Abstract
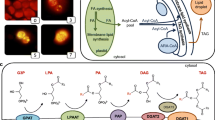
The fresh-water green alga Parietochloris incisa is the richest plant source of the PUFA arachidonic acid (20∶4n−6, AA). To elucidate the biosynthesis of AA in this alga we labeled cultures of P. incisa with radioactive precursors. Pulse chase labeling with acetate resulted in its incorporation via the de novo biosynthesis pathway of FA. However, labeled acetate was also utilized for the elongation of C16 and C18 PUFA. Labeling with [1-14C]oleic acid has shown that the first steps of the lipid-linked FA desaturations utilize cytoplasmic lipids. PC and diacylglyceryltrimethylhomoserine are the major lipids involved as acyl carriers for the Δ12 and Δ6 desaturations of oleic acid, leading sequentially to linoleic and γ-linolenic acids. The latter is released from its lipid carrier and elongated to 20∶3n−6, which is reincorporated primarily into PF and PC and finally desaturated to AA. Galactolipids, mostly monogalctosyldiacylglycerol (MGDG), serve as substrates for the chloroplastic Δ12 desaturase and, apparently, the ω3 desaturation, common to higher plants and many green algae. The predominant sequence desaturates the 18∶1/16∶0 molecular species of MGDG stepwise to the 18∶3n−3/16∶3n−3 molecular species similar to the prokaryotic pathway of higher plants and green algae.
Similar content being viewed by others
Abbreviations
- AA:
-
arachidonic acid (20∶4n−6)
- DGDG:
-
digalactosyldiacylglycerol
- DGTS:
-
diacylglyceryltrimethylhomoserine
- MGDG:
-
monogalactosyldiacylglycerol. Pairs of numbers representing the FA, when separated by a slash, designate the components in the sn-1 and sn-2 positions, respectively, of the molecular species
References
Koletzko, B., and Braun, M. (1991) Arachidonic Acid and Early Human Growth: Is There a Relation? Ann. Nutr. Metabol. 35, 128–131.
Agostoni, C., Riva, E., Bellu, R., Trojan, S., Luotti, D., and Giovannini, M. (1994) Effects of Diet on the Lipid and Fatty Acid Status of Full Term Infants At 4 Months, J. Am. Clin. Nutr. 13, 658–664.
Putnam, J.C., Carlson, S.E., De Voe, P., and Barness, L.A. (1982) The Effect of Variations in Dietary Fatty Acids on the Fatty Acid Composition of Erythrocyte Phosphatidylcholine and Phosphatidylethanolamine in Human Infants, Am. J. Clin. Nutr. 36, 106–114.
Ruyle, M., Connor, W.E., Anderson, G.J., and Lowensohn, R.I. (1990) Placental Transfer of Essential Fatty Acids in Humans: Venous Arterial Differences for Docosahexaenoic Acid in Fetal Umbilical Erythrocytes, Proc. Natl. Acad. Sci. USA 87, 7902–7906.
Carlson, S.E., Werkman, S.H., Peeples, J.M., Cooke, R.J., and Tolley, E.A. (1993) Arachidonic Acid Status Correlates with First Year Growth in Preterm Infants, Proc. Natl. Acad. Sci. USA 90, 1073–1077.
Makrides, M., Neumann, M., Simmer, K., Pater, J., and Gibson, R. (1995) Are Long-Chain Polyunsaturated Fatty Acids Essential in Infancy? Lancet 345, 1463–1468.
Watanabe, S., Hirabashi, S., Boussiba, S., Cohen, Z., Vonshak, A., and Richmond, A. (1996) Parietochloris incisa Comb. Nov. (Trebuxiophyceae, Chlorophyta), Physiol. Res. 44, 107–108.
Bigogno, C., Adlerstein, D., Khozin, I., and Cohen, Z. (1998) Biosynthesis of Arachidonic Acid in the Alga T12, in Advances in Plant Lipid Research (Sanchez, J., Cerda-Olmedo, E., and Martinez-Force, E., eds.), pp. 159–161, Universidad de Sevilla, Seville.
Bigogno, C. (2000) Biosynthesis of Arachidonic Acid (AA) in the Microalga Parietochloris incisa and the Effect of Environmental Conditions on AA Production, Ph.D. Thesis, Ben Gurion University, Israel.
Browse, J., and Somerville, C.R. (1994) Glycerolipids, in Arabidopsis (Meyerowitz, E.M., and Somerville, C.R., eds.), pp. 881–912, Cold Spring Harbor Laboratory Press, Cold Spring Harbor.
