Abstract
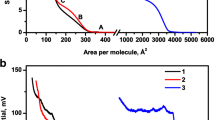
Methoxy ethyl oleate, a nonionic surfactant, has been investigated at the air/water interface for various surface properties by employing the Langmuir film balance technique. The ester forms an expanded isotherm at the air/water interface. The minimum area of packing (A 0), initial area of increase of surface pressure (A i ), collapse pressure (π c ), and area/molecule at collapse pressure (A c ) have been estimated from the isotherm curve.The higher compressibility coefficient (K) suggests that the ester forms a more expanded liquid film than the parent oleic acid. Besides, the ester film is fairly stable as suggested by only about 30% loss in area over a period of 20 min. The relaxation rates of the ester film at different surface pressures of 10, 15, and 20 mN·m−1 have been estimated from changes in the surface area/molecule with time. Interestingly, the surface area (54.2 Å2/molecule) that corresponds to a minimized structure projected for the ester, calculated theoretically, agrees reasonably well with the experimental value (57.2 Å2/molecule).
Similar content being viewed by others
References
Fendler, J.H., Microemulsions, Micelles and Vesicles as Media for Membrane Mimetic Photochemistry, J. Phys. Chem. 84:1485–1491 (1980).
Fendler, J.A., Membrane Mimetic Chemistry, Wiley Interscience, New York, 1982.
Tien, T.H., Bilayer Lipid Membrane (BLM) Theory and Practice, Marcel Dekker, New York, 1974.
Atwood, D., and A.T. Florence, Surfactant Systems, Their Chemistry, Pharmacy, and Biology, Chapman Hill and Hall, New York, Chapter 6, 1983, p. 293.
Shimomura, H., and T. Kunitake, Interaction of Ions with Surface Receptor of the Azobenzene Containing Bilayer Membranes, J. Am. Chem. Soc. 104:1757–1759 (1982).
Adamson, A.W., Physical Chemistry of Surfaces, Wiley Interscience Publications, New York, 1990.
Kenn, R.M., C. Bohm, A.M. Bibo, I.R. Peterson, H. Mohwald, J.A. Nielsen, and K. Kjaer, Mesophases and Crystalline Phases in Fatty Acid Monolayers, J. Phys. Chem. 95:2092–2097 (1991).
Cantor, R.S., and P.M. Mellory, Statistical Thermodynamics of Flexible Chain Surfactants in Monolayer Films 1. Theory of Fluid Phases, J. Chem. Phys. 90:4423–4430 (1989).
Linden, M., and J. Rosenholm, Influence of Multivalent Metal Ions on the Monolayer and Multilayer Properties of Some Unsaturated Fatty Acids, Langmuir 11:4499–4504 (1995).
Peltonen, J.P.K., P. He, and J.B. Rosenholm, The Polymerisation of Monolayers of Some Unsaturated Fatty Acids, Thin Solid Films 210/211:372–374 (1992).
Peltonen, J.P.K., P. He, and J.B. Rosenholm, Phase Controlled Polymerization of Linoleic Acid Monolayers, J. Phys. Chem. 98:12403–12409 (1991).
Bailey, A.E., Industrial Oils and Fats, Intersciences, New York, 1951.
Thomas, J.K., Radiation Induced Reactions in Organised Assemblies, Chem. Rev. 80:283–297 (1980).
Paleos, C.M., Polymerisation in Organised Systems, Chem. Soc. Rev. 14:45–67 (1985).
Geetha, B., A.B. Mandal, and T. Ramasami, Synthesis, Characterisation and Micelle Formation in an Aqueous Solution of Methoxy Poly(ethylene glycol) Macromonomer, Homopolymer and Graft Copolymer, Macromolecules 26:4083–4088 (1993).
Gaines, G.L., Jr., Insoluble Monolayers at Liquid Gas Interfaces, Wiley Interscience, New York, 1966.
MacArthur, B.W., and J.C. Berg, Surface Equations of State for Insoluble Monolayers on Aqueous Solutions, J. Colloid Interface Sci. 68:201–213 (1979).
Official Methods of Analysis, Section 4. Oils, Fats, and Related Substances, Soc. Leather Trade Chemists, 1965.
Tomoaia-Cotisel, M., J. Zsako, A. Mocanu, M. Lupea, and E. Chiju, Insoluble Mixed Monolayers. The Ionisation Characteristics of Some Fatty Acids at the Air-Water Interface, J. Colloid Interface Sci. 117:464–476 (1987).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Baskar, G., Venkatesan, S., Dhathathreyan, A. et al. Two-dimensional surface properties of 2-methoxy ethyl oleate at the air/water interface. J Amer Oil Chem Soc 76, 853–858 (1999). https://doi.org/10.1007/s11746-999-0076-5
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11746-999-0076-5