Abstract
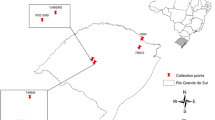
Seven ISSR and eight SRAP primer combinations were used to evaluate the genetic variation within and among nine natural populations of Hertia cheirifolia growing wild in different bioclimatic and geographic regions in Tunisia. The level of genetic diversity within populations varied according to populations and DNA-based markers. A high level of intra-population diversity with ISSR markers (P = 66.34%, H = 0.271, I = 0.394), and a moderate level of genetic diversity with SRAP markers (P = 42.97%, H = 0.164, I = 0.243) were observed. Based on combined data, an intermediate level of polymorphism was observed (P = 59.72%, H = 0.216, I = 0.322).The Mantel test indicated that the genetic differentiation among populations was not correlated to their geographic isolation. For ISSR, SRAP and combined markers, the molecular variation assessed by AMOVA indicated that the most genetic variation resides within populations (71.79%, 51.48% and 65%, respectively) and a high genetic differentiation among populations (ΦST = 0.485, 0.282 and 0.349, respectively). The UPGMA clustering of populations was not related to their geographic and bioclimatic appurtenances. The significant genetic differentiation associated with the low genetic divergence among H. cheirifolia populations detected by both markers suggests that the fragmentation of populations occurred recently. These observations recommend that the appropriate strategy to preserve the gene pool of the species is limiting anthropic pressure and natural habitats fragmentation. Conservation strategies for preserving this plant species are needed in order to increase the size of populations, to avoid genetic erosion and to ensure the in-situ and ex-situ conservations of the species.



Similar content being viewed by others
References
Ammar S, Edziri H, Mahjoub MA, Chatter R, Bouraoui A, Mighri Z (2009) Spasmolytic and anti-inflammatory effects of constituents from Hertia cheirifolia. Phytomed 16(12):1156–1161. https://doi.org/10.1016/j.phymed.2009.03.012
Bagheri A, Abbasi S, Mahmoodi M, Roofigar AA, Blattner FR (2020) Genetic structure and conservation status of Astragalus subrecognitus (Fabaceae): a very rare and narrow endemic species. Plant Ecol Evol 153(1):101–107. https://doi.org/10.5091/plecevo.2020.1648
Bonnet E, Van de Peer Y (2002) zt: a software tool for simple and partial Mantel tests. J Stat Software 7:1–12. https://doi.org/10.18637/jss.v007.i10
Booy G, Hendriks RJJ, Smulders MJM, Van Groenendael JM, Vosman B (2000) Genetic Diversity and the Survival of Populations. Plant boil 2:379–395. https://doi.org/10.1055/s-2000-5958
Duwe VK, Muller LA, Reichel K, Zippel E, Borsch T, Ismail SA (2018) Genetic structure and genetic diversity of the endangered grassland plant Crepis mollis (Jacq.) Asch. as a basis for conservation management in Germany. Conserv Genet 19(3): 527–543. https://doi.org/10.1007/s10592-017-1025-8
Ellstrand NC, Elam DR (1993) Population genetic consequences of small population size: Implications for plant conservation. Ann Rev Ecol Syst 24:217–242. https://doi.org/10.1146/annurev.es.24.110193.001245
Emberger L (1966) Une classification biogéographique des climats. Recherches et Travaux des Laboratoires de Géologie, Botanique et Zoologie. Faculté des Sciences Montpellier (France) 7:1–43
Excoffier L, Smouse PE, Quattro JM (1992) Analysis of molecular variance inferred from metric distances among DNA haplotypes: application to human mitochondrial DNA restriction data. Genet 131:479–491. https://doi.org/10.1093/genetics/131.2.479
Hämälä T, Mattila TM, Savolainen O (2018) Local adaptation and ecological differentiation under selection, migration, and drift in Arabidopsis lyrata. Evol 72(7):1373–1386. https://doi.org/10.1111/evo.13521
Hamrick JL, Murawski DA, Nason JD (1993) The influence of seed dispersal mechanisms on the genetic structure of tropical tree populations. Vegetatio 107:281–297. https://doi.org/10.1007/BF00052230
Jiménez-López FJ, Ortiz M, Berjano R, Talavera S, Terrab A (2016) High population genetic substructure in Hypochaeris leontodontoides (Asteraceae), an endemic rupicolous species of the Atlas Mountains in NW Africa. Alp Bot 126:73–85. https://doi.org/10.1007/s00035-016-0163-9
