Abstract
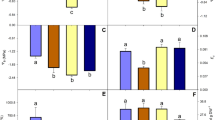
Endemic plants attract attention for their increasing potential for genetic biodiversity, an important phenomenon in all aspects of science. Little is known about the ability of the endangered Turkish endemic plant Cephalaria duzceënsis to generate/remove reactive oxygen species or to adapt its biochemical responses in drought and saline environments. Consequently, changes in growth, relative water content (RWC), chlorophyll fluorescence (Fv/Fm), osmotic potential, hydrogen peroxide (H2O2) and proline contents, lipid peroxidation, and antioxidant enzymes were measured under drought (0, 5, 10, 15% PEG 6000) and salt (0, 75, 150, 225 mM NaCl) stresses. Salt-induced RWC, Fv/Fm, osmotic potential reduction, and lipid peroxidation were enhanced, suggesting that C. duzceënsis is more sensitive to salinity than to drought. However, interestingly, growth increased under both. Although the increase in H2O2 content was greater under drought stress, aside from ascorbate peroxidase, no antioxidant enzyme activity increase was observed under salt stress. Conversely, the increase in peroxidase, ascorbate peroxidase, and catalase activity observed under drought may be related to the better growth performance, less Fv/Fm reduction, better water maintenance, and lower osmotic potential of C. duzceënsis. These results strongly suggest that this endemic plant exhibits moderate drought tolerance and that salt stress has a more detrimental effect on C. duzceënsis than drought stress.





Similar content being viewed by others
References
Abid M, Ali S, Qi LK, Zahoor R, Tian Z, Jiang D, Snider JL, Dai T (2018) Physiological and biochemical changes during drought and recovery periods at tillering and jointing stages in wheat (Triticum aestivum L.). Sci Rep 8:4615. https://doi.org/10.1038/s41598-018-21441-7
Abid G, Ouertani RN, Muhovski Y, Jebara SH, Hidri Y, Ghouili E, Abdelkarim S, Chaieb O, Souissi F, Zribi F, Jebara M (2021) Variation in antioxidant metabolism of faba bean (Vicia faba) under drought stress induced by polyethylene glycol reveals biochemical markers associated with antioxidant defense. Plant Biosyst 155(4):797–806. https://doi.org/10.1080/11263504.2020.1785964
Aebi H (1984) Catalase in vitro. In: Colowick SP, Kaplan NO (eds) Methods in enzymology. Academic Press, Orlando, pp 114–121
Aksoy N, Göktürk RS, Açık L, Çelebi A (2007) Cephalaria duzceënsis (Dipsacaceae), a new species from the western Black Sea region, Turkey. Nord J Bot 25:64–69. https://doi.org/10.1111/j.0107-055X.2007.00100_11.x
Bates LS, Waldren RP, Teare ID (1973) Rapid determination of free proline for water stress studies. Plant Soil 39:205–207. https://doi.org/10.1007/BF00018060
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287. https://doi.org/10.1016/0003-2697(71)90370-8
Bienert GP, Møller ALB, Krsitiansen KA, Schulz A, Møller IM, Schjoerring JK, Jahn TP (2006) Specific aquaporins facilitate the diffusion of hydrogen peroxide across membranes. J Biol Chem 282:1183–1192. https://doi.org/10.1074/jbc.M603761200
Boke Sarikahya N, Goren AC, Kirmizigul S (2019) Simultaneous determination of several flavonoids and phenolic compounds in nineteen different Cephalaria species by HPLC-MS/MS. J Pharm Biomed Anal 173:120–125. https://doi.org/10.1016/j.jpba.2019.05.019
Boke Sarikahya N, Kayce P, Halay E, Göktürk RS, Sümbül H, Kirmizigül S (2013) Phytochemical analysis of the essential oils of 10 endemic Cephalaria species from Turkey. Nat Prod Res 27(9):830–833. https://doi.org/10.1080/14786419.2012.701216
Boke Sarikahya N, Onay Ucar E, Kayce P, Gokturk RS, Sumbul H, Arda N, Kirmizigul S (2015) Fatty acid composition and antioxidant potential of ten Cephalaria species. Rec Nat Prod 9(1):116–123
Bor M, Özdemir F, Türkan I (2003) The effect of salt stres on lipid peroxidation and antioxidants in leaves of sugar beet Beta vulgaris L. and wild beet Beta maritima L. Plant Sci 164:77–84. https://doi.org/10.1016/S0168-9452(02)00338-2
