Abstract
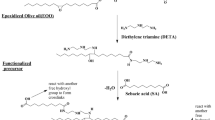
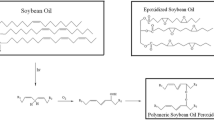
To diversify edible-oil polymer composite, polymeric linoleic acid (PLina) peroxide was obtained by the auto-oxidation of linoleic acid in a simple way for use as a macroinitiator in free radical polymerization of vinyl monomers. Peroxidation, epoxidation, and/or perepoxidation reactions of linoleic acid under air at room temperature resulted in PLina, having soluble fraction more than 91 weight percent (wt%), with molecular weight ranging from 1,644 to 2,763 Da, and containing up to 1.0 wt% of peroxide. PLina initiated the free radical polymerization of ether styrene (S), methyl methacrylate (MMA), or n-butyl methacrylate (nBMA) to give PLina-g-polystyrene (PS), PLina-g-poly-MMA (PMMA), and PLina-g-poly- nBMA (PnBMA) graft copolymers. The polymers obtained were characterized by proton nuclear magnetic resonance (1H NMR), thermal gravimetric analysis (TGA), differential scanning calorimetry (DSC), and gel permeation chromatography (GPC) techniques. Microstructure of the graft copolymers was observed by using scanning electron microscope (SEM). Graft copolymers obtained contained polymeric linoleic acid in a range between 8.5 and 19.3 mol percent (mol%). PLina-g-PS, PLina-g-PMMA and PLina-g-PnBMA graft copolymer samples were also used in cell culture studies. Fibroblast and macrophage cells were strongly adhered and spread on the copolymer film surfaces. These newly synthesized copolymers were tested for their effects on human blood protein adsorption compared with PMMA graft copolymers containing polymeric soybean oil and polymeric linseed oil; interestingly we observed a dramatic decrease in the protein adsorption on the linoleic acid graft copolymer, which is important in tissue engineering.




Similar content being viewed by others
References
Ashby RD, Foglia TA (1998) Poly(hydroxyalkanoate) biosynthesis from triglyceride substrates. Appl Microbiol Biotechnol 49:431–437
Hazer B, Torul O, Borcakli M, Lenz RW, Fuller RC, Goodwin SD (1998) Bacterial production of polyesters from free fatty acids obtained from natural oils by Pseudomonas oleovorans. J Environ Polym Degrad 6:109–113
Li F, Hanson MV, Larock RC (2001) Thermophysical and mechanical properties of novel polymers prepared by the cationic copolymerization of fish oils, styrene and divinylbenzene. Polymer 2:1567–1579
Guner FS, Usta S, Erciyes AT, Yagci Y (2000) Styrenation of triglyceride oils by macromonomer technique. J Coat Technol 72:107–110
Domb AJ, Nudelman R (1995) Biodegradable polymers derived from natural fatty acids. J Polym Sci A: Polym Chem 33:717–725
Thames SH, Yu H, Subramanian R (2000) Cationic ultraviolet curable coatings from castor oil. J Appl Polym Sci 77:8–13
Chen J, Soucek MD, Simonsick WJ, Celikay RW (2002) Synthesis and photopolymerization of norbornyl epoxidized linseed oil. Polymer 43:5379–5389
Cakmakli B, Hazer B, Tekin IO, Kizgut S, Koksal M, Menceloglu Y (2004) Synthesis and characterization of polymeric linseed oil grafted methyl methacrylate or styrene. Macromol Biosci 4:649–655
Cakmakli B, Hazer B, Tekin IÖ, Cömert FB (2005) Synthesis and characterization of polymeric soybean oil-g-methyl methacrylate (and n-butyl methacrylate) graft copolymers: biocompatibility and bacterial adhesion. Biomacromolecules 6:1750–1758
Satriano C, Carnazza S, Guglielmino S, Marletta G (2003) Surface free energy and cell attachment onto ion-beam irradiated polymer surfaces. Nucl Instrum Methods Phys Res B 208:287–293
Johnson WC, Wang J, Chen Z (2005) Surface structures and properties of polystyrene/poly(methyl methacrylate) blends and copolymers. J Phys Chem B 109:6280–6286
Hazer B, Baysal B (1986) Preparation of block copolymers using a new polymeric peroxycarbamate. Polymer 27:961–968
Hazer B, Erdem B, Lenz RW (1994) Styrene polymerization with some new macro or macromonomeric azoinitiators having PEG units. J Polym Sci A: Polym Chem 32:1739–1746
Chung CW, Kim HW, Kim YB, Rhee YH (2003) Poly(ethylene glycol)-grafted poly(3-hydroxyundecenoate) networks for enhanced blood compatibility. Int J Biol Macromol 32:17–22
Yin H, Porter NA (2003) Specificity of the ferrous oxidation of xylenol orange assay: analysis of autoxidation products of cholesteryl arachidonate. Anal Biochem 313:319–326
Ebara M, Yamato M, Hirose M, Aoyagi T, Kikuchi A, Sakai K, Okano T (2003) Copolymerization of 2-carboxyisopropylacrylamide with N-isopropylacrylamide accelerates cell detachment from grafted surfaces by reducing temperature. Biomacromolecules 4(2):344–349
Thayer A (2005) Removing impurities. Chemical and Engineering News C&EN Houston. 83(36):55–58
Poliakoff M, Anastas P (2001) Green chemistry: a principled stance. Nature 413:257–257
Hazer B (1987) Polymerization of vinyl monomers by a new oligoperoxide: oligo(adipoyl-5-peroxy-2,5-dimethyl n-hexyl peroxide). J Polym Sci A: Polym Chem Ed 25:3349–3354
Murty KS, Kishore K (1996) A novel (main chain)-(side chain) polymeric peroxide: synthesis and initiating characteristics of poly(α-(tert-butylperoxymethyl)styrene peroxide). Macromolecules 29:4859–4864
Hazer B, Ayas A, Beşirli N, Saltek N, Baysal BM (1989) Preparation of ABCBA-type block copolymers by use of macro-initiators containing peroxy and azo groups. Makromol Chem 190:1987–1996
Acknowledgment
This work was financially supported by Zonguldak Karaelmas University Research Fund and partially supported by TÜBİTAK research project (Grant no. 104M128).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Çakmaklı, B., Hazer, B., Tekin, İ.Ö. et al. Polymeric Linoleic Acid–Polyolefin Conjugates: Cell Adhesion and Biocompatibility. J Amer Oil Chem Soc 84, 73–81 (2007). https://doi.org/10.1007/s11746-006-1011-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11746-006-1011-7