Abstract
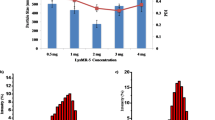
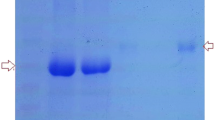
Lactoferricin is an amphipathic, cationic peptide with antimicrobial properties. This study aimed to investigate the combined effect of chitosan loaded with lactoferricin and antibody against Staphylococcus aureus and Pseudomonas aeruginosa. lactoferricin was prepared using the modified method of Law and Reiter. By injecting two inactivated bacteria S. aureus and P. aeruginosa along with chitosan as an adjuvant to the camel neck, antibodies were prepared and their production was confirmed using the Indirect-ELISA method. Lactoferricin and antibody were loaded on chitosan using thiamine method. The combined effect of lactoferricin and antibody loaded on chitosan against bacteria was investigated using a bacterial growth test in the presence or absence (control) of the sample. Based on the results of statistical analysis, the data obtained from the combined effect of lactoferricin and antibody loaded on chitosan reduced the growth rate of S. aureus and P. aeruginosa compared to the control (without lactoferricin and antibody loaded on chitosan). There is a significant interaction (p ≤ 0.01) between different levels of lactoferricin and antibody loaded on chitosan on the growth of both bacteria. Bacterial growth curves (based on optical density) showed that all concentrations of lactoferricin and antibody dilutions significantly decreased the growth of S. aureus and P. aeruginosa. According to the growth curves, it can be concluded that lactoferricin and antibody loaded on chitosan acted more effectively on gram-positive bacteria than gram-negative bacteria. Based on the results, it can be stated that lactoferricin and antibody loaded on chitosan as an antibacterial can be useful in a variety of commercial programs to control the growth and survival of pathogens.








Similar content being viewed by others
References
Alizadeh H, Madani R, Babaie M et al (2012) Preparation and purification of polyclonal antibodies against Mycobacterium avium paratuberculosis antigens in rabbit. J Fasa Univ Med Sci 2(3):168–173 magiran.com/p1074325.
Andresen LO (1999) Development and evaluation of an indirect ELISA for detection of exfoliative toxin ExhA, ExhB or ExhC produced by Staphylococcus hyicus. Vet Microbiol 68(3–4):285–292. https://doi.org/10.1016/s0378-1135(99)00113-3
Annunziato G, Costantino G (2020) Antimicrobial peptides (AMPs): a patent review (2015–2020). Expert Opin Ther Pat 30(12):931–947. https://doi.org/10.1080/13543776.2020.1851679
Behrouz S, Saadat S, Memarzia A et al (2022) The Antioxidant, anti-inflammatory and Immunomodulatory Effects of Camel milk. Front Immunol 13:855342. https://doi.org/10.3389/fimmu.2022.855342
Bellamy W, Takase M, Wakabayashi H et al (1992) Antibacterial spectrum of lactoferricin B, a potent bactericidal peptide derived from the N-terminal region of bovine lactoferrin. J Appl Bacteriol 73(6):472–479. https://doi.org/10.1111/j.1365-2672.1992.tb05007.x
Biasibetti E, Rapacioli S, Bruni N et al (2020) Lactoferrin-derived peptides antimicrobial activity: an in vitro experiment. Nat Pro Res 12,PP:1–5. https://doi.org/10.1080/14786419.2020.1821017
Bruni N, Capucchio MT, Biasibetti E et al (2016) Antimicrobial activity of lactoferrin-related peptides and applications in Human and Veterinary Medicine. Mol 21(6):752. https://doi.org/10.3390/molecules21060752
Duarte LGR, Picone CSF (2022) Antimicrobial activity of lactoferrin-chitosan-gellan nanoparticles and their influence on strawberry preservation. Food Res Int 159:111586. https://doi.org/10.1016/j.foodres.2022.111586
El-Baky NA, Elkhawaga MA, Abdelkhalek ES, e al (2020) De novo expression and antibacterial potential of four lactoferricin peptides in cell-free protein synthesis system. Biotech Rep (Amst) 29:e00583. https://doi.org/10.1016/j.btre.2020.e00583
