Abstract
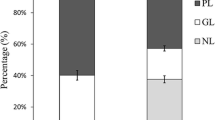
Perkinsus marinus is one of two important protozoan parasites of the eastern oyster, Crassostrea virginica. The other is Haplosporidium nelsoni. Lipids extracted from 7-d-old in vitro cultured P. marinus meronts, incubated with fluorescent-labeled phosphatidylcholine (FL PC) and nonincubated P. marinus meronts, were analyzed by a high-performance liquid chromatography (HPLC) system equipped with a diol phase column, in combination with thin-layer chromatography coupled with a flameionization detector (TLC/FID), and high-performance thin-layer chromatography (HPTLC). Various polar and neutral lipid classes were separated by HPLC using a two-gradient solvent system. Five polar lipid classes—phosphatidylcholine (PC), phosphatidylethanolamine (PE), cardiolipin (CL), sphingomyelin (SM), and phosphatidylserine (PS)—were identified from P. marinus extracts. Four neutral lipid classes—triacylglycerol (TAG), steryl ester (SE), cholesterol (CHO), and fatty alcohol—were distinguished. TLC/FID analysis of meront lipids showed that the weight percentages of PC, PE, CL, SM, PS/PI, TAG, SE, and CHO were 21, 10.7, 4, 2.3, 4.3, 48.7, 7.8, and 1.2%, respectively. HPLC and HPTLC analyses revealed the presence of two SM and PS isomers in P. marinus extracts. Perkinsus marinus effectively incorporated FL PC acquired from the medium and metabolized it to various components (i.e., free fatty acid, monoacylglycerol, diacylglycerol, TAG, PE, and CL). Uptake and interconversion of FL PC in P. marinus meronts increased with time. After 48 h the total uptake of fluorescence (FL) was 28.9% of the FL PC added to the medium, and 43% of the incorporated FL resided in TAG.
Similar content being viewed by others
Abbreviations
- Bodipy:
-
4,4-difluoro-5,7-dimethyl-4-bora-3a,4a-diaza-s-indacene
- CER:
-
ceramide
- CHE:
-
cholesteryl ester
- CHO:
-
cholesterol
- CL:
-
cardiolipin
- DAG:
-
diacylglycerol
- FA:
-
fatty acid
- FAME:
-
fatty acid methyl ester
- FFA:
-
free fatty acids
- FH:
-
fatty alcohol
- FL:
-
fluorescent-labeled
- FTM:
-
fluid thioglycollate medium
- HPLC:
-
high-performance liquid chromatography
- HPTLC:
-
high-performance thin-layer chromatography
- LPC:
-
lysophosphatidylcholine
- MAG:
-
monoacylglycerol
- PC:
-
phosphatidylcholine
- PE:
-
phosphatidylethanolamine
- PG:
-
phosphatidylglycerol
- PI:
-
phosphatidylinositol
- PS:
-
phosphatidylserine
- SE:
-
steryl ester
- SM:
-
sphingomyelin
- TAG:
-
triacylglycerol
- TLC-FID:
-
thin-layer chromatography coupled with a flame-ionization detector
- UV:
-
ultraviolet
- YRW:
-
York river water
References
Levine, N.D. (1978) Perkinsus gen. n. and Other New Taxa in the Protozoan Phylum Apicomplexa, J. Parasitol. 64, 549.
Mackin, J.G., Owen, H.M., and Collier, A. (1950) Preliminary Note on the Occurrence of a New Protistan Parasite, Dermocystidium marinum n. sp., in Crassostrea virginica (Gmelin), Science 111, 328–329.
Chu, F.-L.E. (1996) Laboratory Investigations of Susceptibility, Infectivity and Transmission of Perkinsus marinus in Oysters, J. Shellfish. Res. 15, 57–66.
Perkins, F.O. (1966) Life History Studies of Dermocystidium marinum, an Oyster Pathogen, Ph.D. Thesis, Florida State University, Tallahassee, 273 pp.
Perkins, F.O. (1988) Structure of Protistan Parasites Found in Bivalve Molluscs, Am. Fish. Soc. Spec. Publ. 18, 93–111
La Peyre, J.F., Faisal, M., and Burreson, E.M. (1993) In vitro Propagation of the Protozoan Perkinsus marinus, a Pathogen of the Eastern Oyster, Crassostrea virginica, J. Euk. Microbiol. 40, 304–310.
