Abstract
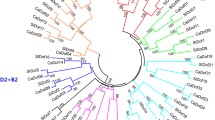
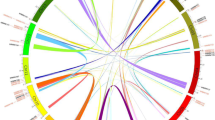
The Dof (DNA binding with one finger) genes encode a family of plant-specific transcription factors and play critical roles in numerous biological processes in plants. Piper nigrum L. (black pepper) is a popular spice crop with high economic value. Phytophthora capsici infection has become a key problem in P. nigrum industry, but the molecular studies about P. capsici have been rarely performed. This study aimed to conduct a genome-wide identification and characterization of the Dof gene family in P. nigrum, and provide some candidates differentially expressed genes under P. capsica infection. A total of 46 Dof genes were identified in the P. nigrum genome, and these genes were unevenly distributed on 20 chromosomes. Afterward, a phylogenetic tree was constructed by the N-J (neighbor-joining) method, and seven groups (not including one group that was not present in P. nigrum but present in Arabidopsis thaliana (L.) Heynh.) were obtained. The Dof genes in the same group had similar gene and protein structures. Selection pressure analysis indicated that duplicated genes underwent purification selection during evolution. Tissue expression analysis showed that P. nigrum Dof genes had significant tissue-specific expression characteristics, and they also expressed significantly different among different groups. In addition, according to the RNA-seq sequence data of Dof genes under the infection of P. capsici, a large number of Dof genes were found with differential expression profiles, and several genes with high response to P. capsici were screened out. This study revealed evolutionary and functional characteristics of P. nigrum Dof genes, and provided important references for the follow-up study of Dof genes in response to the biological stress of P. capsici.







Similar content being viewed by others
Abbreviations
- A. thaliana :
-
Arabidopsis thaliana
- CDF3 :
-
Cycling DOF factor 3
- HMM:
-
Hidden Markov Model
- MW:
-
Molecular Weight
- N-J:
-
Neighbor-joining
- P. capsici :
-
Phytophthora capsici
- PI:
-
Isoelectric point
- P. nigrum :
-
Piper nigrum
- TAIR:
-
The Arabidopsis Information Resource
- TDDF1 :
-
Tomato Dof Daily Fluctuations 1
References
Aravind R, Kumar A, Eapen SJ, Ramana KV (2009) Endophytic bacterial flora in root and stem tissues of black pepper (Piper nigrum L.) genotype: isolation, identification and evaluation against Phytophthora capsici. Lett Appl Microbiol 48:58–64. https://doi.org/10.1111/j.1472-765X.2008.02486.x
Bailey TL, Boden M, Buske FA et al (2009) MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res 37:W202–W208. https://doi.org/10.1093/nar/gkp335
Brasier CM (1992) Evolutionary biology of phytophthora. Annu Rev Phytopathol 30:153–201. https://doi.org/10.1146/annurev.py.30.090192.001101
Camacho C, Goulouris C, Avagyan V et al (2009) Blast+: architecture and applications. BMC Bioinform 10:421. https://doi.org/10.1186/1471-2105-10-421
Cavalar M, Möller C, Offermann S et al (2003) The interaction of DOF transcription factors with nucleosomes depends on the positioning of the binding site and is facilitated by maize HMGB5. Biochemistry 42:2149–2157. https://doi.org/10.1021/bi026761r
Chen C, Chen H, Zhang Y et al (2020) TBtools: an integrative toolkit developed for interactive analyses of big biological data. Mol Plant 13:1194–1202. https://doi.org/10.1016/j.molp.2020.06.009
Corrales A-R, Carrillo L, Lasierra P et al (2017) Multifaceted role of cycling DOF factor 3 (CDF3) in the regulation of flowering time and abiotic stress responses in Arabidopsis. Plant Cell Environ 40:748–764. https://doi.org/10.1111/pce.12894
Domínguez-Figueroa J, Carrillo L, Renau-Morata B et al (2020) The Arabidopsis transcription factor CDF3 is involved in nitrogen responses and improves nitrogen use efficiency in tomato. Front Plant Sci 11. https://doi.org/10.3389/fpls.2020.601558
