Abstract
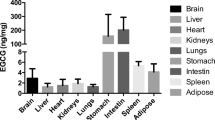
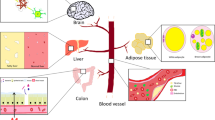
Amyloid-β (Aβ) is secreted from lipogenic organs such as intestine and liver as an apolipoprotein of nascent triacylglycerol rich lipoproteins. Chronically elevated plasma Aβ may compromise cerebrovascular integrity and exacerbate amyloidosis—a hallmark feature of Alzheimer’s disease (AD). Probucol is a hypocholesterolemic agent that reduces amyloid burden in transgenic amyloid mice, but the mechanisms for this effect are presently unclear. In this study, the effect of Probucol on intestinal lipoprotein-Aβ homeostasis was explored. Wild-type mice were fed a control low-fat diet and enterocytic Aβ was stimulated by high-fat (HF) diet enriched in 10% (w/w) saturated fat and 1% (w/w) cholesterol for the duration of 1 month. Mice treated with Probucol had the drug incorporated into the chow at 1% (w/w). Quantitative immunofluorescence was utilised to determine intestinal apolipoprotein B (apo B) and Aβ abundance. We found apo B in both the perinuclear region of the enterocytes and the lacteals in all groups. However, HF feeding and Probucol treatment increased secretion of apo B into the lacteals without any change in net villi abundance. On the other hand, HF-induced enterocytic perinuclear Aβ was significantly attenuated by Probucol. No significant changes in Aβ were observed within the lacteals. The findings of this study support the notion that Probucol suppresses dietary fat induced stimulation of Aβ biosynthesis and attenuate availability of apo B lipoprotein-Aβ for secretion.


Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- Aβ:
-
Amyloid-β
- Apo B:
-
Apolipoprotein B
- HF:
-
High-fat
- LF:
-
Low-fat
- TAG:
-
Triacylglycerol(s)
References
Pastorino L, Lu KP (2006) Pathogenic mechanisms in Alzheimer’s disease. Eur J Pharmacol 545:29–38
Selkoe DJ (1994) Cell biology of the amyloid beta-protein precursor and the mechanism of Alzheimer’s disease. Annu Rev Cell Biol 10:373–403
Kapeller R, Cantley L (1994) Phosphatidylinositol 3-kinase. BioEssays 16:565–576
Luo Y, Hirashima N, Li Y, Alkon D, Sunderland T, Etcheberrigaray R, Wolozin B (1995) Physiological levels of β-amyloid increase tyrosine phosphorylation and cytosolic calcium. Brain Res 681:65–74
Luo Y, Sunderland T, Roth GS, Wolozin B (1996) Physiological levels of β-amyloid peptide promote PC12 cell proliferation. Neurosci Lett 217:125–128
Jekabsone A, Mander P, Tickler A, Sharpe M, Brown G (2006) Fibrillar beta-amyloid peptide Abeta1–40 activates microglial proliferation via stimulating TNF-alpha release and H2O2 derived from NADPH oxidase: a cell culture study. J Neuroinflamm 3:24
Sondag C, Dhawan G, Combs C (2009) Beta amyloid oligomers and fibrils stimulate differential activation of primary microglia. J Neuroinflamm 6:1
Haass C, Koo EH, Mellon A, Hung AY, Selkoe DJ (1992) Targeting of cell-surface beta-amyloid precursor protein to lysosomes-alternative processing into amyloid-bearing fragments. Nature 357:500–503
Panza F, Solfrizzi V, D’lntrono A, Capurso C, Colacicco A, Torres F, Altomare E, Capurso A (2002) Genetics of late-onset Alzheimer’s disease: vascular risk and beta-amyloid metabolism. Recenti Prog Med 93:489–497
Rocchi A, Orsucci D, Tognoni G, Ceravolo R, Siciliano G (2009) The role of vascular factors in late-onset sporadic Alzheimer’s disease. Genetic and molecular aspects. Curr Alzheimer Res 6:224–237