Gauthier, J.D., and Vasta, G.R. (1993) Continuous in vitro Culture of the Eastern Oyster Parasite Perkinsus marinus, J. Invert. Pathol. 62, 321–323.
Kleinschuster, S.J., and Swink, S.L. (1993) A Simple Method for the in vitro Culture of Perkinsus marinus, Nautilus 107, 76–78
Vial, H.J., and Ancelin, M.-L. (1992) Malaria Lipids, an Overview, in Subcellular Biochemistry, Intracellular Parasites (Avila, J.L., and Harris, J.R., eds.), Vol. 18, pp. 259–306, Plenum Press, New York.
Vial, H.J., and Ancelin, M.-L. (1998) Malaria Lipids, in Malaria: Parasite Biology, Pathogenesis, and Protection (Sherman, I.W., ed.), pp. 159–175, ASM Press, Washington, DC.
Smith, T.N., Brooks, T.J., and Lockard, V.G. (1970) In Vitro Studies on Cholesterol Metabolism in the Blood Fluke Schistosoma mansoni, Lipids 5, 854–856.
Meyer, F., Meyer, H., and Bueding, E. (1970) Lipid Metabolism in the Parasitic and Free-living Flatworms. Schistosoma mansoni and Dugesia dorotocephala, Biochim. Biophys. Acta 210, 257–265.
Furlong, S.T., Thibault, K.S., Morbelli, L.M., Quinn, J.J., and Rogers, R.A. (1995) Uptake and Compartmentalization of Fluorescent Lipid Analogs in Larval Schistosoma mansoni, J. Lipid Res. 36, 1–12.
Brouwers, J.F.H.M., Smeenk, I.M.B., van Golde, L.M.G., and Tielens, A.G.M. (1997) The Incorporation, Modification and Turnover of Fatty Acids in Adult Schistosoma mansoni, Mol. Biochem. Parasitol. 88, 175–185.
Brouwers, J.F.H.M., Van Hellemod, J.J., Van Golde, L.M.G., and Tielens, A.G.M. (1998) Ether Lipids and Their Possible Physiological Function in Adult Schistosoma mansoni, Mol. Biochem. Parasitol. 96, 49–58.
Redman, C.A., Kennington, S., Spathopoulou, T., and Kusel, J.R. (1997) Interconversion of Sphingomyelin and Ceramide in Adult Schistosoma mansoni, Mol. Biochem. Parasitol. 90, 145–153.
Coppens, I., Levade, T., and Courtoy, P.J. (1995) Host Plasma Low Density Lipoproteins Particles as a Essential Source of Lipids for the Bloodstream Forms of Trypanosoma brucei, J. Biol. Chem. 270, 5736–5741.
Lujan, H.D., Mowatt, M.R., and Nash, T.E. (1996) Lipid Requirements and Lipid Uptake by Giardia lamblia Trophozoites in Culture, J. Euk. Microbiol. 43, 237–242.
Ellis, J.E., Wyder, M.A., Jarroll, E.L., and Kaneshiro, E.S. (1996) Changes in Lipid Composition During in vitro Encystation and Fatty Acid Desaturase Activity of Giardia lamblia, Mol. Biochem. Parasitol. 81, 13–25.
Christie, W.W. (1987) High-Performance Liquid Chromatography and Lipids, pp. 87–132, Pergamon Press, Oxford.
Christie, W.W. (1986) Separation of Lipid Classes by High-Performance Liquid Chromatography with “Mass Detector”, J. Chromatog. 361, 396–399.
Breton, L., Serkiz, B., Volland, J.-P., and Lepagnol, J. (1989) A New Rapid Method for Phospholipid Separation by High-Performance Liquid Chromatography with Light-scattering Detection, J. Chromatog. B 497, 243–249.
Lutzke, B.S., and Braughler, J.M. (1990) An Improved Method for the Identification and Quantification of Biological Lipids by HPLC Using Laser Light-Scattering Detection, J. Lipid Res. 31, 2127–2130.