Dong C, Hu H, Xie J (2016) Genome-wide analysis of the DNA-binding with one zinc finger (Dof) transcription factor family in bananas. Genome 59:1085–1100. https://doi.org/10.1139/gen-2016-0081
El-Gebali S, Mistry J, Bateman A et al (2019) The Pfam protein families database in 2019. Nucleic Acids Res 47:D427–D432. https://doi.org/10.1093/nar/gky995
Ewas M, Khames E, Ziaf K et al (2017) The tomato DOF daily fluctuations 1, TDDF1 acts as flowering accelerator and protector against various stresses. Sci Rep 7. https://doi.org/10.1038/s41598-017-10399-7
Feng B-H, Han Y-C, Xiao Y-Y et al (2016) The banana fruit Dof transcription factor MaDof23 acts as a repressor and interacts with MaERF9 in regulating ripening-related genes. J Exp Bot 67:2263–2275. https://doi.org/10.1093/jxb/erw032
Gasteiger E, Gattiker A, Hoogland C et al (2003) ExPASy: the proteomics server for in-depth protein knowledge and analysis. Nucleic Acids Res 31:3784–3788. https://doi.org/10.1093/nar/gkg563
Ghahramani Z (2001) An introduction to hidden Markov models and Bayesian networks. World Sci 45:9–41. https://doi.org/10.1142/S021800140100083
Goralogia GS, Liu T-K, Zhao L et al (2017) CYCLING DOF FACTOR 1 represses transcription through the TOPLESS co-repressor to control photoperiodic flowering in Arabidopsis. Plant J 92:244–262. https://doi.org/10.1111/tpj.13649
Guo Y, Qin G, Gu H, Qu L-J (2009) Dof5.6/HCA2, a Dof transcription factor gene, regulates interfascicular cambium formation and vascular tissue development in Arabidopsis. Plant Cell 21:3518–3534. https://doi.org/10.1105/tpc.108.064139
Hao C, Xia Z, Fan R et al (2016) De novo transcriptome sequencing of black pepper (Piper nigrum L.) and an analysis of genes involved in phenylpropanoid metabolism in response to Phytophthora capsici. BMC Genomics 17(1):822. https://doi.org/10.1186/s12864-016-3155-7
Hu L, Xu Z, Wang M et al (2019) The chromosome-scale reference genome of black pepper provides insight into piperine biosynthesis. Nat Commun 10. https://doi.org/10.1038/s41467-019-12607-6
Imaizumi T, Schultz TF, Harmon FG, Ho LA, Kay SA (2005) FKF1 F-box protein mediates cyclic degradation of a repressor of CONSTANS in Arabidopsis. Sci 309:293–297. https://doi.org/10.1126/science.1110586
Konishi M, Donner TJ, Scarpella E, Yanagisawa S (2015) MONOPTEROS directly activates the auxin-inducible promoter of the Dof5.8 transcription factor gene in Arabidopsis thaliana leaf provascular cells. J Exp Bot 66(1):283–291. https://doi.org/10.1093/jxb/eru418
Lamesch P, Berardini TZ, Li D et al (2012) The Arabidopsis information resource (TAIR): improved gene annotation and new tools. Nucleic Acids Res 40:D1202–D1210. https://doi.org/10.1093/nar/gkr1090
Langfelder P, Horvath S (2008) WGCNA: an R package for weighted correlation network analysis. BMC Bioinform 9:559. https://doi.org/10.1186/1471-2105-9-559
Lardon R, Wijinker E, Keurentjes J, Geelen D (2020) The genetic framework of shoot regeneration in Arabidopsis comprises master regulators and conditional fine-tuning factor. Commun Biol 3. https://doi.org/10.1038/s42003-020-01274-9
Larkin MA, Blackshields G, Brown NP et al (2007) Clustal W and Clustal X version2.0. Bioinform 23:2947–2948. https://doi.org/10.1093/bioinformatics/btm404
Letunic I, Bork P (2018) 20 years of the SMART protein domain annotation resource. Nucleic Acids Res 46:D493–D496. https://doi.org/10.1093/nar/gkx922
Lijavetzky D, Carbonero P, Vicente-Carbajosa J (2003) Genome-wide comparative phylogenetic analysis of the rice and Arabidopsis Dof gene families. BMC Evol Biol 3:17. https://doi.org/10.1186/1471-2148-3-17
Liu Y, Liu N, Deng X et al (2020) Genome-wide analysis of wheat DNA-binding with one finger (Dof) transcription factor genes: evolutionary characteristics and diverse abiotic stress responses. BMC Genomics 21. https://doi.org/10.1186/s12864-020-6691-0
