Abstract
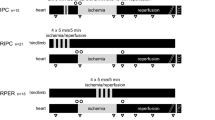
Objective: Ischemic preconditioning (IPC) has been found to protect the myocardium in animal studies. However, clinical studies have been limited and the clinical effects of IPC are still uncertain. The purpose of this study was to assess whether IPC has any protective effect on the human myocardium during minimally invasive CABG (MIDCAB), by means of epicardial electrophysiological testing. Method: Fortyfive patients with left anterior descending artery disease who underwent a MIDCAB procedure were evaluated. In the present study, the electrical potentials which were not affected by cardio-pulmonary bypass or cardioplegia were measured. The ratio of longitudinal to transverse conduction velocity (ΘL/ΘT), and QT, JT dispersions were measured using plaque electrodes in the preischemic state, during a 5-minute coronary occlusion, during the subsequent 5-minute reperfusion, during 5- and 10-minute anastomosis periods, and after anastomosis. Result: The ΘL/ΘT was 2.2±0.2 at baseline. Anisotropy was exaggerated during the 5-minute coronary occlusion (2.6±0.3). During anastomosis, conduction velocities were decreased, but showed no further deterioration (2.4±0.3, and 2.4±0.3, respectively). QT and JT dispersions were improved by reperfusion. Conclusion: The effectiveness of IPC during the MIDCAB procedure was confirmed electrophysiologically. Anisotropy and dispersions were minimized after IPC, therefore IPC demonstrated antiarrhythmic protective effects on the human myocardium.
Similar content being viewed by others
References
Murry CE, Jennings RB, Reimer KA. Preconditioning with ischemia: A delay of lethal cell injury in ischemic myocardium. Circulation 1986; 74: 1124–36.
Malkowski MJ, Kramer CM, Parvizi ST, Dianzumba S, Marquez J, Reichek N, et al. Transient ischemia does not limit subsequent ischemic regional dysfunction in humans: A transesophageal echocardiographic study during minimally invasive coronary artery bypass surgery. J Am Coll Cardiol 1998; 31: 1035–9.
Tzivoni D, Maybaum S. Attenuation of severity of myocardial ischemia during repeated daily ischemic episodes. J Am Coll Cardiol 1997; 30: 119–24.
Lim R, Laskey WK. Ischemic preconditioning in unstable coronary syndromes: evidence for time dependence. J Am Coll Cardiol 1997; 30: 1461–5.
Henein MY, O'Sullivan C, Davies SW, Sigwart U, Gibson DG. Effects of acute coronary occlusion and previous ischaemic injury on left ventricular wall motion in humans. Heart 1997; 77: 338–45.
Eltchaninoff H, Cribier A, Tron C, Derumeaux G, Koning R, Hecketsweiller B, et al. Adaptation to myocardial ischemia during coronary angioplasty demonstrated by clinical, electrocardiographic, echocardiographic, and metabolic parameters. Am Heart J 1997; 133: 490–6.
Dupouy P, Geschwind H, Pelle G, Aptecar E, Hittinger L, El Ghalid A, et al. Repeated coronary artery occlusions during routine balloon angioplasty do not induce myocardial preconditioning in humans. J Am Coll Cardiol 1996; 27: 1374–80.
Kloner RA, Yellon D. Does ischemic preconditioning occur in patients? J Am Coll Cardiol 1994; 24: 1133–42.
Cinca J, Warren M, Carreno A, Tresanchez M, Armadans L, Gomez P, et al. Change in myocardial electrical impedance induced by coronary artery occlusion in pigs with and without preconditioning: correlation with local ST-segment potential and ventricular arrhythmias. Circulation 1997; 96: 3079–86.
Pasceri V, Lanza GA, Patti G, Pedrotti P, Crea F, Maseri A. Preconditioning by transient myocardial ischemia confers protection against ischemia-induced ventricular arrhythmias in variant angina. Circulation 1996; 94: 1850–6.