Bradford MM (1976) A rapid and sensitive method for the quantization of microgram quantities of protein utilizing the principle of the protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1006/abio.1976.9999
Cavalcanti FR, Oliveira JTA, Martins-Miranda AS, Viégas RA, Albenísio J, Silveira G (2004) Superoxide dismutase, catalase and peroxidase activities do not confer protection against oxidative damage in salt-stressed cowpea leaves. New Phytol 163:563–571. https://doi.org/10.1111/j.1469-8137.2004.01139.x
Celenk VU, Boke Sarikahya N, Kirmizigul S (2020) Isolation and structural studies on saponins from three Cephalaria species from Anatolia. Chem Nat Compd 56:180–182. https://doi.org/10.1007/s10600-020-02980-w
Choudhury FK, Rivero RM, Blumwald E, Mittler R (2017) Reactive oxygen species, abiotic stress and stress combination. Plant J 90(5):856–867. https://doi.org/10.1111/tpj.13299
Cia MC, Guimaraes ACR, Medici LO, Chabregas SM, Azevedo RA (2012) Antioxidant responses to water deficit by drought tolerant and sensitive sugarcane varieties. Ann Appl Biol 161:313–324. https://doi.org/10.1111/j.1744-7348.2012.00575.x
Cunha JR, Neto MCL, Carvalho FE, Martins MO, Jardim-Messeder D, Margis-Pinheiro M, Silveira JA (2016) Salinity and osmotic stress trigger different antioxidant responses related to cytosolic ascorbate peroxidase knockdown in rice roots. Environ Exp Bot 131:58–67. https://doi.org/10.1016/j.envexpbot.2016.07.002
Demirevska K, Zasheva D, Dimitrov R, Simova-Stoilova L, Stamenova M, Feller U (2009) Drought stress effects on Rubisco in wheat, changes in the Rubisco large subunit. Acta Physiol Plant 31:1129–1138. https://doi.org/10.1007/s11738-009-0331-2
Devi R, Kaur N, Gupta AK (2012) Potential of antioxidant enzymes in depicting drought tolerance of wheat (Triticum aestivum L.). Indian J Biochem Biophys 49(4):257–265
Force L, Critchley C, van Rensen JJ (2003) New fluorescence parameters for monitoring photosynthesis in plants. Photosynth Res 78:17–33. https://doi.org/10.1023/A:1026012116709
Foyer CH, Halliwell B (1976) The presence of glutathione and glutathione reductase in chloroplasts: A proposed role in ascorbic acid metabolism. Planta 133:21–25. https://doi.org/10.1007/BF00386001
Foyer CH, Noctor G (2011) Ascorbate and glutathione: the heart of the redox hub. Plant Physiol 155(1):2–18. https://doi.org/10.1104/pp.110.167569
García-Martí M, Piñero MC, García-Sanchez F, Mestre TC, López-Delacalle M, Martínez V, Rivero RM (2019) Amelioration of the oxidative stress generated by simple or combined abiotic stress through the K+ and Ca2+ supplementation in tomato plants. Antioxidants 8:81. https://doi.org/10.3390/antiox8040081
Hasanuzzaman M, Alam M, Rahman A, Hasanuzzaman M, Nahar K, Fujita M (2014) Exogenous proline and glycine betaine mediated upregulation of antioxidant defense and glyoxalase systems provides better protection against salt-induced oxidative stress in two rice (Oryza sativa L.) varieties. BioMed Res Int:757219. https://doi.org/10.1155/2014/757219
Hasanuzzaman M, Bhuyan M, Zulfiqar F, Raza A, Mohsin SM, Mahmud JA, Fujita M, Fotopoulos V (2020) Reactive oxygen species and antioxidant defense in plants under abiotic stress: Revisiting the crucial role of a universal defense regulator. Antioxidants 9(8):681. https://doi.org/10.3390/antiox9080681
Hasanuzzaman M, Hossain MA, Fujita M (2011) Nitric oxide modulates antioxidant defense and the methylglyoxal detoxification system and reduces salinity-induced damage of wheat seedlings. Plant Biotechnol Rep 5:353–365. https://doi.org/10.1007/s11816-011-0189-9
Hasanuzzaman M, Nahar K, Anee TI, Khan MIR, Fujita M (2018) Silicon-mediated regulation of antioxidant defense and glyoxalase systems confers drought stress tolerance in Brassica napus L. S Afr J Bot 115:50–57. https://doi.org/10.1016/j.sajb.2017.12.006