Liu J, Meng Q, Xiang H, et al (2021) Genome-wide analysis of Dof transcription factors and their response to cold stress in rice (Oryza sativa L.). BMC Genomics 22. https://doi.org/10.1186/s12864-021-08104-0
Löytynoja A (2021) Phylogeny-aware alignment with PRANK and PAGAN. Methods Mol Biol 2231:17–37. https://doi.org/10.1007/978-1-0716-1036-7-2
Ma J, Li M-Y, Wang F, Tang J, Xiong A-S (2015) Genome-wide analysis of Dof family transcription factors and their responses to abiotic stresses in Chinese cabbage. BMC Genomics 16. https://doi.org/10.1186/s12864-015-1242-9
Qin H, Wang J, Chen X et al (2019) Rice OsDOF15 contributes to ethylene-inhibited primary root elongation under salt stress. New Phytol 223:798–813. https://doi.org/10.1111/nph.15824
Qin L, Hu Y, Wang J et al (2021) Insights into angiosperm evolution, floral development and chemical biosynthesis from the Aristolochia fimbriata genome. Nat Plants 7:1239–1253. https://doi.org/10.1038/s41477-021-00990-2
Rueda-Romero P, Barrero-Sicilia C, Gómez-Cadenas A et al (2012) Arabidopsis thaliana DOF6 negatively affects germination in non-after-ripened seeds and interacts with TCP14. J Exp Bot 63(5):1937–1949. https://doi.org/10.1093/jxb/err388
Ruta V, Longo C, Lepri A et al (2020) The DOF transcription factors in seed and seedling development. Plants 9:218. https://doi.org/10.3390/plants9020218
Salehi B, Zakaria ZA, Gyawali R et al (2019) Piper species: a comprehensive review on their Phytochemistry, biological activities and applications. Molecules 24:1364. https://doi.org/10.3390/molecules24071364
Shaw LM, Mclntyre CL, Gresshoff PM, Xue G-P (2009) Members of the Dof transcription factor family in Triticum aestivum are associated with light-mediated gene regulation. Funct Integr Genomics 9:485–498. https://doi.org/10.1007/s10142-009-0130-2
Skirycz A, Radziejwoski A, Busch W et al (2008) The DOF transcription factor OBP1 is involved in cell cycle regulation in Arabidopsis thaliana. The Plant J 56:779–792. https://doi.org/10.1111/j.1365-313X.2008.03641.x
Subramanian B, Gao S, Lercher MJ et al (2019) Evolview v3: a webserver for visualization, annotation, and management of phylogenetic trees. Nucleic Acids Res 47:W270–W275. https://doi.org/10.1093/nar/gkz357
Sun Z, Guo T, Liu Y, Liu Q, Fang Y (2015) The roles of Arabidopsis CDF2 in transcriptional and posttranscriptional regulation of primary MicroRNAs. PLoS Genet 11:e1005598. https://doi.org/10.1371/journal.pgen.1005598
Suseela Bhai R, Anandaraj M, Sarma YR et al (2007) Screening of black pepper (Piper nigrum L.) germplasm for resistance to foot rot disease caused by Phytophthora capsici Leonian. J Spices Aromatic Crops 16(2):115–117
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38:3022–3027. https://doi.org/10.1093/molbev/msab120
Umemura Y, Ishiduka T, Yamamoto R, Esaka M (2004) The Dof domain, a zinc finger DNA-binding domain conserved only in higher plants, truly functions as a Cys2/Cys2 Zn finger domain. Plant J 37:741–749. https://doi.org/10.1111/j.1365-313x.2003.01997.x
Wang D, Zhang Y, Zhang Z, Zhu J, Yu J (2010) KaKs_Calculator 2.0: a toolkit incorporating gamma-series methods and sliding window strategies. Genomics Proteom Bioinform 1:77–80. https://doi.org/10.1016/S1672-0229(10)60008-3
Wang Y, Tang H, Debarry JD et al (2012) MCScanX: a toolkit for detection and evolutionary analysis of gene synteny and collinearity. Nucleic Acids Res 40:e49. https://doi.org/10.1093/nar/gkr1293
Xu P, Chen H, Ying L, Cai W (2016) AtDOF5.4/OBP4, a DOF transcription factor gene that negatively regulates cell cycle progression and cell expansion in Arabidopsis thaliana. Sci Rep 6. https://doi.org/10.1038/srep27705
Xu P, Chen H, Cai W (2020) Transcription factor CDF4 promotes leaf senescence and floral organ abscission by regulating abscisic acid and reactive oxygen species pathways in Arabidopsis. EMBO Rep 21(7):e48967. https://doi.org/10.15252/embr.201948967
