Abstract
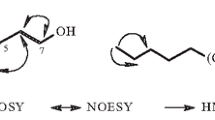
Two novel ceramides, Candidamide A (1) with a phytosphingolipid structure, and Candidamide B (2) with a tertiary amide structure, together with 12 known compounds (3–14) have been isolated from the bulbs of Zephyranthes candida, The structures of 1 and 2 have been elucidated to be 1,3,5,6-tetrahydroxy-2-(2′-hydroxytetracosanoyl amino)-8-(E)-octadecadiene (1) and (2S,3S,4R,8E,2′R)-2-[N-(2′-hydroxyoctadecanoyl)-N-(1′′,2′′-dihydroxyethyl)-amino]-8-hexacosene-1,3,4-triol (2) on the basis of spectroscopic evidence including IR, MS, NMR (1H-NMR, 13C-NMR, DEPT, 1H–1H COSY, HSQC, HMBC). The known compounds were identified as (2S)-3′,7-dihydroxy-4′-methoxyflavan (3), (2S)-4′-hydroxy-7-methoxyflavan (4), (2S)-4′,7-dihydroxyflavan (5), 7-hydroxy-3′, 4′-methylenedioxyflavan (6), ambrettolide (7), β-sitostero1 (8), β-daucosterin (9), rutin (10), pancratistatin (11), lycorine (12), haemanthidine (13), and haemanthamine (14). In the antimicrobial assay, candidamide A (1) and candidamide B (2) displayed moderate activities against bacteria Staphylococcus aureus and Escherichia coli, and fungi Aspergillus niger, Candida albicans and Trichophyton rubrum.






Similar content being viewed by others
Abbreviations
- CC:
-
Column chromatography
- COSY:
-
Correlation spectroscopy
- DEPT:
-
Distortionless enhancement by polarization transfer
- HMBC:
-
Heteronuclear multiple bond correlation
- HSQC:
-
Heteronuclear single quantum correlation
- HR:
-
High resolution
- LCB:
-
Long chain base
- FA:
-
Fatty acid
- MICs:
-
Minimum inhibitory concentrations
References
Thoibi DT, Borua PK (1997) Meiotic behaviour and pollen fertility in three species of Zephyranthes (Amaryllidaceae). Biol Plant 39:355–360
Pettit GR, Gaddamidi V, Cragg GM (1984) Antineoplastic agents, 105. Zephyranthes grandiflora. J Nat Prod 47:1018–1020
Ghosal S, Singh SK, Srivastava RS (1986) Alkaloids of Zephyranthes flava. Phytochemistry 25:1975–1978
Ghosal S, Singh SK, Srivastava RS (1985) Flavans from Zephyranthes flava. Phytochemistry 24:151–153
Ghosal S, Singh SK, Unnikrishnan G (1987) Chemical constituents of Amaryllidaceae. Part 25. Phosphatidylpyrrolophenanthridine alkaloids from Zephyranthes flava. Phytochemistry 26:823–828
Zaida TA, Myrna CR, Iraida SS (1989) Preliminary study of Zephyranthes eggersiana Urban. Revista Cubana de Farmacia 23:147–150
Iraida SS, Zaida TA (1989) Determination of the presence of lactams in the plant Zephyranthes tubispatha Herb, Amaryllidaceae. Revista Cubana de Farmacia 23(1–2):151–154
Kojima K, Mutsuga M, Inoue M, Ogihara Y (1998) Two alkaloids from Zephyranthes carinata. Phytochemistry 48:1199–1202
Mutsuga M, Kojima K, Nose M, Inoue M, Ogihara Y (2001) Cytotoxic activities of alkaloids from Zephyranthes carinata. Nat Med 55:201–204
Herrera MR, Machocho AK, Brun R, Viladomat F, Codina C, Bastida J (2001) Crinane and lycorane type alkaloids from Zephyranthes citrina. Planta Med 67:191–193
Mitsuru N, Tokunaru H, Masao T, Mitsuo M, Shuichi H (1978) Isolation of kaempferol-3-O-rhamnoglucoside, a flavonoid glycoside from Zephyranthes candida. J Biosci 33C:587–588
Ozeki S (1964) Alkaloids of Zephyranthes candida. I. Isolation of bases. Yakugaku Zasshi. 84:1194–1197
Pettit GR, Cragg GM, Singh SB, Duke JA, Doubek DL (1990) Antineoplastic agents, 162. Zephyranthes candida. J Nat Prod 53:176–178
Li X, Sun DD, Chen JW, He LW, Zhang HQ, Xu HQ (2007) New sphingolipids from the root of Isatis indigotica and their cytotoxic activity. Fitoterapia 78:490–495
Li HY, Matsunaga S, Fusetani N (1995) Halicylindrosides, antifungal and cytotoxic cerebrosides from the marine sponge Halichondria cylindroata. Tetrahedron 51:2273–2280
Zhang Y, Wang S, Li XM, Cui CM, Feng C, Wang BG (2007) New sphingolipids with a previously unreported 9-methyl-C20-sphingosine moiety from a marine algous endophytic fungus Aspergillus niger EN-13. Lipids 42:759–764
Chen JH, Cui GY, Liu JY, Tan RX (2003) Pinelloside, an antimicrobial cerebroside from Pinellia ternata. Phytochemistry 64:903–906
