Abstract
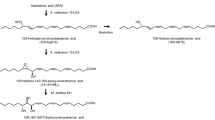
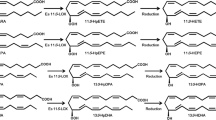
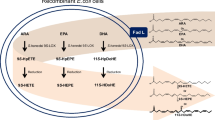
Clavibacter sp. ALA2 oxidized n−3 and n−6 PUFA into a variety of oxylipins. Structures of products converted from EPA and DHA were determined as 15,18-dihydroxy-14,17-epoxy-5(Z),8(Z),11(Z)-eicosatrienoic acid and 17,20-dihydroxy-16,19-epoxy-4(Z),7(Z),10(Z),13(Z)-docosatetraenoic acid by GC-MS and NMR analyses. In contrast, γ-linolenic acid and arachidonic acid were converted to diepoxy bicyclic FA, tetrahydrofuranyl monohydroxy FA, and trihydroxy FA. Thus, the structures of bioconversion products were different between n−3 and n−6 PUFA. Furthermore, strain ALA2 placed hydroxy groups and cyclic structures at the same position from the ω-terminal despite the number of carbons in the chain and the double bonds in the PUFA.
Similar content being viewed by others
References
Gardner, H.W., Recent Investigations into the Lipoxygenase Pathway of Plants, Biochim. Biophys. Acta 1084:221–239 (1991).
Hou, C.T., Value-Added Products Through Bioprocessing: New Hydroxyl Fatty Acids, inform 13:307–316 (2002).
Needleman, P.J., J. Turk, B.A. Jakschik, A.R. Morrison, and J.B. Lefkowith, Arachidonic Acid Metabolism, Annu. Rev. Biochem. 55:69–102 (1986).
Hou, C.T., Biotransformation of Unsaturated Fatty Acids to Industrial Products, Adv. Appl. Microbiol. 47:201–220 (2000).
Wallen, L.L., R.G. Benedict, and R.W. Jackson, The Microbial Production of 10-Hydroxystearic Acid, Arch. Biochem. Biophys. 99:249–253 (1962).
De Andres, C., E. Mercade, G. Guinea, and A. Manresa, 7,10-Dihydroxy-8-(E)-octadecenoic Acid Produced by Pseudomonas 42A2: Evaluation of Different Cultural Parameters of the Fermentation, World J. Microbiol. Biotechnol. 10:106–109 (1994).
El-Sharkawy, S.H., W. Yang, L. Dostal, and J.P.N. Rosazza, Microbial Oxidation of Oleic Acid, Appl. Environ. Microbiol. 58:2116–2122 (1992).
Hou, C.T., Microbial Oxidation of Unsaturated Fatty Acids, Adv. Appl. Microbiol. 41:1–23 (1995).
Hou, C.T., A Novel Compound, 12,13,17-Trihydroxy-9(Z)-octadecenoic acid, from Linoleic Acid by a New Microbial Isolate Clavibacter sp. ALA2, J. Am. Oil Chem. Soc. 73:1359–1362.
Hou, C.T., H.W. Gardner, and W. Brown, 12,13,16-Trihydroxy-9(Z)-octadecenoic Acid, a Possible Intermediate in the Bioconversion of Linoleic Acid to Tetrahydrofuranyl Fatty Acids by Clavibacter sp. ALA2, Ibid.J. Am. Oil Chem. Soc. 78:1167–1169.
Hou, C.T., H. Gardner, and W. Brown, Production of Polyhydroxy Fatty Acids from Linoleic Acid by Clavibacter sp. ALA2, Ibid.J. Am. Oil Chem. Soc. 75:1483–1487.
Gardner, H.W., C.T. Hou, D. Weisleder, and W. Brown, Biotransformation of Linoleic Acid by Clavibacter sp. ALA2: Heterocyclic and Heterobicyclic Fatty Acids, Lipids 35:1055–1060 (2000).