Zareba W, Moss AJ, le Cessie S. Dispersion of ventricular repolarization and arrhythmic cardiac death in coronary artery disease. Am J Cardiol 1994; 74: 550–3.
Surawicz B. The QT interval and cardiac arrhythmias. Annu Rev Med 1987; 38: 81–90.
Shiki K, Hearse DJ. Preconditioning of ischemic myocardium: Reperfusion-induced arrhythmias. Am J Physiol 1987; 253: H1470–6.
Mizumaki K, Fujiki A, Tani M, Misaki T. Effects of acute ischemia on anisotropic conduction in canine ventricular muscle. PACE 1993; 16: 1656–63.
Cardinal R, Vermeulen M, Shenasa M, Roberge F, Page P, Helie F, et al. Anisotropic conduction and functional dissociation of ischemic tissue during reentrant ventricular tachycardia in canine myocardial infarction. Circulation 1988; 77: 1162–76.
Spach MS, Dolber PC, Heidlage JF. Influence of the passive anisotropic properties on directional differences in propagation following modification of the sodium conductance in human atrial muscle: A model of reentry based on anisotropic discontinuous propagation. Circ Res 1988; 62: 811–32.
Kleber AG, Janse MJ, Wilms-Schopmann FJ, Wilde AA, Coronel R. Changes in conduction velocity during acute ischemia in ventricular myocardium of the isolated porcine heart. Circulation 1986; 73: 189–98.
Spach MS, Miller WT 3rd, Geselowitz DB, Barr RC, Kootsey JM, Johnson EA. The discontinuous nature of propagation in normal canine cardiac muscle: Evidence for recurrent discontinuities of intracellular resistance that affect the membrane currents. Circ Res 1981; 48: 39–54.
Gottwald E, Gottwald M, Dhein S. Enhanced dispersion of epicardial activation-recovery intervals at sites of histlogical inhomogeneity during regional cardiac ischaemia and reperfusion. Heart 1998; 79: 474–80.
Stierle U, Giannitsis E, Sheikhzadeh A, Kruger D, Schmucker G, Mitusch R, et al. Relation between QT dispersion and the extent of myocardial ischemia in patients with three-vessel coronary artery disease. Am J Cardiol 1998; 81: 564–8.
Kautzner J, Malik M. QT interval dispersion and its clinical utility. PACE 1997; 20: 2625–40.
Trusz-Gluza M, Wozniak-Skowerska I, Giec L, Szydlo H. Dispersion of the QT inteval as a predictor of cardiac death in patients with coronary heart disease. PACE 1996; 19: 1900–4.
Dillon SM, Allessie MA, Ursell PC, Wit AL. Influences of anisotropic tissue structure on reentrant circuits in the epicardial border zone of subacute canine infarcts. Circ Res 1988; 63: 182–206.
Losano G, Gattullo D, Pagliaro P. Myocardial, neural and vascular aspects of ischemic preconditioning. Life Sci 1996; 59: 1185–92.
Starkopf J, Bugge E, Ytrehus K. Preischemic bradykinin and ischaemic preconditioning in functional recovery of the globally ischaemic rat heart. Cardiovasc Res 1997; 33: 63–70.
Baxter GF. Ischaemic preconditioning of myocardium. Ann Med 1997; 29: 345–52.
Kotoh K, Watanebe G, Ueyama K, Uozaki M, Suzuki M, Misaki T, et al. On-line assessment of regional ventricular wall motion by transesophageal echocardiography with color kinesis during minimally invasive coronary artery bypass grafting. J Thorac Cardiovasc Surg 1999; 117: 912–7.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Doi, Y., Watanabe, G., Kotoh, K. et al. Myocardial ischemic preconditioning during minimally invasive direct coronary artery bypass grafting attenuates ischemia-induced electrophysiological changes in human ventricle. Jpn J Thorac Caridovasc Surg 51, 144–150 (2003). https://doi.org/10.1007/s11748-003-0050-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11748-003-0050-3