Abstract
Objectives
We studied whether histologic subtype according to the new IASLC/ATS/ERS adenocarcinoma classification influences the extent of resection in patients with pathological stage IA lung adenocarcinoma.
Methods
Data on 288 patients with pathological stage IA lung adenocarcinoma were analyzed retrospectively. Recurrence-free survival (RFS) rates were compared according to clinicopathological characteristics, including predominant histologic subtype and extent of resection.
Results
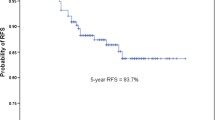
Median follow-up was 38.9 months. Lobectomy was performed in 146 patients, and sublobar resection in 142 patients. When recurrence was compared among the low-grade group (adenocarcinoma in situ, AIS; minimally invasive adenocarcinoma, MIA), intermediate-grade group (lepidic, acinar, and papillary) and high-grade group (solid and micropapillary), the RFS rate decreased as the grade increased (p = 0.037). There was no recurrence in the low-grade or lepidic predominant groups. The recurrence pattern did not differ according to the type of resection or histological subtype. Even in the intermediate- and high-grade groups, the extent of resection was not significantly related to the RFS rate (p = 0.622, p = 0.516). The results were unchanged after adjusting for independent risk factors. The concordance rate between clinical and pathological stage IA was good in low (98.6%) and intermediate grade (84.6%) and poor in high grade (41.2%).
Conclusions
AIS, MIA, and lepidic predominant may be curable by any type of complete resection. Even in invasive subtypes, lobectomy does not offer a recurrence-free advantage over sublobar resection. However, in the high-grade group, less than half of clinical stage IA was actually pathological stage IA. Physicians should exercise caution whenever sublobar resection is planned.



Similar content being viewed by others
References
National Lung Screening Trial Research Team, Aberle DR, Adams AM, Berg CD, Black WC, Clapp JD, Fagerstrom RM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med. 2011;365:395–409.
Ginsberg RJ, Rubinstein LV. Randomized trial of lobectomy versus limited resection for T1 N0 non-small cell lung cancer. Lung Cancer Study Group. Ann Thorac Surg. 1995;60:615–22.
Rusch VW, Hawes D, Decker PA, Martin SE, Abati A, Landreneau RJ, et al. Occult metastases in lymph nodes predict survival in resectable non-small-cell lung cancer: report of the ACOSOG Z0040 trial. J Clin Oncol. 2011;29:4313–9.
Okada M, Nishio W, Sakamoto T, Uchino K, Yuki T, Nakagawa A, et al. Effect of tumor size on prognosis in patients with non-small cell lung cancer: the role of segmentectomy as a type of lesser resection. J Thorac Cardiovasc Surg. 2005;129:87–93.
Koike T, Yamato Y, Yoshiya K, Shimoyama T, Suzuki R. Intentional limited pulmonary resection for peripheral T1 N0 M0 small-sized lung cancer. J Thorac Cardiovasc Surg. 2003;125:924–8.
Yoshikawa K, Tsubota N, Kodama K, Ayabe H, Taki T, Mori T. Prospective study of extended segmentectomy for small lung tumors: the final report. Ann Thorac Surg. 2002;73:1055–8 (discussion 1058–1059).
Nakamura K, Saji H, Nakajima R, Okada M, Asamura H, Shibata T, et al. A phase III randomized trial of lobectomy versus limited resection for small sized peripheral non-small cell lung cancer (JCOG0802/WJOG4607L). Jpn J Clin Oncol. 2010;40:271–4.
Japan Clinical Oncology Group. A non-randomized confirmatory study of limited surgical resection for peripheral early lung cancer defined with thoracic thin-section computed tomography (JCOG0804/WJOG4507L). https://upload.umin.ac.jp/cgi-open-bin/ctr/ctr.cgi?function=brows&action=brows&type=summary&recptno=R000002262&language=J. Accessed 15 Mar 2017.
Japan Clinical Oncology Group. Confirmatory trial of segmentectomy for clinical T1N0 lung cancer dominant with ground glass opacity based on thin-section computed tomography (JCOG1211). https://upload.umin.ac.jp/cgi-open-bin/ctr/ctr.cgi?function=brows&action=brows&type=summary&recptno=R000013286&language=J. Accessed 15 Mar 2017.
National Institutes of Health, National Cancer Institute. CALGB-140503. Phase III randomized study of lobectomy versus sublobar resection in patients with small peripheral stage IA non-small cell lung cancer. http://www.cancer.gov/clinicaltrials/search/view?cdrid=555324&version=HealthProfessional. Accessed 15 Mar 2017.
