Abstract
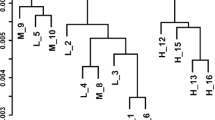
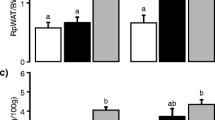
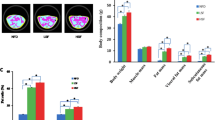
The AIN-76A diet causes fatty liver in rodents when fed for long periods of time. The aim of this study was to utilize fatty acid analysis and transcriptomics to investigate the effects of different fat sources in the AIN-76A diet on tissue lipid profiles and gene expression in male, weanling Fischer-344 rats. Animals were fed isocaloric diets that differed only in the fat source: (1) corn oil (CO) (2) anhydrous milk fat (AMF), and (3) AMF supplemented with 10 % phospholipids from the milk fat globule membrane (AMF-MFGM). There were no differences in food intake, body weight, growth rate, or body fat composition among the groups, and the fatty acid compositions of red blood cells (RBC), plasma, muscle, and visceral adipose tissues reflected the dietary fat sources. Modifying the fat source resulted in 293 genes differentially regulated in skeletal muscle, 1,124 in adipose, and 831 in liver as determined by analysis of variance (ANOVA). Although tissue fatty acid profiles mostly reflected the diet, there were several quantitative differences in lipid classes in the liver and plasma. The AMF diet resulted in the highest level of hepatic triacylglycerols, but the lowest level in plasma. The CO diet resulted in significant accumulation of hepatic unesterified fatty acids and decreased DGAT expression and activity, a potential trigger for steatohepatitis. These results indicate that the fatty acid composition and presence of polar lipids in the AIN-76A diets have significant effects on lipid partitioning, gene expression, and potentially the development of liver pathology.







Similar content being viewed by others
Abbreviations
- ACF:
-
Aberrant crypt foci
- AMF:
-
Anhydrous milk fat
- ANOVA:
-
Analysis of variance
- BHT:
-
Butylated hydroxytoluene
- CE:
-
Cholesteryl esters
- CHD:
-
Coronary heart disease
- CO:
-
Corn oil
- FAME:
-
Fatty acid methyl ester(s)
- FFA:
-
Unesterified fatty acids
- HUFA:
-
Highly unsaturated fatty acid(s)
- MFGM:
-
Milk fat globule membrane
- NAFLD:
-
Nonalcoholic fatty liver disease
- NASH:
-
Nonalcoholic steatohepatitis
- PL:
-
Phospholipid(s)
- PUFA:
-
Polyunsaturated fatty acids
- RBC:
-
Red blood cell(s)
- TAG:
-
Triacylglycerol(s)
- TLC:
-
Thin-layer chromatography
References
Reeves PG, Nielsen FH, Fahey GC Jr (1993) AIN-93 purified diets for laboratory rodents: final report of the American Institute of Nutrition ad hoc writing committee on the reformulation of the AIN-76A rodent diet. J Nutr 123(11):1939–1951
Roebuck BD, Wilpone SA, Fifield DS, Yager JD Jr (1979) Hemorrhagic deaths with AIN-76 diet. J Nutr 109(5):924–925
Nguyen HT, Woodard JC (1980) Intranephronic calculosis in rats: an ultrastructural study. Am J Pathol 100(1):39–56
Medinsky MA, Popp JA, Hamm TE, Dent JG (1982) Development of hepatic lesions in male Fischer-344 rats fed AIN-76A purified diet. Toxicol Appl Pharmacol 62(1):111–120
Nutrition A (1980) Second report of the ad hoc committee on standards for nutritional studies. J Nutr 110:1726
Council NR (1995) Nutrient requirements of laboratory animals, vol 4th Revised. National Academy Press, Washington, p 172
Corpet DE, Tache S (2002) Most effective colon cancer chemopreventive agents in rats: a systematic review of aberrant crypt foci and tumor data, ranked by potency. Nutr Cancer 43(1):1–21
Targher G, Day CP, Bonora E (2010) Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. N Engl J Med 363(14):1341–1350
Bacon BR, Park CH, Fowell EM, McLaren CE (1984) Hepatic steatosis in rats fed diets with varying concentrations of sucrose. Fundam Appl Toxicol 4(5):819–826
Snow DR, Jimenez-Flores R, Ward RE, Cambell J, Young MJ, Nemere I, Hintze KJ (2010) Dietary milk fat globule membrane reduces the incidence of aberrant crypt foci in Fischer-344 rats. J Agric Food Chem 58(4):2157–2163
Jensen RG (1995) Handbook of milk composition: food science and technology international series. Academic, San Diego, p 919
Mcpherson AV, Dash MC, Kitchen BJ (1984) Isolation of bovine-milk fat globule-membrane material from cream without prior removal of caseins and whey proteins. J Dairy Res 51(1):113–121
MacGibbon A, Taylor M (2006) Composition and structure of bovine milk lipids. In: Fox PF, McSweeney PLH (eds) Advanced dairy chemistry. Springer, New York, pp 1–42
Spitsberg VL (2005) Invited review: bovine milk fat globule membrane as a potential nutraceutical. J Dairy Sci 88:2289–2294
Folch J, Lees M, Sloane Stanley GH (1957) A simple method for the isolation and purification of total lipides from animal tissues. J Biol Chem 226(1):497–509
Curtis JM, Berrigan N, Dauphinee P (2008) The determination of n-3 fatty acid levels in food products containing microencapsulated fish oil using the one-step extraction method. Part 1: measurement in the raw ingredient and in dry powdered foods. J Am Oil Chem Soc 85(4):297–305
