Abstract
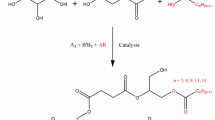
This paper reports the incorporation of an alpha-methylene unit into fatty acid skeletons. Since the new olefin is conjugated with the carboxylate, it is susceptible to 1,4-(Michael) additions. We have used multifunctional thiols and amines for additions at the methylene. The resulting products possess clusters of functionality grouped at one end of a hydrophobic tail. These structural patterns will be of use in the design of new types of bio-based surfactants and polymers. One particularly promising pattern of functionality that can be obtained through oxidation and reduction chemistry is a 2-fatty 1,2,3-propanetriol, or a lipophilized glycerol moiety.




Similar content being viewed by others
References
Huf S, Krügener S, Hirth T, Rupp S, Zibek S (2011) Biotechnological synthesis of long-chain dicarboxylic acids as building blocks for polymers. Eur J Lipid Sci Technol 113:548–561
Hwang H-S, Erhan SZ (2001) Highly selective asymmetric synthesis of 2-hydroxy fatty acid methyl esters through chiral oxazolidinone carboximides. JAOCS 78:205–211
Terés S, Lladó V, Higuera M, Barceló-Coblijn G, Martin ML, Noguera-Salvà MA, Marcilla-Etxenike A, García-Verdugo JM, Soriano-Navarro M, Saus C, Gómez-Pinedo U, Busquets X, Escribá PV (2012) 2-Hydroxyoleate, a nontoxic membrane binding anticancer drug, induces glioma cell differentiation and autophagy. Proc Natl Acad Sci 109:8489–8494
Cordomí A, Prades J, Frau J, Vögler O, Funari SS, Perez JJ, Escribá PV, Barceló F (2010) Interactions of fatty acids with phosphatidylethanolamine membranes: X-ray diffraction and molecular dynamics studies. J Lipid Res 51:1113–1124
Magrioti V, Verger R, Constantinou-Kokotou V (2004) Triacylglycerols based on 2-(N-tert-butoxycarbonylamino)oleic acid are potent inhibitors of pancreatic lipase. J Med Chem 47:288–291
Torres CF, Martín D, Torrelo G, Casado V, Fernández O, Tenllado D, Vázquez L, Morán-Valero MI, Reglero G (2011) Lipids as delivery systems to improve the biological activity of bioactive ingredients. Curr Nutr Food Sci 7:160–169
Hin B, Majer P, Tsukamoto T (2002) Facile synthesis of α-substituted acrylate esters. J Org Chem 67:7365–7368
Best MD, Rowland MM, Bostic HE (2011) Exploiting bioorthogonal chemistry to elucidate protein-lipid binding interactions and other biological roles of phospholipids. Acc Chem Res 44:686–698
Knight HB, Koos RE, Swern D (1953) Addition of formic acid to olefinic compounds. I. Monoolefinic compounds. J Am Chem Soc 75:6212–6215
Zerkowski JA, Solaiman DKY (2006) Synthesis of polyfunctional fatty amines from sophorolipid-derived 17-hydroxy oleic acid. JAOCS 83:621–628
Sakakura A, Kawajiri K, Ohkubo T, Kosugi Y, Ishihara K (2007) Widely useful DMAP-catalyzed esterification under auxiliary base- and solvent-free conditions. J Am Chem Soc 129:14775–14779
Reding MT, Buchwald SL (1995) An inexpensive air-stable titanium-based system for the conversion of esters to primary alcohols. J Org Chem 60:7884–7890
Sharpless KB, Amberg W, Bennani YL, Crispino GA, Hartung J, Jeong KS, Kwong HL, Morikawa K, Wang ZM (1992) The osmium-catalyzed asymmetric dihydroxylation: a new ligand class and a process improvement. J Org Chem 57:2768–2771
Gabriels S, Van Haver D, Vandewalle M, De Clercq P, Viterbo D (1999) On the unexpected stereochemical outcome of the magnesium in methanol-conjugate reduction of an exocyclic α, β-unsaturated ester. Eur J Org Chem 1999:1803–1809
Okuda T, Ishimoto K, Ohara H, Kobayashi S (2012) Renewable biobased polymeric materials: facile synthesis of itaconic anhydride-based copolymers with poly (l-lactic acid) grafts. Macromolecules 45:4166–4174
Hoyle CE, Bowman CN (2010) Thiol–ene click chemistry. Angew Chem Int Ed 49:1540–1573
O’Lenick AJ Jr (2001) Guerbet chemistry. J Surfactants Deterg 4:311–315
Camps F, Coll J, Guitart J (1986) Regiospecific reduction of unsaturated conjugated ketones with sodium dithionite under phase transfer catalysis. Tetrahedron 42:4603–4609
Tai A, Kawasaki D, Sasaki K, Gohda E, Yamamoto I (2003) Synthesis and characterization of 6-O-acyl-2-O-α-d-glucopyranosyl-l-ascorbic acids with a branched-acyl chain. Chem Pharm Bull 51:175–180
Author information
Authors and Affiliations
Corresponding author
Additional information
Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the US Department of Agriculture (USDA). USDA is an equal opportunity provider and employer.
About this article
Cite this article
Zerkowski, J.A., Solaiman, D.K.Y. 2-Fatty Acrylic Acids: New Highly Derivatizable Lipophilic Platform Molecules. J Am Oil Chem Soc 91, 1225–1233 (2014). https://doi.org/10.1007/s11746-014-2459-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-014-2459-5