Abstract
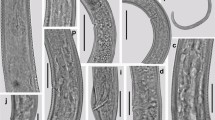
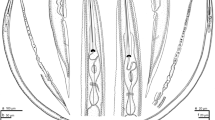
During a survey on the biodiversity of plant-parasitic nematodes in the south of Iran, Hoplolaimus indicus from the grass was recovered. Hoplolaimus are ectoparasites or semiendoparasites of various crops. Hoplolaimus indicus was studied using morphological and molecular techniques. Hoplolaimus indicus is identified based on the lip region with three annuli, five clear and one obscure pharyngeal nucleus, excretory pore at the isthmus level, and spicules with 42–46 µm long. The scan electron microscopy (SEM) observation revealed basal annuli of the lip region divided into six blocks, and one incisure along the body starts from 7–8 annuli posterior to the lip region ended at the tail tip. Principal component analysis (PCA) showed that the present H. indicus and the population of the same species from Iran stand very close to each other. Besides, H. indicus and H. columbus stand separately. Nblast for the 28S rDNA revealed 100% identity of the tested species and other H. indicus from Iran (MF421900) and Myanmar (MN462843). In contrast, the COI of mtDNA showed 97% similarity with several sequences of the American populations of H. columbus (e.g., KP864621). However, the phylogenetic analysis using the mentioned above markers placed H. indicus very close to H. columbus, H. indicus, and H. seinhorsti, particularly using 28S rDNA. Measurements, light microscopy (LM), SEM, and phylogenetic analysis for H. indicus are given. The COI of mtDNA sequence and SEM observation for H. indicus are reported for the first time. In conclusion, the result indicated that 28S rDNA is not a good marker to distinguish H. indicus from the close species, including H. seinhorsti and H. columbus.





Similar content being viewed by others
Data availability
This material is the authors’ own original work, which has not been previously published elsewhere and has no conflict of interest. Two slides of Hoplolaimus indicus were deposited in the Nematology laboratory collection of the University of Limpopo, South Africa. One slide was deposited in the Nematode Collection of the Department of Animal Biology, Plant Biology, and Ecology of the University of Jaén (Spain).
References
Abolafia J (2015) A low-cost technique to manufacture a container to process meiofauna for scanning electron microscopy. Microsc Res Tech 78:771–776. https://doi.org/10.1002/jemt.22538
Abolafia J, Peña-Santiago R (2017) On the identity of Chiloplacus magnus Rashid & Heyns, 1990 and C. insularis Orselli & Vinciguerra, 2002 (Rhabditida: Cephalobidae), two confusable species. Nematology 19:1017–1034. https://doi.org/10.1163/15685411-00003104
Addinsoft, (2007) XLSTAT, Analyse de données et statistique avec MS Excel. Addinsoft, New York
Ahmadi Mansourabad M, Ghaderi R, Kashi L, Karegar A (2016) Observations on Hoplolaimus indicus Sher, 1963 and Hoplolaimus seinhorsti Luc, 1958 (Nematoda: Hoplolaimidae) from Southern Iran. J Crop Protec 5:189–201
Ali Ramaji F, Pourjam E, Karegar A (2006) Some tylenchids associated with pistachio and almond trees in Iran. Proceedings of IVthIS on Pistachio and Almonds. Acta Hort 726:659–666. https://doi.org/10.17660/ActaHortic.2006.726.114
Anderson RV (1983) Morphological characteristics of Hoplolaimus indicus Sher, 1963 in Canada, a parasite of wild rice. J Nematol 15:367–369
Archidona-Yuste A, Cai R, Cantalapiedra-Navarrete C, Carreira JA, Rey A, Viñegla B, Liébanas G, Palomares-Rius JE, Castillo P (2020) Morphostatic speciation within the dagger nematode Xiphinema hispanum-complex species (Nematoda: Longidoridae). Plants 9:1649. https://doi.org/10.3390/plants9121649
Baermann G (1917) Eine einfache Methode zur Auffindung von Ankylostomum (Nematoden) Larven in Erdproben. Geneeskunding Tijdschrift Voor Nederlandsch-Indië 57:131–137
Barooti S, Geraert E (1994) Some plant parasitic nematodes (Tylenchida) from Iran. In: Abstracts of the 22th International Symposium of the European Society of the European Society of Nematologists, Gent, Belgium, p 33
Barooti S, Tanha Maafi Z, Kheiri A, Gol Mohammadzade Khiaban N (2002) Plant parasitic nematodes of southern regions of Baluchistan, Iran. In: Proceedings of 15th Iranian Plant Protection Congress. Volume II: Plant Diseases and Weeds, Kermanshah, Iran, pp 304–305