Ohlrogge, J., and Browse, J. (1995) Lipid Biosynthesis, Plant Cell 7, 957–970.
Shiran, D., Khozin, I., Heimer, Y.M., and Cohen, Z. (1996) Biosynthesis of Eicosapentaenoic Acid in the Microalga Porphyridium cruentum. I: The Use of Externally Supplied Fatty Acids, Lipids 31, 1277–1282.
Khozin, I., Adlerstein, D., Bigogno, C., Heimer, Y.M., and Cohen, Z. (1997) Elucidation of the Biosynthesis of Eicosapentaenoic Acid in the Microalga Porphyridium cruentum, Plant Physiol. 114, 223–230.
Nichols, B.W., and Appleby, R.S. (1969) The Distribution of Arachidonic Acid in Algae, Phytochemistry 8, 1907–1915.
Arao, T., Sakaki, T., and Yamada, M. (1994) Biosynthesis of Polyunsaturated Lipids in the diatom, Phaeodactylum tricornutum, Phytochemistry 36, 629–635.
Arao, T., and Yamada, M. (1994) Biosynthesis of Polyunsaturated Fatty Acids in the Marine Diatom, Phaeodactylum tricornutum, Phytochemistry 35, 1177–1181.
Giroud, C., and Eichenberger, W. (1989) Lipids of Chlamydomonas reinhardtii. Incorporation of [14C]Acetate, [14C]Palmitate and [14C]Oleate into Different Lipids and Evidence for Lipid-Linked Desaturation of Fatty Acids, Plant Cell Physiol. 30, 121–128.
Vogel, G., and Eichenberger, W. (1992) Betaine Lipids in Lower Plants. Biosynthesis of DGTS and DGTA in Ochromonas danica (Chrysophyceae) and the Possible Role of DGTS in Lipid Metabolism, Plant Cell Physiol. 33, 427–436.
Schneider, J.C., and Roessler, P. (1994) Radiolabeling Studies of Lipids and Fatty Acids in Nannochloropsis (Eustigmatophyceae), an Oleaginous Marine Alga, J. Phycol. 30, 594–598.
Stanier, R.Y., Kunisawa, M.M., and Cohen-Bazir, G. (1971) Purification and Properties of Unicellular Blue-Green Algae (Order Chlorococcales), Bacteriol. Rev. 35, 171–201.
Borch, R.F. (1975) Separation of Long Chain Fatty Acids as Phenacylesters by HPLC, Anal. Chem. 47, 2437–2439.
Eichenberger, W., and Gribi, C. (1997) Lipids of Pavlova lutheri: Cellular Site and Metabolic Role of DGCC, Phytochemistry 45, 1561–1567.
Browse, J., and Somerville, C. (1991) Glycerolipid Synthesis: Biochemistry and Regulation, Ann. Rev. Plant Physiol. Plant Mol. Biol. 42, 467–506.
Safford, R., and Nichols, B.W. (1970) Positional Distribution of Fatty Acids in Monogalactosyldiglyceride Fractions from Leaves and Algae, Biochim. Biophys. Acta 210, 57–64.
Khozin, I., and Cohen, Z. (1996) Differential Response of Microalgae to the Substituted Pyridazinone Sandoz 9785 Reveal Different Pathways in the Biosynthesis of Eicosapentaenoic Acid (EPA), Phytochemistry, 42, 1025–1029.
Siljegovich-Hänggi, N., and Eichenberger, W. (1998) Effect of the Substituted Pyridazinone SAN 9785 on the Lipid and Fatty Acid Biosynthesis in Pavlova lutheri (Haptophyceae), in Advances in Plant Lipid Research (Sanchez, J., Cerda-Olmedo, E., and Martinez-Force, E., eds.), pp. 259–261, Universidad de Sevilla, Seville.
Piorreck, M., Baasch, K.H., and Pohl, P. (1984) Biomass Production, Total Protein, Chlorophyll, Lipids and Fatty Acids of Freshwater Green and Blue Algae Under Different Nitrogen Regimes, Phytochemistry 23, 207–216.
Henderson, R.J., and Sargent, J.R. (1989) Lipid Composition and Biosynthesis in Aging Cultures of the Marine Cryptomonad Chroomonas salina, Phytochemistry 28, 1355–1362.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Bigogno, C., Khozin-Goldberg, I., Adlerstein, D. et al. Biosynthesis of arachidonic acid in the oleaginous microalga Parietochloris incisa (Chlorophyceae): Radiolabeling studies. Lipids 37, 209–216 (2002). https://doi.org/10.1007/s11745-002-0882-6
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11745-002-0882-6