Abstract
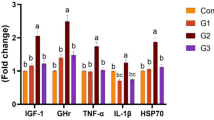
The objective of the present experiment was to investigate the effect of a chelated form of trace minerals in the diet on hatchability, immune system response, and blood parameters in Ross 308 broiler breeders. In this experiment, 560 broiler breeder chickens (520 females and 40 males) of Ross 308 with an average age of 52 weeks were used based on a completely randomized design. There were eight treatments with five replicates and in each replicate there were 13 hens and one rooster. The experimental diets were the same except for the form of the trace minerals. Treatment 1 (negative control): Mn, Se, Cu, Fe and Zn provided as non-chelated form; treatment 2 (positive control): Mn, Se, Cu, Fe and Zn provided as chelated form; treatment 3: Mn, Se, Cu, Fe and Zn provided as 50% chelated form and 50% non-chelated form; treatment 4: Zn provided as chelated form; treatment 5: Cu provided as chelated form; treatment 6: Fe provided as chelated form; treatment 7: Mn provided as chelated form; treatment 8: Se provided as chelated form. In all treatment groups, the quantities of Zn, Cu, Fe, Mn, Se and I were added as much as 110, 10, 50, 120, 0.3 and 2 mg kg− 1 feed based on their requirements, respectively. Data on hatchability, immune system response, and blood parameters were analyzed using SPSS software. The results showed that the number and percentage of hatched chicks were affected by treatment throughout experimental period (p < 0.05), such that treatments 2 and 3 had the highest values compared with the other treatments (p < 0.05); however, the number and percentage of unhatched chicks were not affected by treatment. The number and percentage of fertilized eggs throughout the production period were not affected by the treatments. Hatch residue analysis revealed that the percentage of mild embryonic mortality throughout the production period was affected by the experimental treatments (p < 0.05), with the lowest rate in treatment 2. In addition, no significant difference was found between the experimental treatments in terms of early embryonic mortality, late embryonic mortality, contaminated, external pip, and cracked throughout the production period. The results of the immune system examination and blood parameters showed that treatments 2 and 3 had the highest antibody titers against Newcastle (p < 0.05). In contrast to the levels of low-density lipoproteins (LDL) and high-density lipoprotein (HDL), the levels of estrogen and cholesterol were affected by the experimental treatments (p < 0.05) and there was a significant difference between the experimental treatments in the level of liver enzyme alkaline phosphatase (p < 0.05). The research results indicate an improvement in the hatchability and immune system of broilers fed experimental diets containing chelated forms of Zn, Fe, Cu, Se, and Mn (treatment 2) and a basal diet containing 50% of the minerals Zn, Fe, Cu, Se, and Mn in the sulfate form and 50% in the chelate form (treatment 3).
Similar content being viewed by others
Data Availability
Raw data is available from corresponding author upon reasonable request.
References
Ajayi OB, Odutuga A (2004) Effect of low-Zn status and essential fatty acids deficiency on the activities of aspartate aminotransferase and alanine aminotransferase in liver and serum of albino rats. Mol Nutr Food Res 48(2):88–90. https://doi.org/10.1002/food.200300316
Al-Daraji HJ, Amen MHM (2011) Effect of dietary zn on certain blood traits of broiler breeder chickens. Int Poult Sci 10(10):807–813. https://doi.org/10.3923/ijps.2011.807.813
Aminullah NTM, Prabhu JN, Suresh BN, Indresh HC (2021) Performance of Swarnadhara breeder hens supplemented with reduced levels of different copper forms. Vet World 14(5):1371–1379. https://doi.org/10.14202/vetworld.2021.1371-1379
Ao T, Pierce JL, Power R, Pescatore AJ, Cantor AH, Dawson KA, Ford MJ (2009) Effects of feeding different forms of Zn and copper on the performance and tissue mineral content of chicks. Poult Sci 88:2171–2175. https://doi.org/10.3382/ps.2009-00117