Cantalapiedra-Navarrete C, Navas-Cortés JA, Liébanas G, Vovlas N, Subbotin SA, Palomares-Rius JE, Castillo P (2013) Comparative molecular and morphological characterisations in the nematode genus Rotylenchus: Rotylenchus paravitis n. sp., an example of cryptic speciation. Zool Anz 252:246–268. https://doi.org/10.1016/j.jcz.2012.08.002
Chaturvedi Y, Khera S (1979) Studies on taxonomy, biology and ecology of nematodes associated with jute crop. Tech Monog Zool Surv India 2:1–105
Cobb NA (1913) New nematode genera found inhabiting fresh water and non-brackish soils. J Wash Acad Sci 3:432–444
Darriba D, Taboada GL, Doallo R, Posada D (2012) jModelTest 2: more models, new heuristics and parallel computing. Nat Methods 9:772. https://doi.org/10.1038/nmeth.2109
De Grisse A (1969) Redescription ou modifications de quelques techniques utilisées dans l’étude des nematodes phytoparasitaire]. Mededelingen Rijksfaculteit der Landbouwwetenschappen Gent 34:351–369
De Man JG (1876) Onderzoekingen over vrij in de aarde levende Nematoden. Tijdschrift der Nederlandsche Dierkundige Vereeniging 2:78–196
De Man JG (1881) Die einheimischen, frei in der reinen Erde und im süssen Wasser lebenden Nematoden. Vorläufiger Bericht und descriptiv-systematischer Theil. Tijdschrift Nederlandsche Dierkundige Vereeniging 5:1–104
De Ley P, Felix MA, Frisse LM, Nadler SA, Sternberg PW, Thomas WK (1999) Molecular and morphological characterisation of two reproductively isolated species with mirror-image anatomy (Nematoda: Cephalobidae). Nematology 2:591–612. https://doi.org/10.1163/156854199508559
Derycke S, Vanaverbeke G, Rigaux A, Backljau T, Moens T (2010) Exploring the use of cytochrome oxidase c subunit 1 (COI) for DNA barcoding of free-living marine nematodes. PLoS ONE 10:1–9. https://doi.org/10.1371/journal.pone.0013716
Divsalar N, Shokoohi E, Marais M, Mashela PW (2020) Molecular and morphological characters of Helicotylenchus species from Iran with a note on the identity of H. pseudorobustus. Zootaxa 4789(1):266–280. https://doi.org/10.11646/zootaxa.4789.1.10
Fassuliotis G (1974) Host range of the Columbia lance nematode Hoplolaimus columbus. Plant Dis Rep 58:1000–1002
Filipjev IN (1934) The classification of the free-living nematodes and their relation to the parasitic nematodes. Smithsonian Misc Collect 89(6):1–63
Fortuner R (1991) The Hoplolaiminae. In: Nickle WR (ed) Manual of agricultural Nematology. Marcel Dekker Inc., New York, pp 669–719
Gazaway WS, McLean KS (2003) A survey of plant parasitic nematodes associated with cotton in Alabama. J Cotton Sci 7:1–7
Golden AM, Minton NA (1970) Description and larval heteromorphism of Hoplolaimus concaudajuvencus, n. sp. (Nematoda: Hoplolaimidae). J Nematol 2(2):161–166
Guindon S, Gascuel O (2003) A simple, fast and accurate method to estimate large phylogenies by maximum-likelihood. Syst Biol 52:696–704. https://doi.org/10.1080/10635150390235520
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/ 98NT. Nucl Acids Symp Ser 41:95–98
Handoo ZA, Golden M (1992) A key and diagnostic compendium to the species of the genus Hoplolaimus Daday, 1905 (Nematoda: Hoplolaimidae). J Nematol 24:45–53
Holguin CM, Baeza JA, Mueller JD, Agudelo P (2015) High genetic diversity and geographic subdivision of three lance nematode species (Hoplolaimus spp.) in the United States. Ecol Evol 5(14):2929–2944. https://doi.org/10.1002/ece3.1568
Khan E, Chawla ML (1975) CIH description of plant parasitic nematodes Set 5. Commonwealth Institute of Helminthology, St. Albans
Luc M (1958) Nematodes and wilting in cotton in Southwestern Madagascar. Cot Fibr Trap 13:239–256
Ma X, Agudelo P, Mueller JD, Knap HT (2011) Molecular characterization and phylogenetic analysis of Hoplolaimus stephanus. J Nematol 43:25–34
Ma X, Robbins RT, Bernard EC, Holguin CM, Agudelo P (2019) Morphological and molecular characterisation of Hoplolaimus smokyensis n. sp. (Nematoda: Hoplolaimidae), a lance nematode from Great Smoky Mountains National Park, USA. Nematology 21:923–935. https://doi.org/10.1163/15685411-00003266
Martin SB, Mueller JD, Saunders JA, Jones WI (1994) A survey of South Carolina cotton fields for plant-parasitic nematodes. Plant Dis 78:717–719. https://doi.org/10.1094/PD-78-0717
Nguyen TH, Bui QD, Trinh PQ (2015) Description of Hoplolaimus bachlongviensis sp. n. (Nematoda: Hoplolaimidae) from banana soil in Vietnam. Biodiv Data J 3:e6523. https://doi.org/10.3897/BDJ.3.e6523