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts, I. kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Hernández JA, Ferrer MA, Jiménez A, Barceló AR, Sevilla F (2001) Antioxidant systems and O2.−/H2O2 production in the apoplast of pea leaves. Its relation with salt-induced necrotic lesions in minor veins. Plant Physiol 127:817–831. https://doi.org/10.1104/pp.010188
Hunt R, Causton DR, Shipley B, Askew AP (2002) A modern tool for classical plant growth analysis. Ann Bot 90:485–488. https://doi.org/10.1093/aob/mcf214
Hussain MI, Elnaggar A, El-Keblawy A (2020) Eco-physiological adaptations of Salsola drummondii to soil salinity: role of reactive oxygen species, ion homeostasis, carbon isotope signatures and anti-oxidant feedback. Plant Biosyst. https://doi.org/10.1080/11263504.2020.1840453
Ibrahim W, Qiu CW, Zhang C, Cao F, Shuijin Z, Wu F (2019) Comparative physiological analysis in the tolerance to salinity and drought individual and combination in two cotton genotypes with contrasting salt tolerance. Physiol Plant 165(2):155–168. https://doi.org/10.1111/ppl.12791
IUCN (2001) IUCN Red List categories. Version 3.1. Prepared by the IUCN Species Survival Commission. World Conservation Union, Gland, Switzerland and Cambridge, United Kingdom
Jaleel CA, Manivannan P, Wahid A, Farooq M, Al-Juburi J, Somasundaram R, Panneerselvam R (2009) Drought stress in plants: A review on morphological characteristics and pigments composition. Int J Agric Biol 11:100–105
Karaoğlu MM (2006) Cephalaria syriaca addition to wheat flour dough and effect on rheological properties. Int J Food Sci Technol 41:37–46. https://doi.org/10.1111/j.1365-2621.2006.01272.x
Kaur G, Asthir B (2015) Proline: a key player in plant abiotic stress tolerance. Biol Plant 59:609–619. https://doi.org/10.1007/s10535-015-0549-3
Kaya C (2021) Nitrate reductase is required for salicylic acid-induced water stress tolerance of pepper by upraising the AsA-GSH pathway and glyoxalase system. Physiol Plant 172:351–370. https://doi.org/10.1111/ppl.13153
Kaya C, Ashraf M (2021) Nitric oxide is required for aminolevulinic acid-induced salt tolerance by lowering oxidative stress in maize (Zea mays). J Plant Growth Regul 40:617–627. https://doi.org/10.1007/s00344-020-10126-z
Kaya C, Ashraf M, Alyemenic MN, Ahmad P (2020) The role of endogenous nitric oxide in salicylic acid-induced up-regulation of ascorbate-glutathione cycle involved in salinity tolerance of pepper (Capsicum annuum L.) plants. Plant Physiol Biochem 147:10–20. https://doi.org/10.1016/j.plaphy.2019.11.040
Kościelniak J, Władysław F, Biesaga-Kościelniak J (2006) The effect of drought stress on chlorophyll fluorescence in Lolium-Festuca hybrids. Acta Physiol Plant 28:149–158. https://doi.org/10.1007/s11738-006-0041-y
Li T, Hu Y, Du X, Tang H, Shen C, Wu J (2014) Salicylic acid alleviates the adverse effects of salt stress in Torreya grandis cv. merrillii seedlings by activating photosynthesis and enhancing antioxidant systems. PLoS ONE 9(10):e109492. https://doi.org/10.1371/journal.pone.0109492
Liang X, Zhang L, Natarajan SK, Becker DF (2013) Proline mechanism of stress survival. Antioxid Redox Signal 19(9):998–1011. https://doi.org/10.1089/ars.2012.5074
Liu J, Hasanuzzaman M, Wen H, Zhang J, Peng T, Sun H, Zhao Q (2019) High temperature and drought stress cause abscisic acid and reactive oxygen species accumulation and suppress seed germination growth in rice. Protoplasma 256:1217–1227. https://doi.org/10.1007/s00709-019-01354-6
Liu HT, Liu YY, Pan QH, Yang HR, Zhan JC, Huang WD (2006) Novel interrelationship between salicylic acid, abscisic acid and PIP2-specific-phospholipase C in heat acclimation-induced thermotolerance in pea leaves. J Exp Bot 57:3337–3347. https://doi.org/10.1093/jxb/erl098
Liu J, Lu B, Xu LL (2000) An improved method for the determination of hydrogen peroxide in leaves. Prog Biochem Biophys 27:548–551