Association of Official Analytical Chemistry (1994) Official methods of analysis of the Association of Official Analytical Chemists. Vols I & II, Association of Analytical chemists, Arlington
Attia YA, Abdalah AA, Zeweil HS, Bovera F, Tag El-Din AA, Araft MA (2011) Effect of inorganic or organic copper additions on reproductive performance, lipid metabolism and morphology of organs of dualpurpose breeding hens. Arch Geflügelk 75(3):169–178
Brown AJ, Jessup W (1999) Oxysterols and atherosclerosis. Atherosclerosis 142:1–28. https://doi.org/10.1016/S0021-9150(98)00196-8
Bun SD, Guo YM, Gue F, Cao H (2011) Influence of organic zinc supplementation on the antioxidant status and immune responses of broilers challenged with Eimeria tenella. Poult Sci 90:1220–1226. https://doi.org/10.3382/ps.2010-01308
Chen S, Li T, Yeh R, Lien T, Huang C, Chen K (2011) Impact of high dietary copper on the reproductive characteristics of laying hens and growth of newly hatched chicks. J Chin Soc Anim Sci 43(3):11–24. https://doi.org/10.1016/j.psj.2020.10.033
Das A, Mishra SK, Swain RK, Swain P, Dhama K, Sahoo G, Behura NC, Sethi K, Chichilichi B, Behera T, Mishra SR (2014b) Effect of organic minerals supplementation on growth, bioavailability and immunity in grower birds. Int J Pharm 10:380–388. https://doi.org/10.3923/ijp.2014.380.388
Ebeid TA (2012) Vitamin E and organic selenium enhances the antioxidative status and quality of chicken cockerel semen under high ambient temperature. Br Poult Sci 5:708–717. https://doi.org/10.1080/00071668.2012.722192
Faghih-Mohammadi F, Seidavi AR, Bouyeh M (2023) The effect of the chelated form of trace elements in diet on weight gain, production traits, egg specific gravity, immune system, blood parameters, liver enzymes, and progesterone hormone in Ross 308 broiler breeder chickens. Ital J Anim Sci 22:524–536. https://doi.org/10.1080/1828051X.2023.2215248
Favero A, Vieira SL, Angel CR, Bess F, Cemin HS, Ward TL (2013a) Reproductive performance of Cobb 500 breeder hens fed diets supplemented with zn, manganese, and copper from inorganic and amino acid-complexed sources. J Appl Poult Res 22:80–91. https://doi.org/10.3382/japr.2012-00607
Favero A, Vieira SL, Angel CR, Bos-Mikich A, Lothhammer N, Taschetto D, Cruz RFA, Ward TL (2013b) Development of bone in chick embryos from Cobb 500 breeder hens fed diets supplemented with zinc, manganese, and copper from inorganic and amino acid-complexed sources. Poult Sci 92:402–411. https://doi.org/10.3382/ps.2012-02670
Feng J, Ma WQ, Niu HH, Wu XM, Wang Y (2010) Effects of Zn glycine chelate on growth, hematological, and immunological characteristics in broilers. Biol Trace Elem Res 133:203–211. https://doi.org/10.1007/s12011-009-8431-9
Hajilari D, Shams Shargh M, Ashayerizadeh O (2019) Effects of dietary organic and inorganic zn and copper supplements on performance, footpad dermatitis, carcass characteristics, and blood profile of broiler chickens. Poult Sci J 7(1):15–23
Hoffmann PR (2007) Mechanisms by which selenium influences immune responses. Arch Immunol Ther Exp 55:289–297. https://doi.org/10.1007/s00005-007-0036-4
Idowu OMO, Ajuwon RO, Oso AO, Akinloye OA (2011) Effect of zinc supplementation on laying performance, serume, chemistry and zn residue in tibia bone, liver, excreta and egg shell of laying hens. Int J Poult Sci 10:225–230. https://doi.org/10.3923/ijps.2011.225.230
Johnson J (1990) Ranking countries for minerals exploration,“ Natural Resources Forum. Blackwell Publishing 14(3):178–186. https://doi.org/10.1111/j.1477-8947.1990.tb00516.x
Jongbloed AW, Kemme PA, De Groote G, Lippens M, Meschy F (2002) Bioavailability of major and trace minerals. International Association of the European (EU) Manufacturers of major, trace and specific Fed mineral materials (EMFEMA), Brussels, Belgium
M’Sadeq SA, Wu SB, Choct M, Swick RA (2018) Influence of trace mineral sources on broiler performance, lymphoid organ weights, apparent digestibility, and bone mineralization. Poult Sci 97:3176–3182. https://doi.org/10.3382/ps/pey197
Mahfouz MM, Kawano H, Kummerow FA (1997) Effect of cholesterol-rich diets with and without added vitamins E and C on the severity of atherosclerosis in rabbits. Am J Clin Nutr 66:1240–1249. https://doi.org/10.1093/ajcn/66.5.1240