Travis WD, Brambilla E, Noguchi M, Nicholson AG, Geisinger KR, Yatabe Y, et al. International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society International Multidisciplinary Classification of Lung Adenocarcinoma. J Thorac Oncol. 2011;6:244–85.
Yoshizawa A, Motoi N, Riely GJ, Sima CS, Gerald WL, Kris MG, et al. Impact of proposed IASLC/ATS/ERS classification of lung adenocarcinoma: prognostic subgroups and implications for further revision of staging based on analysis of 514 stage I cases. Mod Pathol. 2011;24:653–64.
Dembitzer FR, Flores RM, Parides MK, Beasley MB. Impact of histologic subtyping on outcome in lobar vs sublobar resections for lung cancer. A pilot study. Chest. 2014;146:175–81.
Mitsudomi T, Morita S, Yatabe Y, Negoro S, Okamoto I, Tsurutani J, et al. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomised phase 3 trial. Lancet Oncol. 2010;11:121–8.
Mok TS, Wu YL, Thongprasert S, Yang CH, Chu DT, Saijo N, et al. Gefitinib or carboplatin–paclitaxel in pulmonary adenocarcinoma. N Engl J Med. 2009;361:947–57.
Goldstraw P, Crowley J, Chansky K, Giroux DJ, Groome PA, Rami-Porta R, et al. The IASLC lung cancer staging project: proposals for the revision of the TNM stage groupings in the forthcoming (seventh) edition of the TNM classification of malignant tumours. J Thorac Oncol. 2007;2:706–14.
Yoshida J, Ishii G, Yokose T, Aokage K, Hishida T, Nishimura M, et al. Possible delayed cut-end recurrence after limited resection for ground-glass opacity adenocarcinoma, intraoperatively diagnosed as Noguchi type B, in three patients. J Thorac Oncol. 2010;5:546–50.
Asamura H, Hishida T, Suzuki K, Koike T, Nakamura K, Kusumoto M, Japan Clinical Oncology Group Lung Cancer Surgical Study Group, et al. Radiographically determined noninvasive adenocarcinoma of the lung: survival outcomes of Japan Clinical Oncology Group 0201. J Thorac Cardiovasc Surg. 2013;146:24–30.
Sawabata N, Miyaoka E, Asamura H, Nakanishi Y, Eguchi K, Mori M, Japanese Joint Committee for Lung Cancer Registration, et al. Japanese lung cancer registry study of 11,663 surgical cases in 2004: demographic and prognosis changes over decade. J Thorac Oncol. 2004;2011(6):1229–35.
Hung JJ, Jeng WJ, Chou TY, Hsu WH, Wu KJ, Huang BS, et al. Prognostic value of the new International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society lung adenocarcinoma classification on death and recurrence in completely resected stage I lung adenocarcinoma. Ann Surg. 2013;258:1079–86.
Hung JJ, Yeh YC, Jeng WJ, Wu KJ, Huang BS, Wu YC, et al. Predictive value of the international association for the study of lung cancer/American Thoracic Society/European Respiratory Society classification of lung adenocarcinoma in tumor recurrence and patient survival. J Clin Oncol. 2014;32:2357–64.
Tsutani Y, Miyata Y, Nakayama H, Okumura S, Adachi S, Yoshimura M, et al. Prediction of pathologic node-negative clinical stage IA lung adenocarcinoma for optimal candidates undergoing sublobar resection. J Thorac Cardiovasc Surg. 2012;144:1365–71.
Tsutani Y, Miyata Y, Nakayama H, Okumura S, Adachi S, Yoshimura M, et al. Prognostic significance of using solid versus whole tumor size on high resolution computed tomography for predicting the pathological malignant grade of tumors in clinical stage IA lung adenocarcinoma: a multicenter study. J Thorac Cardiovasc Surg. 2012;143:607–12.
Nitadori J, Bograd AJ, Kadota K, Sima CS, Rizk NP, Morales EA, et al. Impact of micropapillary histologic subtype in selecting limited resection vs lobectomy for lung adenocarcinoma of 2 cm or smaller. J Natl Cancer Inst. 2013;105:1212–20.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Rights and permissions
About this article
Cite this article
Ito, H., Nakayama, H., Murakami, S. et al. Does the histologic predominance of pathological stage IA lung adenocarcinoma influence the extent of resection?. Gen Thorac Cardiovasc Surg 65, 512–518 (2017). https://doi.org/10.1007/s11748-017-0790-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11748-017-0790-0