Juaneda, P., Rocquelin, G., and Astorg, P.O. (1990) Separation and Quantification of Heart and Liver Phospholipid Classes by High-Performance Liquid Chromatography Using a New Light-Scattering Detector, Lipids 25, 756–759.
Letter, W.S. (1992) A Rapid Method for Phospholipid Class Separation by HPLC Using an Evaporative Light-Scattering Detector, J. Liq. Chromatogr. 15, 253–266.
Bunger, H., and Pison, U. (1995) Quantitative Analysis of Pulmonary Surfactant Phospholipids by High-Performance Liquid Chromatography and Light-Scattering Detection, J. Chromatogr. B 672, 25–31.
Kiuchi, K., Ohta, T., and Ebine, H. (1975) High-Speed Liquid Chromatography Separation of Glycerides, Fatty Acids and Sterols, J. Chromatogr. Sci. 13, 461–466.
Aitzetmüller, K., and Koch, J. (1978) Liquid Chromatography Analysis of Serum Lipids and Other Lipids of Medical Interest, J. Chromatogr. B 145, 195–202.
Gillan, F.T., and Johns, R.B. (1983) Normal-Phase HPLC Analysis of Microbial Carotenoids and Neutral Lipids, J. Chromatogr. Sci. 21, 34–39.
Palmer, D.N., Anderson, M.A., and Jolly, R.D. (1984) Separation of Some Neutral Lipids by Normal-Phase High-Performance Liquid Chromatography on a Cyanopropyl Column: Ubiquinone, Dolichol, and Cholesterol Levels in Sheep Liver, Anal. Biochem. 140, 315–319.
Lapin, B.P., Pisareva, N.A., Rubtsova, T.E., and Jakevich, M.L. (1986) Separation of Lipophilic Fractions by High-Performance Liquid Chromatography, J. Chromatogr. 365, 229–235.
Carunchio, V., Nicoletti, I., Frezza, L., and Sinibaldi, M. (1984) High-Performance Liquid Chromatography Separation of Phospholipids on Chemically Bonded Silica Gel Part 1, Ann. Chim. (Rome) 74, 331–339.
Mallet, A.I., Cunningham, F.M., and Daniel, R. (1984) Rapid Isocratic High-Performance Liquid Chromatographic Purification of Platelet Activating Factor (PAF) and 1 Lyso-PAF from Human Skin, J. Chromatogr. B 309, 160–164.
Andrews, A.G. (1984) Estimation of Amniotic Fluid Phospholipids by High-Performance Liquid Chromatography, J. Chromatogr. B 336, 139–150.
Kuhnz, W., Zimmermann, B., and Nau, H. (1985) Improved Separation of Phospholipids by High-Performance Liquid Chromatography, J. Chromatogr. B 344, 309–312.
Soudant, P., Marty, Y., Moal, J., and Samain, J.-F. (1995) Separation of Major Polar Lipids in Pecten maximus by High-Performance Liquid Chromatography and Subsequent Determination of Their Fatty Acids Using Gas Chromatography, J. Chromatogr. B 673, 15–26.
Silversand, C., and Haux, C. (1997) Improved High-Performance Liquid Chromatographic Method for the Separation and Quantification of Lipid Classes: Application to Fish Lipids, J. Chromatogr. B 703, 7–14.
Schlager, S.I., and Jordi, H. (1981) Separation of Cellular Phospholipid, Neutral Lipid and Cholesterol by High-Pressure Liquid Chromatography, Biochim. Biophys. Acta 665, 355–358.
Yandrasitz, J.R., Berry, G., and Segal, S. (1981) High-Performance Liquid Chromatography of Phospholipids with UV Detection: Optimization of Separations on Silica, J. Chromatogr. B 225, 319–328.
Dugan, L.L., Demediuk, L., Pendley, C.E. II, and Horrocks, L.A., (1986) Separation of Phospholipids by High-Performance Liquid Chromatography: All Major Classes, Including Ethanolamine and Choline Plasmalogens, and Most Minor Classes, Including Lysophosphatidylethanolamine. J. Chromatogr. B 378, 317–327.
Juaneda, P., and Rocquelin, G. (1986) Complete Separation of Phospholipids from Human Heart Combining two HPLC Methods, Lipids 21, 239–240.
