Abstract
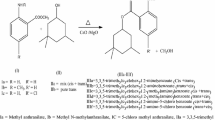
Methoxylation of methyl oleate into methyl methoxy stearate was carried out in a batch reactor at temperatures ranging from 150 to 190°C in the presence of dealuminated H-Y faujasites as catalysts. In the presence of an excess of methanol, the H-Y faujasite with a Si/Al ratio of 15 was shown to achieve the title reaction with a yield of methyl methoxy stearate that does not exceed 40% because of the parallel formation, at comparable rates, of methyl oleate isomers identified as methyl elaidate, methyl trans-vaccenate, and methyl cis-vaccenate. Isomerization reactions were confirmed to occur rapidly in an independent manner. FFA are also present in small amounts owing to the in situ dehydration of methanol and subsequent hydrolysis of the esters. Finally, starting from pure oleic acid leads to results similar to those obtained with methyl oleate as the starting material.
Similar content being viewed by others
References
Wagner, H., R. Luther, and T. Mang, Lubricant Base Fluids Based on Renewable Raw Materials: Their Catalytic Manufacture and Modification, Appl. Catal. A: Gen. 221:429–442 (2001).
Geller, D.P., and J.W. Goodrum, Effects of Specific Fatty Acid Methyl Esters on Diesel Fuel Lubricity, Fuel 83:2351–2356 (2004).
Gunstone, F.D., and R.P. Inglis, Application of the Oxymercuration-Demercuration Reaction to Long-Chain Unsaturated Esters, Chem. Phys. Lipids 10:73–88 (1973).
Gunstone, F.D., and R.P. Inglis, Fatty Acids. Neighboring Group Participation in the Oxymercuration-Demercuration Reaction. Another Route to 1–4 and 1–5 Epoxides, Chem. Phys. Lipids 10:89–104 (1973).
Carballeira, N.M., M.V. Gonzales, and M. Pagan, Neighbouring Methoxyl Participation in the Acid-Catalyzed Methoxylation of Methylene-Interrupted Fatty Acids, Chem Phys. Lipids 89:91–96 (1997).
Madrigal, R.V., M.O. Bagby, and E.H. Pryde, The Acid-Catalyzed Addition of Alkoxy Groups to the Olefinic Double Bonds of Soybean Oil, J. Am. Oil Chem. Soc. 65:1508–1510 (1988).
Sheldon, R.A., and H. van Bekkum, Introduction, in Fine Chemicals Through Heterogeneous Catalysis, edited by R.A. Sheldon and H. van Bekkum, Wiley-VCH, Weinheim, 2001, pp. 1–11.
Sheldon, R.A., and H.U. Blaser, Catalysis Applied to Fine Chemicals, Adv. Synth. Catal. 345:413 (2003).
Corma, A., State of the Art and Future Challenges of Zeolites as Catalysts, J. Catal. 216:298–312 (2003).
Zhang, S., and Z. Zhang, Fatty Acid Isomerization with Mesoporous Zeolites, Patent WO 03/082464 (2003).
Tomifuji, T., H. Abe, Y. Matsumura, and Y. Sakuma, Process for the Preparation of Branched Chain Fatty Acids and Alkyl Esters Thereof, U.S. Patent 5,677,473 (1997).
Hodgson, W.R., C.M. Lok, G. Roberts, and W.T. Koestsier, Fatty Acid Isomerisation, Patent EP 0,774,451 (1997).
Kohashi, H., and T.A. Foglia, Addition of Aromatic Compounds to Oleic Acid Catalyzed by Heterogeneous Acid Catalysis, J. Am. Oil Chem. Soc. 61:1048–1051 (1984).
Foglia, T.A., and T. perlstein, Process for the Preparation of Branched Chain Fatty Acids and Esters, U.S. Patent 4,371,469 (1983).
Augustine, R.L., Heterogeneous Catalysis for the Synthetic Chemist, Marcel Dekker, New York, 1996.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Lansalot-Matras, C., Lozano, P., Pioch, D. et al. Methoxylation of methyl oleate in the presence of dealuminated Y faujasites in their protonic form. J Amer Oil Chem Soc 83, 725–729 (2006). https://doi.org/10.1007/s11746-006-5030-1
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11746-006-5030-1