Crossgrove J, Li G, Zheng W (2005) The choroid plexus removes beta-amyloid from brain cerebrospinal fluid. Exp Biol Med (Maywood) 230:771–776
Deane R, Sagare A, Hamm K, Parisi M, LaRue B, Guo H, Wu Z, Holtzman D, Zlokovic B (2005) IgG-assisted age-dependent clearance of Alzheimer’s amyloid beta peptide by the blood-brain barrier neonatal Fc receptor. J Neurosci 25:11495–11503
Mamo JCL, Jian L, James AP, Flicker L, Esselmann H, Wiltfang J (2008) Plasma lipoprotein β-amyloid in subjects with Alzheimer’s disease or mild cognitive impairment. Ann Clin Biochem 45:395–403
Burges B, McIsaac S, Naus K, Chan J, Tansley G, Yang J, Miao F, Ross C, van Eck M, Hayden M, van Nostrand W, St George-Hyslop P, Westaway D, Wellington C (2006) Elevated plasma triglyceride levels precede amyloid deposition in Alzheimer’s disease mouse models with abundant A beta in plasma. Neurobiol Dis 24:114–127
Namba Y, Tsuchiya H, Ikeda K (1992) Apolipoprotein B immunoreactivity in senile plaque and vascular amyloids and neurofibrillary tangles in the brains of patients with Alzheimer’s disease. Neurosci Lett 134:264–266
Takechi R, Galloway S, Pallebage-Gamarallage MMS, Wellington CL, Johnsen RD, Dhaliwal SS, Mamo JCL (2010) Differential effects of dietary fatty acids on the cerebral distribution of plasma-derived apo B lipoproteins with amyloid-β. Br J Nutr 103:652–662
Funatsu T, Suzuki K, Goto M, Arai Y, Kakuta H, Tanaka H, Yasuda S, Ida M, Nishijima S, Miyata K (2001) Prolonged inhibition of cholesterol synthesis by atorvastatin inhibits apo B-100 and triglyceride secretion from HepG2 cells. Atherosclerosis 157:107–115
Watts GF, Chan DC, Barrett PHR, O’Neill FH, Thompson GR (2003) Effect of a statin on hepatic apolipoprotein B-100 secretion and plasma campesterol levels in the metabolic syndrome. Int J Obes Relat Metab Disord 27:862–865
Pal S, Allister E, Thomson A, Mamo JCL (2002) Cholesterol esters regulate apoB48 secretion in CaCo2 cells. Atherosclerosis 161:55–63
Staels B, Dallongeville J, Auwerx J, Schoonjans K, Leitersdorf E, Fruchart J-C (1998) Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation 98:2088–2093
Dufouil C, Richard F, Fievet N, Dartigues J, Ritchie K, Tzourio C, Amouyel P, Alperovitch A (2005) APOE genotype, cholesterol level, lipid-lowering treatment, and dementia: the three-city study. Neurology 64:1531–1538
Rockwood K, Kirkland S, Hogan D, MacKnight C, Merry H, Verreault R, Wolfson C, McDowell I (2002) Use of lipid-lowering agents, indication bias, and the risk of dementia in community-dwelling elderly people. Arch Neurol 59:223–227
Mamo J, Elsegood C, Umeda Y, Hirano T, Redgrave T (1993) Effect of probucol on plasma clearance and organ uptake of chylomicrons and VLDLs in normal and diabetic rats. Arterioscler Thromb 13:231–239
Mellies MJ, Gartside PS, Glatfelter L, Vink P, Guy G, Schonfeld G, Glueck CJ (1980) Effects of probucol on plasma cholesterol, high and low density lipoprotein cholesterol, and apolipoproteins A1 and A2 in adults with primary familial hypercholesterolemia. Metabolism 29:956–964
Gershkovich P, Hoffman A (2005) Uptake of lipophilic drugs by plasma derived isolated chylomicrons: linear correlation with intestinal lymphatic bioavailability. Eur J Pharm Sci 26:394–404
