer risks in relatives of male breast cancer patients. J Natl Cancer Inst 1992;84:1114–7.
Karlsson CT, Malmer B, Wiklund F, Gronberg H. Breast cancer as a second primary in patients with prostate cancer-estrogen treatment or association with family history of cancer? J Urol 2006;176(2):538–43.
Leibowitz SB, Garber JE, Fox EA, et al. Male patients with diagnoses of both breast cancer and prostate cancer. Breast J 2003;9(3):208–12.
Thellenberg C, Malmer B, Tavelin B, Gronberg H. Second primary cancers in men with prostate cancer: an increased risk of male breast cancer. J Urol 2003;169(4):1345–8.
Yu H, Diamandis EP, Sutherland DJA. Immunoreactive prostate specific antigen levels in female and male breast tumors and its association with steroid hormone receptors and patient age. Clin Biochem 1994;27:75–9.
Inskip PD, Stovall M, Flannery JT. Lung cancer risk and radiation dose among women treated for breast cancer. J Natl Cancer Inst 1994;86(13):983–8.
Wang LE, Bondy ML, de Andrade M, et al. Gender difference in smoking effect on chromosome sensitivity to gamma radiation in a healthy population. Radiat Res 2000;154(1):20–7.
Peto R, Darby S, Deo H, et al. Smoking, smoking cessation, and lung cancer in the UK since 1950: combinationof national statistics with two case–control studies. BMJ 2000;321(7257):323–9.
Neugut AI, Meadows AT, Robinson E. Introduction. In: Neugut AI, Meadows AT, Robinson E, editors. Multiple primary cancers. Philadelphia, PA: Lippincott Williams & Wilkins; 1999, p. 1–12.
Matesich SM, Shapiro CL. Second cancers after breast cancer treatment. Semin Oncol 2003;30(6):740–8.
Galper S, Gelman R, Recht A, et al. Second nonbreast malignancies after conservative surgery and radiation therapy for early-stage breast cancer. Int J Radiat Oncol Biol Phys 2002;52(2):406–14.
Acknowledgements
We would like to thank Dr Natan Cherny for his helpful comments during the preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grenader, T., Goldberg, A. & Shavit, L. Second cancers in patients with male breast cancer: a literature review. J Cancer Surviv 2, 73–78 (2008). https://doi.org/10.1007/s11764-008-0042-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11764-008-0042-5