Blazejczyk M, Miron M, Nadon R (2007) FlexArray: a statistical data analysis software for gene expression microarrays. http://genomequebec.mcgill.ca/FlexArray. Accessed 8 Oct 2012
Long AD, Mangalam HJ, Chan BY, Tolleri L, Hatfield GW, Baldi P (2001) Improved statistical inference from DNA microarray data using analysis of variance and a Bayesian statistical framework: analysis of global gene expression in Escherichia coli K12. J Biol Chem 276(23):19937–19944
Hochberg Y, Benjamini Y (1990) More powerful procedures for multiple significance testing. Stat Med 9(7):811–818
Tong W, Cao X, Harris S, Sun H, Fang H, Fuscoe J, Harris A, Hong H, Xie Q, Perkins R, Shi L, Casciano D (2003) ArrayTrack–supporting toxicogenomic research at the US Food and Drug Administration National Center for Toxicological Research. Environ Health Perspect 111(15):1819–1826
Lopez-Parra M, Titos E, Horrillo R, Ferre N, Gonzalez-Periz A, Martinez-Clemente M, Planaguma A, Masferrer J, Arroyo V, Claria J (2008) Regulatory effects of arachidonate 5-lipoxygenase on hepatic microsomal TG transfer protein activity and VLDL-triglyceride and apoB secretion in obese mice. J Lipid Res 49(12):2513–2523
Abramoff MD, Magalhaes PJ, Ram SJ (2004) Image processing with ImageJ. Biophotonics Int 11(7):36–42
Ko JS, Ryu SY, Kim YS, Chung MY, Kang JS, Rho MC, Lee HS, Kim YK (2002) Inhibitory activity of diacylglycerol acyltransferase by tanshinones from the root of Salvia miltiorrhiza. Arch Pharm Res 25(4):446–448
Coleman RA (1992) Diacylglycerol acyltransferase and monoacylglycerol acyltransferase from liver and intestine. Methods Enzymol 209:98–104
McFie PJ, Stone SJ (2011) A fluorescent assay to quantitatively measure in vitro acyl CoA:diacylglycerol acyltransferase activity. J Lipid Res 52(9):1760–1764
Harris WS, Von Schacky C (2004) The omega-3 index: a new risk factor for death from coronary heart disease? Prev Med 39(1):212–220
Lands B (2008) A critique of paradoxes in current advice on dietary lipids. Prog Lipid Res 47(2):77–106
O’Rourke EJ, Soukas AA, Carr CE, Ruvkun G (2009) C. elegans major fats are stored in vesicles distinct from lysosome-related organelles. Cell Metab 10(5):430–435
Yamaguchi K, Yang L, McCall S, Huang J, Yu XX, Pandey SK, Bhanot S, Monia BP, Li YX, Diehl AM (2007) Inhibiting triglyceride synthesis improves hepatic steatosis but exacerbates liver damage and fibrosis in obese mice with nonalcoholic steatohepatitis. Hepatology 45(6):1366–1374
Romestaing C, Piquet MA, Bedu E, Rouleau V, Dautresme M, Hourmand-Ollivier I, Filippi C, Duchamp C, Sibille B (2007) Long term highly saturated fat diet does not induce NASH in Wistar rats. Nutr Metab (Lond) 4:4
Li-Hawkins J, Gafvels M, Olin M, Lund EG, Andersson U, Schuster G, Bjorkhem I, Russell DW, Eggertsen G (2002) Cholic acid mediates negative feedback regulation of bile acid synthesis in mice. J Clin Invest 110(8):1191–1200
Gafvels M, Olin M, Chowdhary BP, Raudsepp T, Andersson U, Persson B, Jansson M, Bjorkhem I, Eggertsen G (1999) Structure and chromosomal assignment of the sterol 12alpha-hydroxylase gene (CYP8B1) in human and mouse: eukaryotic cytochrome P-450 gene devoid of introns. Genomics 56(2):184–196
Noh SK, Koo SI (2004) Milk sphingomyelin is more effective than egg sphingomyelin in inhibiting intestinal absorption of cholesterol and fat in rats. J Nutr 134(10):2611–2616
Kamili A, Wat E, Chung RW, Tandy S, Weir JM, Meikle PJ, Cohn JS (2010) Hepatic accumulation of intestinal cholesterol is decreased and fecal cholesterol excretion is increased in mice fed a high-fat diet supplemented with milk phospholipids. Nutr Metab (Lond) 7:90
Wat E, Tandy S, Kapera E, Kamili A, Chung RW, Brown A, Rowney M, Cohn JS (2009) Dietary phospholipid-rich dairy milk extract reduces hepatomegaly, hepatic steatosis and hyperlipidemia in mice fed a high-fat diet. Atherosclerosis 205(1):144–150
Kersten S (2005) Regulation of lipid metabolism via angiopoietin-like proteins. Biochem Soc Trans 33(Pt 5):1059–1062
Backhed F, Ding H, Wang T, Hooper LV, Koh GY, Nagy A, Semenkovich CF, Gordon JI (2004) The gut microbiota as an environmental factor that regulates fat storage. Proc Natl Acad Sci U S A 101(44):15718–15723
Acknowledgments
This study was supported by both a seed grant from the Center for Integrated Biosystems at Utah State University and by the Utah Agricultural Experiment Station and was approved as journal paper number 8290. We are grateful to Dallin Snow for preparing RNA samples for Microarray analysis and Dr. Aaron Olsen and Kent Udy for assistance with animal studies.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Zhou, A.L., Hintze, K.J., Jimenez-Flores, R. et al. Dietary Fat Composition Influences Tissue Lipid Profile and Gene Expression in Fischer-344 Rats. Lipids 47, 1119–1130 (2012). https://doi.org/10.1007/s11745-012-3729-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-012-3729-3