Farnaud S, Patel A, Odell EW et al (2004) Variation in antimicrobial activity of lactoferricin-derived peptides explained by structure modelling. FEMS Microbiol Lett 238(1):221–226. https://doi.org/10.1111/j.1574-6968.2004.tb09759.x
Farshbaf M (2021) Encapsulating Staphylococcus aureus bacteria by calcium alginate and chitosan and investigating its adjuvant properties to produce effective nanoparticles against Staphylococcus aureus. Dissertation, Islamic Azad University of Mashhad
Gifford JL, Hunter HN, Vogel HJ (2005) Lactoferricin. Cell Mol Life ScI 62,22,PP. 2588–2598. https://doi.org/10.1007/s00018-005-5373-z
Ho TM, Zou Z, Bansal N (2022) Camel milk: a review of its nutritional value, heat stability, and potential food products. Food Res Int 153:110870. https://doi.org/10.1016/j.foodres.2021.110870
Huan Y, Kong Q, Mou H et al (2020) Antimicrobial peptides: classification, design, application and research progress in multiple Fields. Front Microbiol 11:582779. https://doi.org/10.3389/fmicb.2020.582779
Jackson N, Wu TZ, Adams-Sapper S et al (2019) A multiplexed, indirect enzyme-linked immunoassay for the detection and differentiation of E. coli from other Enterobacteriaceae and P. aeruginosa from other glucose non-fermenters. J Microbiol Methods 158:52–58. https://doi.org/10.1016/j.mimet.2019.01.014
Jovčevska I, Muyldermans S (2020) The therapeutic potential of nanobodies. BioDrugs 34(1):11–26. https://doi.org/10.1007/s40259-019-00392-z
Kaintura T, Sharma P, Singh S et al (2015) Gelatin nanoparticles as a delivery system for proteins. J Nanomed Res 2(1). https://doi.org/10.15406/jnmr.2015.02.00018
Kell DB, Heyden EL, Pretorius E (2020) The Biology of Lactoferrin, an Iron-binding protein that can help defend against viruses and Bacteria. Front Immunol 11:1221. https://doi.org/10.3389/fimmu.2020.01221
Kim JH, Yu D, Eom SH et al (2017) Synergistic Antibacterial Effects of Chitosan-Caffeic Acid Conjugate against Antibiotic-Resistant Acne-Related Bacteria. Mar Drugs 15(6):167. https://doi.org/10.3390/md15060167
Lee B, Hwang JS, Lee DG (2020) Antibacterial action of lactoferricin B like peptide against Escherichia coli: reactive oxygen species-induced apoptosis‐like death. J Appl Microbiol 129(2):287–295. https://doi.org/10.1111/jam.14632
Mahlapuu M, Håkansson J, Ringstad L et al (2016) Antimicrobial peptides: an emerging category of Therapeutic Agents. Front Cell Infect Microbiol 6:194. https://doi.org/10.3389/fcimb.2016.00194
Moradi A, Pourseif MM, Jafari B et al (2020) Nanobody-based therapeutics against colorectal cancer: Precision therapies based on the personal mutanome profile and tumor neoantigens. Pharmacol Res 156:104790. https://doi.org/10.1016/j.phrs.2020.104790
Mousavi Parsa M (2021) Loading of prepared nanobody against Staphylococcus aureus bacteria on chitosan and alginate. Dissertation, Islamic Azad University of Mashhad
Navarrete-Rodríguez EM, Ríos-Villalobos LA, Alcocer-Arreguín CR et al (2018) Cross-over clinical trial for evaluating the safety of Camel’s milk intake in patients who are allergic to cow’s milk protein. Allergol Immunopathol 46(2):149–154. https://doi.org/10.1016/j.aller.2017.06.005
Pashnayar S, Asaran Darban R, Zibaei S (2021) Purification of nanobody produced against Pseudomonas aeruginosa bacteria using ion exchange chromatography. Appl Res Anim Sci 10(38):67–74. https://doi.org/10.22092/aasrj.2021.124293
Ponnaganti M, Kishore Babu A (2021) Preparation, characterization and evaluation of Chitosan Nanobubbles for the targeted delivery of Ibrutinib. Nat Volatiles Essent Oils 8(6):5017–5037
Raei M, Rajabzadeh G, Zibaei S et al (2015) Nano-encapsulation of isolated lactoferrin from camel milk by calcium alginate and evaluation of its release. Int J Biol Macromol 79:669–673. https://doi.org/10.1016/j.ijbiomac.2015.05.048
Ren Z, Zhang X, GuoY et al (2016) Preparation and in vitro delivery performance of chitosan–alginate microcapsule for IgG. Food Agric Immunol 28(1):1–13. https://doi.org/10.1080/09540105.2016.1202206