Jiménez-Mejías P, Fernández-Mazuecos M, Gutiérrez L, Álvarez I, Vargas P (2017) Narrow endemics in Mediterranean scrublands: high gene flow buffers genetic impoverishment in the annual monospecific Castrilanthemum (Asteraceae)΄. Biol Cons 26(11):2607–2626. https://doi.org/10.1007/s10531-017-1374-0
King LM, Schaal BA (1990) Genotypic variation within asexual lineages of Taraxacum officinale. Proc Natl Acad Sci USA 87:998–1002. Doi: https://doi.org/10.1073/pnas.87.3.998
Koopman MM, Carstens BC (2010) Conservation genetic inferences in the carnivorous pitcher plant Sarracenia alata (Sarraceniaceae). Conserv Genet 11(5):2027–2038. https://doi.org/10.1007/s10592-010-0095-7
Kovach WL (1999) A Multivariate Statistical Package for Windows, ver 3.1. Kovach Computing Services, Pentraeth, UK
Le Floc’h E (1983) Contribution à une étude ethnobotanique de la flore tunisienne. Imprimerie officielle de la république Tunisienne, Tunisie
Li G, Quiros CF (2001) Sequence-related amplified polymorphism (SRAP), a new marker system based on a simple PCR reaction: its application to mapping and gene tagging in Brassica. Theor Appl Genet 103:455–461. https://doi.org/10.3732/apps.1400017
Liu C, Xue GE, Cheng B, Wang X, He J, Liu GH, Yang WJ (2015) Genetic diversity analysis of Capparis spinosa L. populations by using ISSR markers. Genet Mol Res 14:16476–16483. https://doi.org/10.4238/2015.December.9.19
Luan S, Chiang TY, Gong XUN (2006) High genetic diversity vs. low genetic differentiation in Nouelia insignis (Asteraceae), a narrowly distributed and endemic species in China, revealed by ISSR fingerprinting. Ann Bot 98(3):583–589. https://doi.org/10.1093/aob/mcl129
Majouli K, Hamdi A, Msaada K, Kenani A (2017) A bioactivity guided study on the antibacterial activity of Hertia cheirifolia L. extracts. Microb Pathog 106:113–118. https://doi.org/10.1016/j.micpath.2016.10.015
Maurice T, Matthies D, Muller S, Colling G (2016) Genetic structure of colline and montane populations of an endangered plant species. AoB Plants 8. https://doi.org/10.1093/aobpla/plw057
McGlaughlin M, Karoly K, Kaye T (2002) Genetic variation and its relationship to population size in reintroduced populations of pink sand verbena, Abronia umbellate subsp. Breviflora (Nyctaginaceae). Conserv Genet 3(4): 411–420. https://doi.org/10.1023/A:1020507416654
Meena B, Singh N, Mahar KS, Sharma YK, Rana TS (2019) Molecular analysis of genetic diversity and population genetic structure in Ephedra foliata: an endemic and threatened plant species of arid and semi-arid regions of India. Physiol Mol Biol Plants 25(3):753–764. https://doi.org/10.1007/s12298-019-00648-6
Messaoud C, Afif M, Boulila A, Rejeb MN, Boussaid M (2007) Genetic variation of Tunisian Myrtus communis L. (Myrtaceae) populations assessed by isozymes and RAPDs. Ann For Sci 64:845–853. https://doi.org/10.1051/forest:2007061
Müller CM, Schulz B, Lauterbach D, Ristow M, Wissemann V, Gemeinholzer B (2017) Geropogon hybridus (L.) Sch. Bip.(Asteraceae) exhibits micro-geographic genetic divergence at ecological range limits along a steep precipitation gradient. Plant Syst Evol 303(1):91–104. https://doi.org/10.1007/s00606-016-1354-y
Nei M (1972) Genetic distance between populations. Am Nat 106:283–292
Nybom H (2004) Comparison of different nuclear DNA markers for estimating intraspecific genetic diversity in plants. Mol Ecol 13(5):1143–1155. https://doi.org/10.1111/j.1365-294X.2004.02141.x
Petrova G, Petrov S, Delcheva M, Bancheva S (2017) Genetic diversity and conservation of Bulgarian endemic Verbascum tzar-borisii (Scrophulariaceae). Ann Bot Fenn 54:307–316
Pometti CL, Bessega CF, Vilardi JC, Ewens M, Saidman BO (2016) Genetic variation in natural populations of Acacia visco. Plant Syst Evol 302:901–910. https://doi.org/10.1007/s00606-016-1306-6
Pottier-Alapetite G (1981) In: Tunisienne IOR (ed) Flore de la Tunisie: Angiospermes Dicotylédones, Gamopétales. Scientifiques Tunisiennes, Ministère de l’Enseignement Supe´rieur et de la Recherche Scientifique, Ministère de l’Agriculture, Tunis