Yanagisawa S (1995) A novel DNA-binding domain that may form a single zinc finger motif. Nucleic Acids Res 23:3403–3410. https://doi.org/10.1093/nar/23.17.3403
Yanagisawa S (2002) The Dof family of plant transcription factors. Trends Plant Sci 7:555–560. https://doi.org/10.1016/s1360-1385(02)02362-2
Yanagisawa S (2004) Dof domain proteins: plant-specific transcription factors associated with diverse phenomena unique to plants. Plant Cell Physiol 45:386–391. https://doi.org/10.1093/pcp/pch055
Yanagisawa S, Lzui K (1993) Molecular cloning of two DNA-binding proteins of maize that are structurally different but interact with the same sequence motif. J Biol Chem 268:16028–16036. https://doi.org/10.1016/S0021-9258(18)82353-5
Yanagisawa S, Schmidt RJ (1999) Diversity and similarity among recognition sequences of Dof transcription factors. Plant J 17:209–214. https://doi.org/10.1046/j.1365-313x.1999.00363.x
Yanagisawa S, Sheen J (1998) Involvement of maize Dof zinc finger proteins in tissue-specific and light-regulated gene expression. Plant Cell 10:75–89. https://doi.org/10.1105/tpc.10.1.75
Yang X, Tuskan GA, Cheng ZM (2006) Divergence of the Dof gene families in poplar, Arabidopsis, and rice suggests multiple modes of gene evolution after duplication. Plant Physiol 142:820–830. https://doi.org/10.1104/pp.106.083642
Yu H, Ma Y, Lu Y, Yue J, Ming R (2021) Expression profiling of the Dof gene family under abiotic stresses in spinach. Sci Rep 11. https://doi.org/10.1038/s41598-021-93383-6
Zeng HC, Ho HH, Zheng FC (2005) Pythium vexans causing patch canker of rubber trees on Hainan Island, China. Mycopathologia 159(4):601–606. https://doi.org/10.1007/s11046-005-5258-6
Zhou Y, Cheng Y, Wan C et al (2020) Genome-wide characterization and expression analysis of the Dof gene family related to abiotic stress in watermelon. Peer J 8:e8358. https://doi.org/10.7717/peerj.8358
Zou H-F, Zhang Y-Q, Wei W et al (2013) The transcription factor AtDOF4.2 regulates shoot branching and seed coat formation in Arabidopsis. Biochem J 449:373–388. https://doi.org/10.1042/BJ20110060
Zou Z, Zhu J, Zhang X (2019) Genome-wide identification and characterization of the Dof gene family in cassava (Manihot esculenta). Gene 687:298–307. https://doi.org/10.1016/j.gene.2018.11.053
Acknowledgments
We are very grateful to the spice and Beverage Research Institute of Chinese Academy of Tropical Agricultural Sciences for the technical support, and to all the members of our laboratory.
Funding
This research was founded by the Innovation Platform for Academicians of Hainan Province (No. YSPTZX202154), and China Agriculture Research System of MOF and MARA (NO. CARS-11). We would to thank all authors contributing to this manuscript.
Author information
Authors and Affiliations
Contributions
J.L. conceived and supervised this study. J.L., and F.Z. designed the experiments. F.Z. carried out the experiments. F.Z. analyzed and interpreted the data. D.Y. and F.Z. conduct data collation and chart modification. J.L., R.F., L.Y., L.H., and F.S. participated in the discussion and provided valuable advice and practical contributions. J.L. and F.Z. wrote the manuscript. All authors reviewed, edited, and approved the final manuscript. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Consent of publication
Not applicable.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 29 kb)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, F., Fan, R., Yan, L. et al. Genome-wide identification of black pepper (Piper nigrum L.) Dof gene family and the differential gene screening in resistance to Phytophthora capsici. Biologia 78, 655–666 (2023). https://doi.org/10.1007/s11756-022-01232-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01232-2