Abstract
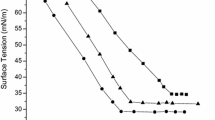
A series of three cationic surfactants with different chain lengths, i.e. O-decyl-, O-dodecyl- and O-tetradecyl-N,N′-diisopropylisourea hydrochloride were synthesized. The chemical structures of the prepared compounds were confirmed using chemical analysis, Fourier transform infra red spectroscopy and nuclear magnetic resonance spectra and chromatography coupled with mass spectrometry. Several surface properties of the synthesized surfactants were studied in aqueous solution. Surface tension, conductivity and the foaming power were measured and the critical micelle concentration (CMC) was determined at 25 °C. These results were correlated to the surfactant chemical structure. The surfactant that provided the best surface activity was the O-dodecyl-N,N′-diisopropylisourea hydrochloride. The CMC was found to be significantly dependent on the hydrophobic chain length and decreased with increasing of the carbon number in the alkyl chain. The surfactants synthesized have an interesting foaming power. The surfactant with the longest alkyl chain exhibited the highest foamability.




Similar content being viewed by others
References
Chlebicki J, Wegrzynska J, Maliszewska I, Oswiecimska M (2005) Preparation, surface-active properties, and antimicrobial activities of bis-quaternary ammonium salts from amines and epichlorohydrin. J Surfactants Deterg 8:227–232
Hafiz AA, Badawi AM, El-Deeb FI, Soliman EA, El-Awady MY, Mohamed DE (2009) Ferrocene-based cationic surfactants: surface and antimicrobial properties. J Surfactants Deterg 13:165–172
Kozak M, Domka L, Jurga S (2007) Interactions of cationic surfactants with DPPC. J Therm Anal Calorim 88:395–399
Mc Carron PA, Donnelly RF, Marouf W, Calvert DE (2007) Anti-adherent and antifungal activities of surfactant-coated poly(ethylcyanoacrylate) nanoparticles. Int J Pharm 340:182–190
Caillier L, de Givenchy TE, Levy R, Vandenberghe Y, Géribaldi S, Guittard F (2009) Synthesis and antimicrobial properties of polymerizable quaternary ammoniums. Eur J Med Chem 44:3201–3208
Pérez L, Pinazo A, Garcıa MT, Lozano M, Manresa A, Angelet M, Vinardell MP, Mitjans M, Pons R, Infante MR (2009) Cationic surfactants from lysine: synthesis, micellization and biological evaluation. Eur J Med Chem 44:1884–1892
Asefi D, Arami M, Mahmoodi NM (2010) Electrochemical effect of cationic gemini surfactant and halide salts on corrosion inhibition of low carbon steel in acid medium. Corros Sci 52:794–800
Deyab MA (2007) Effect of cationic surfactant and inorganic anions on the electrochemical behavior of carbon steel in formation water. Corros Sci 49:2315–2328
Asefi D, Arami M, Sarabi AA, Mahmoodi NM (2009) The chain length influence of cationic surfactant and role of nonionic co-surfactants on controlling the corrosion rate of steel in acidic media. Corros Sci 51:1817–1821
Wang X, Yang H, Wang F (2010) A cationic gemini-surfactant as effective inhibitor for mild steel in HCl solutions. Corros Sci 52:1268–1276
Jada A, Florentin C, Mariotti S (2004) Study of the electrical properties of cationic bitumen emulsions by microelectrophoresis. Adv Colloid Interface Sci 108–109:127–132
Chappat M (1994) Some applications of emulsions. Colloids Surf A 91:57–77
Dopierala K, Prochaska K (2008) The effect of molecular structure on the surface properties of selected quaternary ammonium salts. J Colloid Interface Sci 321:220–226
Mata J, Varade D, Bahadur P (2005) Aggregation behavior of quaternary salt based cationic surfactants. Thermochim Acta 428:147–155
Hac-Wydro K, Wydro P, Dynarowicz-Latka P (2004) A study of the interaction between dialkyldimethylammonium bromides and tri-n-octylphosphine oxide (topo) in mixed monolayers at the air/water interface. J Colloid Interface Sci 278:206–214
Shimizu S, El Seoud OA (2003) 2-(Acylaminoethyl)trimethylammonium chloride surfactants: synthesis and properties of aqueous solutions. Colloid Polym Sci 282:21–31
Badache L, Bauduin G, Boutevin B, Rahal S (1998) Synthèse et propriétés de O-fluoro alkyl N,N′-dialkylisourées. J Fluor Chem 92:53–58
Badache L, Bauduin G, Djadi D, Rahal S (2002) Synthèse et application d’une série d’isourées fluorées dans le domaine de la corrosion. J Soc Alger Chem 12:63–70
Badache L, Boschet F, Lehanine Z, Boutevin B, Ameduri B (2011) Synthesis and surface properties of a series of surfactants based on O-alkyl and O-perfluoro-N,N′-diisopropylisoureas. J Fluor Chem 132:382–388
Lehanine Z, Badache L (2011) Thermodynamics of micellization of cationic surfactants based on O-alkyl and O-perfluoro-N,N′-diisopropylisourea. J Chem Thermodyn 43:1342–1348
Mathias LJ (1979) Esterification and alkylation reactions employing isoureas. Synthesis Stuttgart 561–576
Bakibaev AA, Shtrykova VV (1995) Isoureas: synthesis, properties, and applications. Russ Chem Rev 64:929–938
Ulrich H (2007) Chemistry and technology of carbodiimides. Wiley, Chichester, UK
Lamoureux G, Aguero C (2009) A comparison of several modern alkylating agents. Special Issue Reviews and accounts, Arkivoc, pp 251–264
Chighine A, Crosignani S, Arnal MC, Bradley M, Linclau B (2009) Microwave-assisted ester formation using O-alkylisoureas: a convenient method for the synthesis of esters with inversion of configuration. J Org Chem 74:4753–4762
Crosignani S, White PD, Linclau B (2004) Polymer-supported O-alkylisoureas: useful reagents for O-alkylation of carboxylic acids. J Org Chem 69:5897–5905
Rosen MJ (2004) Surfactants and interfacial phenomena, 3rd edn. Wiley, New York
Tanford C (1980) The hydrophobic effect-formation of micelles and biological membranes, 2nd edn. Wiley, New York
Mukerjee P, Mysels KJ (1971) Critical micelle concentrations of aqueous surfactant systems. NSRDS, Washington
Mukerjee P (1967) The nature of the association equilibria and hydrophobic bonding in aqueous solution of association. Adv Colloid Interface Sci 1:241–275
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Badache, L., Lehanine, Z. & Naït Abderrahmane, W. Synthesis and Surface Properties Study of a Series of Cationic Surfactants with Different Hydrophobic Chain Lengths. J Surfact Deterg 15, 715–720 (2012). https://doi.org/10.1007/s11743-012-1351-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11743-012-1351-2