Rahali N, Mehdi S, Younsi F, Boussaid M, Messaoud C (2017) Antioxidant, α-amylase and acetylcholinesterase inhibitory activities of Hertia cheirifolia essential oils: Influence of plant organs and seasonal variation. Int J Food Prop 20: 1637–1651. https://doi.org/1080/10942912.2017.1352597
Rahali N, Younsi F, Boussaid M, Messaoud C (2019) Chemical variability of Hertia cheirifolia (L.) Kuntze essential oils and incidence on antioxidant and anticholinesterase activities. J Essent Oil Res 1622603. https://doi.org/10.1080/10412905
Song MF, Guan YH, Zhang Y, Li HT, Zhang LX, Zhang ZL (2021) Genetic Diversity Assessment of a Chinese Medicinal Endemic Species, Aspidopterys obcordata var. obcordata, by Combined Molecular Marker Methods (ISSR & SRAP). Biochem Genet 59:283–299. https://doi.org/10.1007/s10528-020-10001-2
Talebi SM, Askary M, Khalili N, Matsyura A, Ghorbanpour M, Kariman K (2021) Genetic structure and essential oil composition in wild populations of Salvia multicaulis Vahl. Biochem Syst Ecol 96. https://doi.org/10.1016/j.bse.2021.104269
Tiwari G, Singh R, Singh N, Choudhury DR, Paliwal R, Kumar A, Gupta V (2016) Study of arbitrarily amplified (RAPD and ISSR) and gene targeted (SCoT and CBDP) markers for genetic diversity and population structure in Kalmegh [Andrographis paniculata (Burm. f.) Nees]. Ind Crops Prod 86:1–11. https://doi.org/10.1016/j.indcrop.2016.03.031
Toczydlowski RH, Waller DM (2019) Drift happens: Molecular genetic diversity and differentiation among populations of jewelweed (Impatiens capensis Meerb.) reflect fragmentation of floodplain forests. Mol Ecol 28(10):2459–2475. https://doi.org/10.1111/mec.15072
Uzun A, Yesiloglu T, Aka-Kacar Y, Tuzcu O, Gulsen O (2009) Genetic diversity and relationships within Citrus and related genera based on sequence related amplified polymorphism markers (SRAPs). Sci Hortic 121(3):306–312. https://doi.org/10.1016/j.scienta.2009.02.018
Vidaller C, Dutoit T, Ibrahim Y, Hanslin HM, Bischoff A (2018) Adaptive differentiation among populations of the Mediterranean dry grassland species Brachypodium retusum: The role of soil conditions, grazing, and humidity. Am J Bot 105(7):1123–1132. https://doi.org/10.1002/ajb2.1116
Vogt R, Oberprieler Ch (2008) Chromosome numbers of North African phanerogams 6 VIII. More counts in Compositae. Willdenowia 38:497–519. https://doi.org/10.3372/wi.38.38210
Wang Q, Zhang ML, Yin LK (2016) Genetic diversity and population differentiation of Capparis spinosa (Capparaceae) in Northwestern China. Biochem Syst Ecol 66: 1–7. https://doi.org/1016/j.bse.2016.02.034
Yeh FC, Yang R, Boyle T (1999) POPGENE: Microsoft Windowbased freeware for population genetic analysis: version 1.3: manual.Edmonton: University of Alberta. 29p
Younsi F, Nahali R, Mehdi S, Boussaid M, Messaoud C (2018) Relationship between chemotypic and genetic diversity of natural populations of Artemisia herba-alba Asso growing wild in Tunisia. Phytochem 148:48–56. https://doi.org/10.1016/j.phytochem.2018.01.014
Zietkiewicz E, Rafalski A, Labuda D (1994) Genome fingerprinting by simple sequence repeat (SSR)-anchored polymerase chain reaction amplification. Genomics 20:176–183. https://doi.org/10.1006/geno.1994.1151
Zeng X, Michalski SG, Fischer M, Durka W (2012) Species diversity and population density affect genetic structure and gene dispersal in a subtropical understory shrub. J Plant Ecol 5(3):270–278. https://doi.org/10.1093/jpe/rtr029
Acknowledgements
The research was supported by the Tunisian Ministry of Scientific Research and Technology and the National Institute of Applied Science and Technology University of Carthage (UR17ES22).
Author information
Authors and Affiliations
Contributions
NR performed the laboratory works and wrote the draft. IY contributed to revision and interpretation. MB designed the study and reviewed the manuscript. CM conducted the data analysis and reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest/competing interests
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rahali, N., Yangui, I., Boussaid, M. et al. Assessment of genetic diversity and population structure of the endemic Hertia cheirifolia (L.) Kuntze based on ISSR and SRAP molecular markers. Biologia 77, 3429–3439 (2022). https://doi.org/10.1007/s11756-022-01166-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01166-9