Manangi MK, Vazques-Anon M, Richards JD, Carter S, Knight CD (2015) The impact of feeding supplemental chelated trace minerals on shell quality, tibia breaking strength, and immune response in laying hens. J Appl Poult Res 24:316–326. https://doi.org/10.3382/japr/pfv029
McQuoid D (2000) Operation of multiple-stage and single-stage incubation systems. World Poult 18(2):18–22
Mroczek-Sosnowska N, Sawosz E, Vadalasetty K, Łukasiewicz M, Niemiec J, Wierzbicki M, Kutwin M, Jaworski S, Chwalibog A (2015) Nanoparticles of copper stimulate angiogenesis at systemic and molecular level. Int J Mol Sci 16(3):4838–4849. https://doi.org/10.3390/ijms16034838
Muszynski S, Tomaszewska E, Kwiecie´n M, Dobrowolski P, Tomczyk A (2018) Effect of dietary phytase supplementation on bone and hyaline cartilage development of broilers fed with organically complexed copper in a cu-deficient diet. Biol Trace Elem Res 182:339–353. https://doi.org/10.1007/s12011-017-1092-1
Narinc D, Aygun A, Sari T (2013) Effects of cage type and mating ratio on fertility in japanese quails (Coturnix coturnix japonica) eggs. Agr Sci Dev 2:4–7
Oviedo-Rondon EO, Leandro NM, Ali R, Koci M, Moraes V, Brake J (2013) Broiler breeder feeding programs and trace minerals on maternal antibody transfer and broiler humoral immune response1. J Appl Poult Res 22:499–510. https://doi.org/10.3382/japr.2012-00708
Pineda L, Sawosz E, Vadalasetty KP, Chwalibog A (2013) Effect of copper nanoparticles on metabolic rate and development of chicken embryos. Anim Feed Sci Technol 186(1–2):125–129. https://doi.org/10.1016/j.anifeedsci.2013.08.012
Rajeswari S, Swaminathan S (2014) Role of copper in health and diseases. Int J Curr Sci 10:94–107
Rama Rao SV, Bhukya P, Mantena VLNR, Arun KP, Saharia P, Orugonda KM (2013) Effect of supplementing organic selenium on performance, carcass traits, oxidative parameters and immune responses in commercial broiler chickens. Asian Aust J Anim Sci 26(2):247–252. https://doi.org/10.5713/ajas.2012.12299
Renema RA (2019) Reproductive responses to Sel-PlexR organic selenium in male and female broiler breeders: Impact on production traits and hatchability. Poult Indus https://en.engormix.com/poultry-industry/articles/sel-plex-organic-selenium-in-male-female-broiler-breeders-t33862.htm; Accessed 01 June 2021
Aviagen (2009) Investigating hatchery practice. Aviagen
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG (1973) Selenium: biochemical role as a component of glutathione peroxidase. J Poult Sci 179:588–590. https://doi.org/10.1126/science.179.4073.588
Saber S, Kutlu HR, Yusuf UZ, Celik L, Yucelt O, Baylan M (2020) Effects of form of dietary trace mineral premix on fertility and hatchability of broiler breeder hens and post-hatch performance and carcass parameters of their progenies. Kafkas Üniv Vet Fakültesi Derg 26(2):171–180. https://doi.org/10.9775/kvfd.2019.22647
Saito Y, Yoshida Y, Niki E (2007) Cholesterol is more susceptible to oxidation than linoleates in cultured cells under oxidative stress induced by selenium deficiency and free radicals. FEBS Letters 581:4349–4354. https://doi.org/10.1016/j.febslet.2007.08.010
Sakumoto R, Hayashi KG, Takahashi T (2014) Different expression of PGE synthase, PGF receptor, TNF, Fas and oxytocin in the bovine corpus luteum of the estrous cycle and pregnancy. Rep Biol 14:115–121. https://doi.org/10.1016/j.repbio.2013.12.003
Sapp RL, Rekaya R, Misztal I, Wing T (2004) Male and female fertility and hatchability in chickens: a longitudinal mixed model approach. Poult Sci 83:1253–1259. https://doi.org/10.1093/ps/83.8.1253
Scott A, Vadalasetty KP, Sawosz E, Łukasiewicz M, Vadalasetty RKP, Jaworski S, Chwalibog A (2016) Effect of copper nanoparticles and copper sulphate on metabolic rate and development of broiler embryos. Anim Feed Sci Technol 220:151–158. https://doi.org/10.1016/j.anifeedsci.2016.08.009
Scott A, Prasad K, André VC, Sawosz E (2018) Copper nanoparticles as an alternative feed additive in poultry diet: a review. Nanotech Rev 7(1):69–93. https://doi.org/10.1515/ntrev-2017-0159
