Abstract
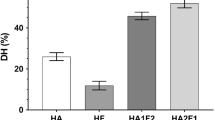
Protein isolate from sunflower seeds was used as the starting material for preparation of an extensively hydrolyzed peptide product. Protein was hydrolyzed using an endopeptidase (Alcalase), an exopeptidase (Flavourzyme), or both enzymes sequentially. Combined use of these proteases generated the highest degree of hydrolysis, 54.2%, and highest solubility, around 90%, between pH 2.5 and 7. Molecular weight profiles of the hydrolysates were characterized by gel filtration chromatography and denaturing electrophoresis. Amino acid compositions and solubilities of the different hydrolysates also were studied.
Similar content being viewed by others
References
Matthews, D.M., F.R.C. Path, and S.A. Adibi, Progress in Gastroenterology, Peptide Absorption, Gastroenterology 71:151–161 (1976).
Hessov, I., and P. Wara, Improved Nutritional Intake After Colonic Surgery, Acta Chir. Scan. 516:26–28 (1983).
Adler-Nissen, J., A Review of Food Protein Hydrolysis—Specific, in Enzymic Hydrolysis of Food Proteins, edited by J. Adler-Nissen, Elsevier Science Publishers Ltd., Essex, England, 1986, pp. 263–313.
Lühs, W., and W. Friedt, The Major Oil Crops, in Designer Oil Crops, Breeding, Processing and Biotechnology, edited by D.J. Murphy, VCH, Weinheim, Germany, 1994, pp. 5–58.
Parrado, J., J. Bautista, and A. Machado, Production of Soluble Enzymatic Protein Hydrolysate from Industrially Defatted Nondehulled Sunflower Meal, J. Agric. Food Chem. 39:447–450 (1991).
Lusas, E.W., Sunflower Seed Protein, in New Protein Foods, edited by A.M. Altschul and H.L. Wilcke, Academic Press, London, 1985, Vol. 5, pp. 393–429.
Villanueva, A., J. Vioque, R. Sánchez-Vioque, A. Clemente, J. Bautista, and F. Millán, Production of an Extensive Sunflower Protein Hydrolysate by Sequential Hydrolysis with Endo- and Exopeptidases, Grasas Aceites 50: in press (1999).
Parrado, J., F. Millán, I. Hernández-Pinzón, J. Bautista, and A. Machado, Characterization of Enzymatic Sunflower Protein Hydrolysates. J. Agric. Food Chem. 41:1821–1825 (1993).
AOAC Official Methods of Analysis, 15th edn., Association of Official Analytical Chemists, Arlington, 1990, pp. 1–11, Method Aa 5–38.
Adler-Nissen, J., Determination of the Degree of Hydrolysis of Food Protein Hydrolyzates by Trinitrobenzenesulfonic acid. J. Agric. Food Chem. 27:1256–1262 (1979).
Laemmli, U.K., Cleavage of Structural Proteins During Assembly of the Head of Bacteriophage T4, Nature 227:680–685 (1970).
Alaiz, M., J.L. Navarro, J. Girón, and E. Vioque. Amino Acid Analysis by High-Performance Liquid Chromatography After Derivatization with Diethyl Ethoxymethylenemalonate. J. Chromatogr. 591:181–186 (1992).
FAO/WHO, Protein Quality Evauation. Food and Agricultural Organization of the United Nations, Rome, Italy, 1991, p. 66.
Plumb, G.W., and N.A. Lambert, Comparison of the Trypsinolysis of Nine 11S Globulin Species, Food Hydrocolloids 3:465–473 (1990).
Larré, C., M. Chiarello, S. Dudek, M. Chenu, and J. Guéguen, Action of Transglutaminase on the Constitutive Polypeptides of Pea Legumin, J. Agric. Food Chem. 41:1816–1820 (1993).
Zayas, J.F., Solubility of Proteins, in Functionality of Proteins in Foods. Springer-Verlag, Berlin, 1997, pp. 6–67.
Mahmoud, M.I., Physicochemical and Functional Properties of Protein Hydrolysates in Nutritional Products, Food Technol. 48:89–95 (1994).
Chobert, J.M., C. Bertrand-Harb, and M.G. Nicolas, Solubility and Emulsifying Properties of Caseins and Whey Proteins Modified Enzymatically by Trypsin. J. Agric. Food Chem. 36:883–892 (1988).
Chobert, J.M., L. Briand, J. Guéguen, Y. Popineau, C. Larré, and T. Haertlé, Recent Advances in Enzymatic Modifications of Food Proteins for Improving Their Functional Properties, Nahrung 40:177–182 (1996).
Schmidl, M.K., S.L. Taylor, and J.A. Nordlee, Use of Hydrolysate-Based Products in Special Medical Diets, Food Technol. 48:77–85 (1994).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Villanueva, A., Vioque, J., Sánchez-Vioque, R. et al. Peptide characteristics of sunflower protein hydrolysates. J Amer Oil Chem Soc 76, 1455–1460 (1999). https://doi.org/10.1007/s11746-999-0184-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11746-999-0184-2