Abstract
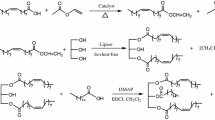
A novel phosphonium salt methodology was utilized for the first time to synthesize 1,3-, and 1,2-diphosphatidylglycerol. Optically active 1,2-di-O-acyl-sn-glyceryl phosphate was coupled with unprotected glycerol in the presence of pyridiniumbromide perbromide and triethylamine to yield, after final removal of phosphate protecting group, the title compounds. The 1,2-diphosphatidylglycerol (1,2-isomer of cardiolipin) may be a member of a new class of phospholipids for industrial applications similar to other phosphocholines.


Similar content being viewed by others
Abbreviations
- ATR:
-
Attenuated total reflectance
- CL:
-
Cardiolipin
- DIPEA:
-
Diisopropylethylamine
- ESI:
-
Electron spray ionization
- IR:
-
Infra red
- MS:
-
Mass spectrometry
- NMR:
-
Nuclear magnetic resonance
- PA:
-
Phosphatidic acid
- PBP:
-
Pyridiniumbromide perbromide
- PG:
-
Phosphatidylglycerol
- rt:
-
Room temperature
- TBHP:
-
tert-Butylhydroperoxide
- THF:
-
Tetrahydrofuran
- TLC:
-
Thin layer chromatography
References
Arrigo PD, Ferra LD, Fantoni GP, Scarcelli D, Servi S, Strini A (1996) Enzyme-mediated synthesis of two diastereoisomeric forms of phosphatidylglycerol and of diphosphatidylglycerol (cardiolipin). J Chem Soc Perkin Trans 1(21):2657–2660
Keana JFW, Shimiju M, Jernstedt KK (1986) A short, flexible route to symmetrically and unsymmetrically substituted diphosphatidylglycerols (cardiolipin). J Org Chem 51:2297–2299
Mishina IM, Vasilenko AE, Stepanov AE, Shvets VI (1987) Studies on complex lipids. Synthesis of diphosphatidylglycerol (cardiolipin) with unsaturated fatty acids. Bioorg Khim 13:1110–1115
Mishina IM, Vasilenko AE, Stepanov AE, Shvets VI (1984) Study of lipids. Synthesis of phosphatidylglycerol and diphosphatidylglycerol. Zh Org Khim 20:985–988
Mishina IM, Vasilenko AE, Stepanov AE, Shvets VI (1985) Synthesis of diphosphatidylglycerol (cardiolipin) with unsaturated fatty acids. Bioorg Khim 11:992–994
Ioannou PV, Marecek JF (1986) Studies on the chemical synthesis and stability of cardiolipins and related compounds. Chem Chorn 15:205–220
Ramirez F, Ioannou PV, Marecek JF, Dodd GH, Golding BT (1977) Synthesis of phospholipids by means of cyclic enediol pyrophosphates. Tetrahedron 33:599–608
Ramirez F, Ioannou PV, Marecek JF, Golding BT, Dodd GH (1976) Application of cyclic enediol pyrophosphates to the synthesis of phospholipids. Diphosphatidylglycerol (cardiolipin). Synthesis 11:769–770
Inoue K, Nojima S (1968) Immunochemical studies of phosoholipids. II. Synthesis of cardiolipin and its analogues. Chem Pharm Bull 16:76–81
Inoue K, Nojima S (1963) On the cardiolipin analogues. Synthesis of dipalmitoyl-d,l-α-glycerylphosphoryl-propanol sodium salt and bis(dipalmitoyl-d,l-α-glycerylphosphoryl)-1,3-propanediol disodium salt. Chem Pharm Bull 11:1150–1156
Saunders RM, Schwarz HP (1966) Synthesis of phosphatidylglycerol and diphosohatidylglycerol. J Am Chem Soc 88:3844–3847
Duralski AA, Spooner PJR, Rankin SE, Watts A (1998) Synthesis of isotopically labelled cardiolipins. Tetrahedron Lett 39:11607–1610
Krishna UM, Ahmad MU, Ahmad I (2004) Phosphoramidite approach for the synthesis of cardiolipin. Tetrahedron Lett 45:2077–2079
Lin Z, Ahmad MU, Ali SM, Ahmad I (2004) An efficient and novel method for the synthesis of cardiolipin and its analogs. Lipids 39:285–290
Krishna UM, Ahmad MU, Ali SM, Ahmad I (2004) A short, concise route to diphosphatidylglycerol (cardiolipin) and its variants. Lipids 39:595–600
Browne JE, Driver MJ, Russel JC, Sammes PG (2000) Preparation of phospholipid analogues using the phosphoramidite route. J Chem Soc Perkin Trans 1(5):653–657
Beaucage SL, Iyer RP (1993) The synthesis of specific ribonucleotides and unrealated phosphorylated biomolecules by the phosphoramidite method. Tetrahedron 49:10441–10488
Watanaba Y, Hirofuji H, Ozaki S (1994) Synthesis of a phosphatidylinositol 3, 4, 5-triphosphate. Tetrahedron Lett 35:123–124
Watanaba Y, Nakamura T, Mitsumoto H (1997) Protection of phosphate with the 9-fluorenylmethyl group. Synthesis of unsaturated-acyl phosphatidylinositol 4, 5-bisphosphate. Tetrahedron Lett 38:7407–7410
Acknowledgments
We thank the Bioanalytical group of Pharmacokinetics, Safety and Efficacy (PSE) Department at NeoPharm for the Mass spectral analyses.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Ahmad, M.U., Krishna, U.M., Ali, S.M. et al. Synthesis of 1,3- and 1,2-Diphosphatidylglycerol. Lipids 42, 291–296 (2007). https://doi.org/10.1007/s11745-007-3020-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11745-007-3020-1