Seidavi A, Asadpour L, Dadashbeiki M, Payan-Carreira R (2014) Effects of dietary fish oil and green tea powder supplementation on broiler chickens immunity. Acta Sci Vet 42(1):1–13
Sun Q, Guo Y, Li J, Zhang T, Wen J (2012) Effects of methionine hydroxy analog chelated Cu/Mn/Zn on laying performance, egg quality, enzyme activity and mineral retention of laying hens. J Poult Sci 49(1):20–25. https://doi.org/10.2141/jpsa.011055
Sun X, Lu L, Liao X, Zhang L, Lin X, Luo X, Ma Q (2018) Effect of in ovo zinc injection on the embryonic development and epigenetics-related indices of zinc-deprived broiler breeder eggs. Biol Trace Elem Res 185(2):456-464. https://doi.org/10.1007/s12011-018-1260-y
Surai PF, Kochish II, Fisinin VI, Kidd MT (2019) Antioxidant defence systems and oxidative stress in poultry biology: an update. Antioxidants 8:235. https://doi.org/10.3390/antiox8070235
Światkiewicz S, Arczewska-Wlosek A, Jozefiak D (2014) The efficacy of organic minerals in poultry nutrition: review and implications of recent studies. J World Poult Sci 703:475–486. https://doi.org/10.1017/S0043933914000531
Tabatabaie MM, Aliarabi H, Saki AA, Ahmadi A, Siyar SA (2007) Effect of different sources and levels of Zn on egg quality and laying hen performance. Pak J Biol Sci 10(19):3476–3347
Vieira MM, Ribeiro AML, Kessler AM, Moraes ML, Kunrath MA, Ledur VS (2013) Different sources of dietary zn for broilers submitted to immunological, nutritional, and environmental challenge. J Appl Int Poult Res 22:855–861. https://doi.org/10.3382/japr.2013-00753
Wang G, Liu LJ, Tao WJ, Xiao ZP, Pei X, Liu J, Wang MQ, Lin G, Ao TY (2019) Effects of replacing inorganic trace minerals with organic trace minerals on the production performance, blood profiles, and antioxidant status of broiler breeders. Int J Poult Sci 98:2888–2895. https://doi.org/10.3382/ps/pez035
Xie C, Elwan HAM, Elnesr SSM, Dong X, Feng J, Zou XT (2019) Effects of iron glycine chelate on laying performance, antioxidant activities, serum biochemical indices, iron concentrations and transferrin mRNA expression in laying hens. J Anim Physiol Anim Nutr 103:547–554. https://doi.org/10.1111/jpn.13061
Yenice E, Mrzrak C, Gultekin M (2015) Effects of organic and inorganic forms of manganese, zinc, copper, and chromium on bioavailability of these minerals and calcium in late-phase laying hens. Biol Trace Elem Res 167:300–307. https://doi.org/10.1007/s12011-015-0313-8
Zhang ZW, Wang QH, Zhang JL, Li S, Wang XL, Xu SW (2012) Effects of oxidative stress on immunosuppression induced by selenium deficiency in chickens. Biol Trace Elem Res 149:352–361. https://doi.org/10.1007/s12011-012-9439-0
Zhao J, Shirley RB, Vazquez-Anon M, Dibner JJ, Richards JD, Fisher P, Hampton T, Christensen KD, Allard JP, Giesen AF (2010) Effects of chelated trace minerals on growth performance, breast meat yield, and footpad health in commercial meat broilers. J Appl Poult Res 19:365–372. https://doi.org/10.3382/japr.2009-00020
Zia MW, Khalique A, Naveed S, Hussain J (2018) Organic and inorganic selenium in poultry: a review. Indian J Anim Res 52(4):483–489. https://doi.org/10.18805/ijar.v0iOF.6837
Funding
This manuscript is prepared based on Ph.D. thesis of the first author at Rasht Branch, Islamic Azad University, Rasht, Iran. Financial support by Rasht Branch, Islamic Azad University, grant number 17.16.1.462 is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
FFM, AS and MB participated in preparing the manuscript equally, and all authors approved of the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest/Competing interests
There is no conflict of interest.
Ethics approval
The study was approved by the ethics committees of the Islamic Azad University.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Faghih-Mohammadi, F., Seidavi, A. & Bouyeh, M. Effects of the dietary chelated form of trace minerals on hatchability, immune system response, blood constitutes, and estrogen hormone in Ross 308 broiler breeders. Biologia 78, 3427–3439 (2023). https://doi.org/10.1007/s11756-023-01480-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-023-01480-w