Poirier J (2003) Apolipoprotein E and cholesterol metabolism in the pathogenesis and treatment of Alzheimer’s disease. Trends Mol Med 9:94–101
Poirier J (2005) Apolipoprotein E, cholesterol transport and synthesis in sporadic Alzheimer’s disease. Neurobiol Aging 26:355–361
Galloway S, Jian L, Johnsen R, Chew S, Mamo JCL (2007) β-Amyloid or its precursor protein is found in epithelial cells of the small intestine and is stimulated by high-fat feeding. J Nutr Biochem 18:279–284
Pallebage-Gamarallage MM, Galloway S, Johnsen R, Jian L, Dhaliwal S, Mamo JCL (2009) The effect of exogenous cholesterol and lipid-modulating agents on enterocytic amyloid-β abundance. Br J Nutr 101:340–347
Galloway S, Pallebage-Gamarallage MM, Takechi R, Jian L, Johnsen RD, Dhaliwal SS, Mamo JC (2008) Synergistic effects of high fat feeding and apolipoprotein E deletion on enterocytic amyloid-beta abundance. Lipids Health Dis 22:7–15
Takechi R, Galloway S, Pallebage-Gamarallage M, Johnsen R, Mamo J (2008) Three-dimensional immunofluorescent double labelling using polyclonal antibodies derived from the same species: enterocytic colocalization of chylomicrons with Golgi apparatus. Histochem Cell Biol 129:779–784
Lairon D (2008) Macronutrient intake and modulation on chylomicron production and clearance. Atheroscler Suppl 9:45–48
Galloway S, Takechi R, Pallebage-Gamarallage M, Dhaliwal S, Mamo J (2009) Amyloid-beta colocalizes with apolipoprotein B in absorptive cells of the small intestine. Lipids Health Dis 8:46
Smith D, Watts GF, Dane-Stewart C, Mamo JC (1999) Post-prandial chylomicron response may be predicted by a single measurement of plasma apolipoprotein B48 in the fasting state. Eur J Clin Invest 29:204–209
Ohtani H, Hayashi K, Hirata Y, Dojo S, Nakashima K, Nishio E, Kurushima H, Saeki M, Kajiyama G (1990) Effects of dietary cholesterol and fatty acids on plasma cholesterol level and hepatic lipoprotein metabolism. J Lipid Res 31:1413–1422
Sharon A, Michael IM, Paul ND (2000) The effects of fatty acids on apolipoprotein B secretion by human hepatoma cells (HEP G2). Atherosclerosis 150:255–264
Runz H, Rietdorf J, Tomic I, de Bernard M, Beyreuther K, Pepperkok R, Hartmann T (2002) Inhibition of intracellular cholesterol transport alters presenilin localisation and amyloid precursor protein processing in neuronal cells. J Neurosci 22:1679–1689
Tawara K, Tomikawa M, Abiko Y (1986) Mode of action of probucol in reducing serum cholesterol in mice. Jpn J Pharmacol 40:123–133
Champagne D, Pearson D, Dea D, Rochford J, Poirier J (2003) The cholesterol-lowering drug probucol increases apolipoprotein E production in the hippocampus of aged rats: implications for Alzheimer’s disease. Neuroscience 121:99–110
Acknowledgments
The National Health and Medical Research Council of Australia, and the Australian Technology Network Centre supported this research financially.
Conflict of interest
The authors have no conflicts of interest to declare in relation to this article.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Pallebage-Gamarallage, M.M., Galloway, S., Takechi, R. et al. Probucol Suppresses Enterocytic Accumulation of Amyloid-β Induced by Saturated Fat and Cholesterol Feeding. Lipids 47, 27–34 (2012). https://doi.org/10.1007/s11745-011-3595-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-011-3595-4