Ma X, Zheng J, Zhang X, Hu Q, Qian R (2017) Salicylic acid alleviates the adverse effects of salt stress on Dianthus superbus (Caryophyllaceae) by activating photosynthesis, protecting morphological structure, and enhancing the antioxidant system. Front Plant Sci 8:600. https://doi.org/10.3389/fpls.2017.00600
Menezes-Silva PE, Sanglard LMVP, Ávila RT, Morais LE, Martins SCV, Nobres P, Patreze CM, Ferreira MA, Araújo WL, Fernie AR, DaMatta FM (2017) Photosynthetic and metabolic acclimation to repeated drought events play key roles in drought tolerance in coffee. J Exp Bot 68(15):4309–4322. https://doi.org/10.1093/jxb/erx211
Michel BE, Kaufmann MR (1973) The osmotic potential of polyethylene glycol 6000. Plant Physiol 51:914–916. https://doi.org/10.1104/pp.51.5.914
Mika A, Lüthje S (2003) Properties of guaiacol peroxidase activities isolated from corn root plasma membranes. Plant Physiol 132:1489–1498. https://doi.org/10.1104/pp.103.020396
Miller G, Shulaev V, Mittler R (2008) Reactive oxygen signaling and abiotic stress. Physiol Plant 133:481–489. https://doi.org/10.1111/j.1399-3054.2008.01090.x
Mittler R (2017) ROS are good. Trends Plant Sci 22(1):11–19. https://doi.org/10.1016/j.tplants.2016.08.002
Munns R (2002) Comparative physiology of salt and water stress. Plant Cell Environ 25(2):239–250. https://doi.org/10.1046/j.0016-8025.2001.00808.x
Nahar K, Hasanuzzaman M, Fujita M (2016) Roles of osmolytes in plant adaptation to drought and salinity. In: Iqbal N, Nazar RA, Khan N (eds) Osmolytes and plants acclimation to changing environment: emerging omics technologies. Springer, New Delhi, pp 37–68
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. https://doi.org/10.1093/oxfordjournals.pcp.a076232
Ozkur O, Ozdemir F, Bor M, Turkan I (2009) Physiochemical and antioxidant responses of the perennial xerophyte Capparis ovata Desf. to drought. Environ Exp Bot 66(3):487–492. https://doi.org/10.1016/j.envexpbot.2009.04.003
Potters G, Horemans N, Jansen MSK (2010) The cellular redox state in plant stress biology – a charging concept. Plant Physiol Biochem 48:292–300. https://doi.org/10.1016/j.plaphy.2009.12.007
Rizhsky L, Liang H, Mittler R (2002) The combined effect of drought stress and heat shock on gene expression in tobacco. Plant Physiol 130(3):1143–1151. https://doi.org/10.1104/pp.006858
Sales CR, Ribeiro RV, Silveira JA, Machado EC, Martins MO, Lagôa AM (2013) Superoxide dismutase and ascorbate peroxidase improve the recovery of photosynthesis in sugarcane plants subjected to water deficit and low substrate temperature. Plant Physiol Biochem 73:326–336. https://doi.org/10.1016/j.plaphy.2013.10.012
Santa-Cruz A, Martinez-Rodriguez MM, Perez-Alfocea F, Romero-Aranda R, Bolarin MC (2002) The rootstock effect on the tomato salinity response depends on the shoot genotype. Plant Sci 162:825–831. https://doi.org/10.1016/S0168-9452(02)00030-4
Seckin B, Türkan I, Sekmen AH, Ozfidan C (2010) The role of antioxidant defense system at differential salt tolerance of Hordeum marinum Huds. (sea barleygrass) and Hordeum vulgare L. (cultivated barley). Environ Exp Bot 69:76–85. https://doi.org/10.1016/j.envexpbot.2010.02.013
Sekmen AH, Ozgur R, Uzilday B, Turkan I (2014) Reactive oxygen species scavenging capacities of cotton (Gossypium hirsutum) cultivars under combined drought and heat induced oxidative stress. Environ Exp Bot 99:141–149. https://doi.org/10.1016/j.envexpbot.2013.11.010
Sezen I, Demircan N, SezenKaraoğlan E (2018) Biodiversity and urban gardens: Medicinal and aromatic plants that can used in Erzurum (Turkey) urban gardens. J Architect Sci App 3(1):81–88. https://doi.org/10.30785/mbud.457781
Sezgin M, Tezcan H, Sahin M, Arslan Y, Subasi I, Demir I, Koc A (2017) Determination of yield and quality values of some type Cephalaria syriaca L. cultivated in different ecological conditions in Turkey. KSU J Agric Nat 20:192–195. https://doi.org/10.31015/jaefs.18036