Sharbafi R, Moradian F, Rafiei A et al (2013) Isolation and purification of lactoferrin from cow’s milk. J Maz Univ Med Sci 21(84):44–51. http://jmums.mazums.ac.ir/article-1-668-fa.html
Sharbafi R, Moradian F, Rafiei A (2015) Investigating the antimicrobial activity of lactoferrin protein on the growth of Pseudomonas aeruginosa and Escherichia coli. J Babol Univ Med Sci 18(7):19–23. http://jbums.org/article-1-5958-fa.html
Sinha M, Kaushik S, Kaur P et al (2013) Antimicrobial lactoferrin peptides: the hidden players in the protective function of a multifunctional protein. Int J of Pep PP:2013. https://doi.org/10.1155/2013/390230
Soleimani sh, Zibaei S (2022) Combined effect of lactoferrin and nanobody loaded on chitosan against Staphylococcus aureus and Pseudomonas aeruginosa. App Ani Sci Res J PP 7–16. https://doi.org/10.22092/aasrj.2022.128151
Stetefeld J, McKenna SA, Patel TR (2016) Dynamic light scattering: a practical guide and applications in biomedical sciences. Bio rev 8:409–427. https://doi.org/10.1007/s12551-016-0218-6
Svendsen JSM, Grant TM, Rennison D et al (2019) Very short and stable lactoferricin-derived antimicrobial peptides: Design Principles and potential uses. Acc Chem Res 52(3):749–759. https://doi.org/10.1021/acs.accounts.8b00624
Tanhaeian A, Mohammadi E, Mansury D et al (2020) Assessment of a novel antimicrobial peptide against clinically isolated animal pathogens and prediction of its Thermal-Stability. Microb Drug Resist 26(4):412–419. https://doi.org/10.1089/mdr.2019.0062
Tomita M, Takase M, Bellamy W et al (1994) A review: the active peptide of lactoferrin. Acta Paediatr Jpn 36(5):585–591. https://doi.org/10.1111/j.1442-200X.1994.tb03250.x
Trinidad-Calderón PA, Varela-Chinchilla CD, García-Lara S (2021) Natural peptides inducing Cancer Cell Death: Mechanisms and Properties of specific candidates for Cancer therapeutics. Molecules 26(24):7453. https://doi.org/10.3390/molecules26247453
Unciti-Broceta JD, Del Castillo T, Soriano M et al (2013) Novel therapy based on camelid nanobodies. Ther Deliv 4(10):1321–1336. https://doi.org/10.4155/tde.13.87
Wakabayashi H, Bellamy W, Takase M et al (1992) Inactivation of Listeria monocytogenes by Lactoferricin, a potent antimicrobial peptide derived from cow’s milk. J Food Prot 55(4):238–240. https://doi.org/10.4315/0362-028X-55.4.238
Yang Y, Zhao X, Li X et al (2017) Effects of anti-CD44 monoclonal antibody IM7 carried with chitosan polylactic acid-coated nano-particles on the treatment of ovarian cancer. Oncol Lett 13(1):99–104. https://doi.org/10.3892/ol.2016.5413
Ye X, Yoshida S, Ng TB (2000) Isolation of lactoperoxidase, lactoferrin, alpha-lactalbumin, beta-lactoglobulin B and beta-lactoglobulin A from bovine rennet whey using ion exchange chromatography. Int J Biochem Cell Biol 32(11–12):1143–1150. https://doi.org/10.1016/s1357-2725(00)00063-7
Zhang QY, Yan ZB, Meng YM et al (2021) Antimicrobial peptides: mechanism of action, activity and clinical potential. Mil Med Res 8(1):48. https://doi.org/10.1186/s40779-021-00343-2
Acknowledgements
The support of the Razi Vaccine & Serum Research Institute during this study is appreciated. This manuscript is prepared based on the Ph.D thesis of the first author at Rasht Branch, Islamic Azad University, Rasht, Iran.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical considerations
Adherence to the principles of research ethics.this research has been approved by the ethics committee of Lahijan Azad University with the code IR.IAU.LIAU.REC.1400.055.
Conflict of interest
Authors declare that there are no known conflicts of interest associated with this publication.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Soleimani, S., Zibaee, S. & Anvari, M. Combined effect of chitosan loaded with lactoferricin and antibody against Staphylococcus aureus and Pseudomonas aeruginosa. Biologia 79, 275–289 (2024). https://doi.org/10.1007/s11756-023-01540-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-023-01540-1