Riascos-Ortiz D, Mosquera-Espinosa AT, De Agudelo FV, de Oliveira C, Muñoz-Flórez JE (2019) Morpho-molecular characterization of Colombian and Brazilian populations of Rotylenchulus associated with Musa spp. J Nematol 51:e2019-47. https://doi.org/10.21307/jofnem-2019-047
Robbins RT (1982) Description of Hoplolaimus magnistylus n. sp. (Nematoda: Hoplolaimidae). J Nematol 14(4):500–506
Ronquist F, Huelsenbeck J (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574. https://doi.org/10.1093/bioinformatics/btg180
Schuurmans Stekhoven JH, Teunissen RJH (1938) Nématodes libres terrestres. Expl Parc Nat Albert, Mission de Witte (1933–35) Bruxelles. Institut des Parcs Nationaux du Congo Belge 2, pp 1–229
Shamsi MA (1979) Basirolaimus gen.n. (Nematoda: Hoplolaimidae) with the description of Basirolaimus sacchari n.sp. from India. Nematol Mediterr 7:15–19
Sher SA (1963) Revision of the Hoplolaiminae (Nematoda). II. Hoplolaimus Daday, 1905 and Aorolaimus n. gen. Nematologica 9:267–295. https://doi.org/10.1163/187529263X00476
Shokoohi E, Marais M, Fourie H, Panahi H (2018) Morphological and molecular characterization of three Helicotylenchus species from Iran. Zootaxa 4388:207–220. https://doi.org/10.11646/zootaxa.4388.2.4
Shokoohi E, Abolafia J, Mashela PW, Divslar N (2019) New data on known species of Hirschmanniella and Pratylenchus (Rhabditida, Pratylenchidae) from Iran and South Africa. J Nematol 51:e2019-41. https://doi.org/10.21307/jofnem-2019-041
Shokoohi E, Abolafia J, Mashela PW (2020) Redescription of Paratrophurus anomalus from South Africa. Nematology 22:543–554. https://doi.org/10.1163/15685411-00003322
Shokoohi E, Marais M, Mashela PW, Divsalar N (2021) Variation within Helicotylenchus vulgaris. Biologia 76(2):549–563. https://doi.org/10.2478/s11756-020-00551-6
Siddiqi MR (2000) Tylenchida parasites of plants and insects, 2nd edn. CABI Publishing, Wallingford, p 833
Singh MK (1995) Studies on the morphology and bio-ecology of nematode fauna of Rewa. PhD thesis. University of Rewa, India, pp 317
Steiner G (1945) Helicotylenchus, a new genus of plant-parasitic nematodes and its relationship to Rotylenchus Filipjev. Proc Entomol Soc Wash 12:34–38
Straube D, Juen A (2013) Storage and shipping of tissue samples for DNA analyses: A case study on earthworms. Europ J Soil Biol 57:13–18. https://doi.org/10.1016/j.ejsobi.2013.04.001
Thompson JD, Higgins DG, Gibson TJ (1994) CLUSTAL W: improving the sensitivity of progressive multiple sequence alignment through sequence weighting, position-specific gap enalties and weight matrix choice. Nuc Acids Res 22:4673–4680
Thorne G (1935) Notes on free-living and plant-parasitic nematodes II. Proc Helm Soc Wash 2:96–98
von Daday E (1905) Untersuchungen über die Süsserwasser-Mikrofauna Paraguays. Zool Stuttgart 18:1–349
Vovlas N (1983) Morphology of Hoplolaimus seinhorsti as seen by scanning electron microscope. Nematol Medit 11:145–149
Wrather JA, Niblack TL, Milam MR (1992) Suvey of plant-parasitic nematodes in Missouri cotton fields. J Nematol 24:779–782
Acknowledgements
The authors thank the research support plans "PAIUJA 2019/2020: EI_RNM02_2019" and "PAIUJA 2021/2022: EI_RNM02_2021" of the University of Jaén, Spain. The SEM pictures were obtained with the assistance of technical staff (Amparo Martínez-Morales) and equipment of “Centro de Instrumentación Científico-Técnica (CICT)” from the University of Jaén. The authors also acknowledge Mr. Hadi Panahi for providing the samples to the Iranian population.
Funding
This study was financially supported by the University of Jaén (Spain) for the SEM photographs. The rest of the project was supported by ES.
Author information
Authors and Affiliations
Contributions
ES conducted the study, identified the species and analysed the data, wrote and revised the manuscript. PWM revised the manuscript. JA revised the manuscript, analyzed the data, taken LM and SEM of the species. ES and JA contributed to the final draft of the manuscript.
Corresponding author
Ethics declarations
Ethical statement
The paper reflects the authors’ own research and analysis in a truthful and complete manner.
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shokoohi, E., Abolafia, J. & Mashela, P.W. Redescription of Hoplolaimus indicus Sher, 1963 (Rhabditida, Hoplolaimidae) from Iran, including the first SEM study of the species. Biologia 77, 2161–2172 (2022). https://doi.org/10.1007/s11756-022-01104-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01104-9