Wiley, M.G., Pretakiewicz, M., Takahashi, M., and Lowenstein, J.M. (1992) An Extended Method for Separating and Quantitating Molecular Species of Phospholipids, Lipids 27, 295–301.
Chu, F.-L.E., Soudant, P., Volety, A.K., and Huang, Y. (2000) Perkinsus marinus: Uptake and Interconversion of Fluorescent Lipid Analogs in the Parasite of the Oyster, Crassostrea virginica, Exp. Parasitol. 95, 240–251.
Bligh, E.G., and Dyer, W.J. (1959) A Rapid Method of Total Lipid Extraction and Purification, Can. J. Biochem. Physiol. 37, 911–917.
Metcalfe, L.D., and Schmitz, A.A. (1961) The Rapid Preparation of Fatty Acid Esters for Gas Chromatography Analysis, Anal. Chem. 33, 363–364.
Marty, Y., Delaunay, F., Moal, J., and Samain, J.F. (1992) Change in the Fatty Acid Composition of Pecten maximus (L.), J. Exp. Mar. Biol. Ecol. 163, 221–234.
Chu, F.-L.E., and Ozkizilcik, S. (1995) Lipid and Fatty Acid Composition of Striped Bass (Morone saxatilis) Larvae During Development, Comp. Biochem. Physiol. 111B, 665–674.
Olsen, R.E., and Henderson, R.J. (1989) The Rapid Analysis of Neutral and Polar Marine Lipids using Double Development HPTLC and Scanning Densitometry, J. Exp. Mar. Biol. Ecol. 129, 189–197.
Christie, W.W. (1982) Lipid Analysis, 2nd edn., pp. 107–134, Pergamon Press, Oxford.
Sherman, I.W. (1979) Biochemistry of Plasmodium (malaria parasites), Microbiol. Rev. 43, 453–495.
Beach, D.H., Holz, G.G., Jr., and Anekwe, G.E. (1979) Lipid of Leishmania Promastigotes, J. Parasitol. 65, 203–216.
Smith, J.D. (1993) Phospholipid Biosynthesis in Protozoa, Prog. Lipid Res. 32, 47–60.
Gurr, M.T., and Harwood, J.L. (1991) Lipid Biochemistry, An Introduction, 4th edn., pp. 295–337, Chapman and Hall, New York.
Mitschler, R.R., Welti, R., and Upton, S.J. (1994) A Comparative Study of Lipid Composition of Cryptosporidium parvum (Apicomplexan) and Madin-Darby Bovine Kidneys Cells, J. Euk. Microbiol. 41, 8–12.
Stevens, T.L., Gibson, G.R., Adam, R., Maier, J., Allison-Ennis, M., and Das, S. (1997) Uptake and Cellular Localization of Exogenous Lipids by Giardia lamblia, a Primitive Eukaryote, Exp. Parasitol. 86, 133–143
Dixon, H., and Williamson, J. (1970) The Lipid Composition of Blood and Culture Forms of Trypanosoma lewisi and Trypanosoma rhodesiense Compared with That of Their Environment, Comp. Biochem. Physiol. 33, 111–128.
Vial, H.J., Thuet, M.J., and Philippot, J.R. (1982) Phospholipid Biosynthesis in Synchronous Plasmodium falciparum Cultures, J. Protozool. 29, 258–263.
Vial, H.J., Ancelin, M.-L., Thuet, M.J., and Philippot, J.R. (1989) Phospholipid Metabolism in Plasmodium-Infected Erythrocytes: Guidelines for Further Studies Using Radioactive Precursor Incorporations, Parasitology 98, 351–357.
Kasurinen, J. (1992) A Novel Fluorescent Fatty Acid, 5-Methyl-BDY-dodecanoic Acid, Is a Potential Probe in Lipid Transport Studies by Incorporating Selectively to Lipids of BHK cells, Biochem. Biophys. Res. Comm. 187, 1594–1601.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Soudant, P., Chu, FL.E. & Marty, Y. Lipid class composition of the protozoan Perkinsus marinus, an oyster parasite, and its metabolism of a fluorescent phosphatidylcholine analog. Lipids 35, 1387–1396 (2000). https://doi.org/10.1007/s11745-000-0656-1
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11745-000-0656-1