Abstract
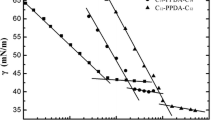
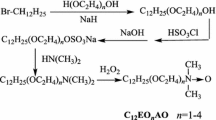
A series of gemini n-alkylphenol polyoxyethylene surfactants (GAP) were successfully synthesized and their molecular structure were confirmed by NMR and FTIR spectrum. Using the same synthesis route, a gemini nonylphenol polyoxyethylene surfactant (GNP) was synthesized using an industrial nonylphenol product and paraformaldehyde, and its molecular structure was also characterized by 1H-NMR and FTIR spectra. The optimal reaction conditions were established. The critical micelle concentration (CMC) values of GAP were determined by means of Wilhelmy plate method and steady-state fluorescence probe method. The experimental results show how the lengths of the hydrophilic polyoxyethylene chain and the hydrophobic tail alter the CMC values. The CMC values of the GAP are found to be much lower than those of corresponding conventional single tail nonionic surfactants of the polyethoxylated alkylphenol type, which indicates that the gemini species exhibit a better surface activity.




Similar content being viewed by others
References
FitzGerald PA, Carr MW, Davey TW, Serelis AK, Such CH, Warr GG (2004) Preparation and dilute solution properties of model gemini nonionic surfactants. J Colloid Interface Sci 275:649–658
Zhou TT, Yang H, Xu XH, Wang XY, Wang JB, Dong GJ (2008) Synthesis, surface and aggregation properties of nonionic poly(ethylene oxide) gemini surfactants. Colloids Surf A 317:339–343
Matsubara H, Eguchi T, Takumi H, Tsuchiya K, Takiue T, Aratono M (2009) Surface adsorption and aggregate formation of cationic gemini surfactant and long-chain alcohol mixtures. J Phys Chem B 113(26):8847–8853
Yoshimura T, Sakato A, Tsuchiya K, Ohkubo T, Sakai H, Abe M, Esumi K (2007) Adsorption and aggregation properties of amino acid-based N-alkyl cysteine monomeric and N, N′-dialkyl cystine gemini surfactants. J Colloid Interface Sci 308:466–473
Zhu S, Cheng F, Wang J, Yu JG (2006) Anionic gemini surfactants: synthesis and aggregation properties in aqueous solutions. Colloids Surf A 281:35–39
Rosen MJ, Mathias JH, Davenport LH (1999) Aberrant aggregation behavior in cationic gemini surfactants investigated by surface tension, interfacial tension, and fluorescence methods. Langmuir 15:7340–7346
De S, Aswal VK, Goyal PS, Bhattacharya S (1996) Role of spacer chain length in dimeric micellar organization. Small angle neutron scattering and fluorescence studies. J Phys Chem 100:11664–11671
Aswal DS, Goyal VK, Bhattacharya PS (1998) Novel gemini micelles from dimeric surfactants with oxyethylene spacer chain. Small angle neutron scattering and fluorescence studies. J Phys Chem B 102:6152–6160
Pinazo A, Wen X, Peırez L, Infante MR, Franses EI (1999) Aggregation behavior in water of monomeric and gemini cationic surfactants derived from arginine. Langmuir 15(9):3134–3142
Danino D, Talmon Y, Levy H, Beinert G, Zana R (1995) Branched threadlike micelles in an aqueous solution of a trimeric surfactant. Science 269:1420–1421
In M, Bec V, Chariol OA, Zana R (2000) Quaternary ammonium bromide surfactant oligomers in aqueous solution: self-association and microstructure. Langmuir 16:141–148
Bai GY, Yan HK, Thomas RK (2001) Microcalorimetric studies on the thermodynamic properties of cationic gemini surfactants. Langmuir 17:4501–4504
Rosen MJ, Song LD (1996) Dynamic surface tension of aqueous surfactant solutions. 8. Effect of spacer on dynamic properties of gemini surfactant solutions. J Colloid Interface Sci 179:261–268
Baumann HP, Switzerland E (1995) Organic compounds. U.S. Patent 5,470,355
Tracy DJ, Li RX (1998) Novel nonylphenol nonionic gemini surfactants. W.O. Patent 98/37062
Kalyanasundaram K, Thomas JK (1977) Environmental effects on vibronic band intensities in pyrene monomer fluorescence and their application in studies of micellar systems. J Am Chem Soc 99:2039–2044
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Yang, F., Li, G., Xu, N. et al. Synthesis and Critical Micelle Concentration of a Series of Gemini Alkylphenol Polyoxyethylene Nonionic Surfactants. J Surfact Deterg 14, 339–345 (2011). https://doi.org/10.1007/s11743-011-1245-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11743-011-1245-8