Iwasaki, Y., W. Brown, and C.T. Hou, Biosynthetic Pathway of Diepoxy Bicyclic Fatty Acids from Linoleic Acid by Clavibacter sp. ALA2, J. Am. Oil Chem. Soc. 79:369–372 (2002).
Hosokawa, M., C.T. Hou, D. Weisleder, and W. Brown, Isolation and Structure Determination of 12,13,16-Trihydroxy-9(Z)-Octadecenoic Acid Produced from Linoleic Acid by Clavibacter sp. ALA2 and Biosynthesis of Tetrahydrofuranyl Fatty Acids, Ibid. 80:145–149 (2003).
Hosokawa, M., C.T. Hou, and D. Weisleder, Production of Novel Tetrahydrofuranyl Fatty Acids from α-Linolenic Acid by Clavibacter sp. ALA2, Appl. Environ. Microbiol. 69:3868–3873 (2003).
Hosokawa, M., C.T. Hou, D. Weisleder, and W. Brown, Formation of Cyclic Products from Polyunsaturated Fatty Acids by Clavibacter sp. ALA2, in Essential Fatty Acids and Eicosanoids: Invited Papers from the Fifth International Congress, edited by Y.-S. Huang, S.-J. Lin, and P.-C. Huang, AOCS Press, Champaign, 2003, pp. 36–41.
Miura, Y., and A.J. Fulco, ω−1, ω−2 and ω−3 Hydroxylation of Long-Chain Fatty Acids, Amides and Alcohols by a Soluble Enzyme System from Bacillus megaterium, Biochim. Biophys, Acta 388:305–317 (1975).
Bains, S.K., S.M. Gardiner, K. Mannweiler, D. Gillett and G.G. Gibson, Immunochemical Study on the Contribution of Hypolipidaemic-Induced Cytochrome P-452 to the Metabolism of Lauric Acid and Arachidonic Acid, Biochem. Pharm. 34:3221–3229 (1985).
Porter, T.D., and M.J. Coon, Cytochrome P-450. Multiplicity of Isoforms, Substrates, and Catalytic and Regulatory Mechanisms, J. Biol. Chem. 266:13469–13472 (1991).
Moghaddam, M.F., K. Motoba, B. Borhan, F. Pinot, and B.D. Hammock. Novel Metabolic Pathways for Linoleic Acid and Arachidonic Acid Metabolism, Biochim. Biophys. Acta 1290:327–339.
Kato, T., Y. Yamaguchi, N. Abe, T. Uyehara, T. Nakai, S. Yamanaka, and N. Harada, Unsaturated Hydroxy Fatty Acids, the Self-Defensive Substances in Rice Plant Against Blast Disease, Chem. Lett. 25:409–412 (1984).
Hou, C.T., and R.J. Forman III, Growth Inhibition of Plant Pathogenic Fungi by Hydroxy Fatty Acids, J. Ind. Microbiol. Biotechnol. 24:275–276.
Kawagishi, H., M. Ando, T. Mizuno, H. Yokota, and S. Konish, A Novel Fatty Acid from Mushroom Hericium erinaceum, Agri. Biol. Chem. 54:1329–1331 (1990).
Markaverich, B.M., M.A. Alejandro, D. Markaverich, L. Zitzow, N. Casajuna, N. Camarao, J. Hill, K. Bhirdo, R. Faith, J. Turk, and J.R. Crowley, Identification of an Endocrine Disrupting Agent from Corn with Mitogenic Activity, Biochem. Biophys. Res. Commun. 291:692–700 (2002).
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Hosokawa, M., Hou, C.T. & Weisleder, D. Bioconversion of n−3 and n−6 PUFA by Clavibacter sp. ALA2. J Amer Oil Chem Soc 80, 1085–1091 (2003). https://doi.org/10.1007/s11746-003-0824-8
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11746-003-0824-8