Cateni F, Zilic J, Falsone G, Scialino G, Banfi E (2003) New cerebrosides from Euphorbia peplis L.: antimicrobial activity evaluation. Bioorg Med Chem Lett 13:4345–4350
Mishra PK, Singh N, Ahmad G, Dube A, Maurya R (2005) Glycolipids and other constituents from Desmodium gangeticum with antileishmanial and immunomodulatory activities. Bioorg Med Chem Lett 15:4543–4546
Cateni F, Zilic J, Falsone G, Hollan F, Frausin F, Scarcia V (2003) Preliminary biological assay on cerebroside mixture from Euphorbia nicaeensis All. Isolation and structure determination of five glucocerebrosides. IL Farmaco 58:809–817
Natori T, Morita M, Akimoto K, Koezuka Y (1994) Agelasphins, novel antitumor and immunostimulatory cerebrosides from the marine sponge Agelas mauritianus. Tetrahedron 50:2771–2784
Gao JM, Yang X, Wang CY, Liu JK (2001) Armillaramide, a new sphingolipid from the fungus Armillaria mellea. Fitoterapia 72:857–972
Higuchi R, Inagaki M, Togawa K, Miyamoto T, Komori T (1994) Isolation and structure of cerebrosides from the sea cucumber Pentacta australis. Liebigs Ann Chem 7:653–658
Ouyang MA, Liu R, Kuo YH (2005) A new cerebrosides, asperiamide A, from the marine fungus Asperillus sp. J Asian Nat Prod Res 7:761–765
Simo CCF, Kouam SF, Poumale HMP, Simo IK, Ngadjui BT, Green IR, Krohn K (2008) Benjaminamide: a new ceramide and other compounds from the twigs of Ficus benjamina (Moraceae). Biochem Syst Ecol 36:238–243
Chen XS, Wu YL, Chen DH (2002) Structure determination and synthesis of a new cerebrosides isolation from the traditional Chinese medicine Typhonium giganteum Engl. Tetrahedron Lett 43:3529–3532
Kumar N, Singh B, Gupta AP, Kaul VK (2006) Lonijaposides, novel cerebrosides from Lonicer japonica. Tetrahedron 62:4317–4322
Chen XS, Chen DH, Si JY, Guang ZT (2001) Chemical constituents of Typhonium giganteum Engl. J Asian Nat Prod Res 3:277–283
Su BN, Takaishi Y (1999) Morinins H-K, four novel phenylpropanol ester lipid metabolites from Morina chinensis. J Nat Prod 62:1325–1327
Shibuya H, Kawashima K, Sakagami M, Kawanishi H, Shimomura M, Ohashi K, Kitagawa I (1990) Sphingolipids and glycerolipids.I. Chemical structure and ionophoretic activities of soyacerebrosides I and II from soybean. Chem Pharm Bull 38:2933–2938
Liu A, Zou ZM, Xu LZ, Yang SL (2005) A new cerebroside from Uvaria tonkinensis var. subglabra. J Asian Nat Prod Res 7:861–865
Oueslati MH, Mighri Z, Jannet HB, Abreu PM (2005) New ceramides from Rantherium suaveolens. Lipids 40:1075–1079
Lin YP, Yan J, Qiu MH (2006) Novel imine from Hemsleya macrocarpa var. clavata. Lipids 41:97–99
Masaoud M, Ripperger H, Porzel A (1995) Flavonoids of dragon’s blood from Dracaena cinnabari. Phytochemistry 38:745–749
Achenbach H, Stocker M, Manael AC (1988) Flavonoid and other constituents of Bauhinia manca. Phytochemistry 27:1835–1842
San V, Seoan E (1982) Synthesis of ambrettolide from phloionolic acid. Perkin Trans I 7:1837–1838
Likhitwitayawuid K, Angerhofer CK, Chai H, Pezzuto JM, Cordell GA (1993) Cytotoxic and antimalarial alkaloids from the bulbs of Crinum amabile. J Nat Prod 8:1331–1338
Kihara M, Konishi K, Xu L, Kobayashi S (1991) Alkaloidal constituents of the flowers of Lycoris radiata Herb (Amaryllidaceae). Chem Pharm Bull 39:1849–1853
Acknowledgments
This work was financially supported in part by the Natural Science Foundation of Jiangsu Province (BK2004062). The authors wish to acknowledge Professor Y.-L. Wu at State Key Laboratory of Bio-organic and Natural Products Chemistry, Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences, Shanghai China, for making suggestions on this paper.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Wu, Zp., Chen, Y., Xia, B. et al. Two Novel Ceramides with a Phytosphingolipid and a Tertiary Amide Structure from Zephyranthes candida . Lipids 44, 63–70 (2009). https://doi.org/10.1007/s11745-008-3246-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-008-3246-6