Sharma A, Shahzad B, Kumar V, Kohli SK, Sidhu G, Bali AS, Handa N, Kapoor D, Bhardwaj R, Zheng B (2019) Phytohormones regulate accumulation of osmolytes under abiotic stress. Biomolecules 9(7):285. https://doi.org/10.3390/biom9070285
Smart RE, Bingham GE (1974) Rapid estimates of relative water content. Plant Physiol 53:258–260. https://doi.org/10.1104/pp.53.2.258
Su LJ, Zhang JH, Gomez H, Murugan R, Hong X, Xu D, Jiang F, Peng ZY (2019) Reactive oxygen species-induced lipid peroxidation in apoptosis, autophagy, and ferroptosis. Oxid Med Cell Longev:5080843.https://doi.org/10.1155/2019/5080843
Sun J, Gu J, Zeng J (2013) Changes in leaf morphology, antioxidant activity and photosynthesis capacity in two different drought- tolerant cultivars of chrysanthemum during and after water stress. Sci Hortic 161:249–258. https://doi.org/10.1016/j.scienta.2013.07.015
Torun H, Novák O, Mikulík J, Pěnčík A, Strnad M, Ayaz FA (2020) Timing-dependent effects of salicylic acid treatment on phytohormonal changes, ros regulation, and antioxidant defense in salinized barley (Hordeum vulgare L.). Sci Rep 10:13886. https://doi.org/10.1038/s41598-020-70807-3
Uzilday B, Turkan I, Ozgur R, Sekmen AH (2014) Strategies of ROS regulation and antioxidant defense during transition from C3 to C4 photosynthesis in the genus Flaveria under PEG-induced osmotic stress. J Plant Physiol 171:65–75. https://doi.org/10.1016/j.jplph.2013.06.016
Vukićević DR, Stevanović DD, Genčić MS, Blagojević PD, Radulović NS (2016) Essential-oil constituents and alkanes of Cephalaria ambrosioides Roem. & Schult. (family Caprifoliaceae, subfamily Dipsacaceae) and (chemo)taxonomic discernment of the subfamilies Dipsacaceae and Morinaceae. Chem Biodivers 13(2):198–209. https://doi.org/10.1002/cbdv.201500050
Yildiztugay E, Ozfidan-Konakci C, Kucukoduk M (2013) Sphaerophysa kotschyana, an endemic species from Central Anatolia: antioxidant system responses under salt stress. J Plant Res 126(5):729–742. https://doi.org/10.1007/s10265-013-0573-3
Yildiztugay E, Sekmen AH, Turkan I, Kucukoduk M (2011) Elucidation of physiological and biochemical mechanisms of an endemic halophyte Centaurea tuzgoluensis under salt stress. Plant Physiol Biochem 49(8):816–824. https://doi.org/10.1016/j.plaphy.2011.01.021
Wang X, Liu H, Yu F, Hu B, Jia Y, Sha H, Zhao H (2019) Differential activity of the antioxidant defence system and alterations in the accumulation of osmolyte and reactive oxygen species under drought stress and recovery in rice (Oryza sativa L.) tillering. Sci Rep 9(1):8543. https://doi.org/10.1038/s41598-019-44958-x
Wang J, Meng Y, Li B, Ma X, Lai Y, Si E, Yang K, Xu X, Shang X, Wang H, Wang D (2015) Physiological and proteomic analyses of salt stress response in the halophyte Halogeton glomeratus. Plant Cell Environ 38(4):655–669. https://doi.org/10.1111/pce.12428
Wang W, Vinocur B, Altman A (2003) Plant responses to drought, salinity and extreme temperatures: towards genetic engineering for stress tolerance. Planta 218(1):1–14. https://doi.org/10.1007/s00425-003-1105-5
Acknowledgements
The authors would like to thank Nuriye Peaci for her corrections and suggestions for the English grammar in this manuscript. This work was supported by funding from the Düzce University Research Foundation (2018.11.01.724).
Author information
Authors and Affiliations
Contributions
HT designed the research, analyzed the data and wrote the paper. HA collected the plant materials. HT and HA carried out the experiments.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Torun, H., Aydın, H. Ecophysiological responses of endemic Cephalaria duzceënsis to drought and salt stress. Biologia 77, 2435–2447 (2022). https://doi.org/10.1007/s11756-022-01079-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11756-022-01079-7