Abstract
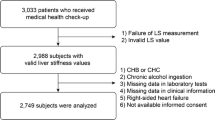
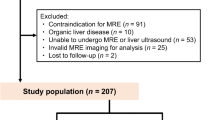
Patients with non-advanced non-alcoholic fatty liver disease (NAFLD) have an increased cardiovascular risk. The present study was designed to evaluate the relationship between liver stiffness measurement (LSM) by transient elastography (TE) and myocardial deformation indices of all cardiac chambers in NAFLD patients without overt heart disease. All consecutive NAFLD patients diagnosed with LSM < 12.5 kPa on TE between September 2021 and December 2021 entered the study. All participants underwent blood tests, TE and two-dimensional (2D) transthoracic echocardiography (TTE) implemented with speckle-tracking echocardiography (STE) analysis of left ventricular (LV) global longitudinal strain (GLS), global circumferential strain (GCS) and global radial strain (GRS), right ventricular (RV) GLS, left atrial (LA) total global strain (TGSA) and right atrial (RA) TGSA. Main independent predictors of impaired LV-GLS (defined as absolute value less negative than − 20%) were evaluated. A total of 92 NAFLD patients (54.0 ± 11.1 years, 50% males) were prospectively analyzed. Mean LSM was 6.2 ± 2.4 kPa. Fibroscan results revealed that 76.1% of patients had F0-F1, 5.4% F2 and 18.5% F3 liver fibrosis. Despite normal biventricular systolic function on 2D-TTE, LV-GLS, LV-GCS and LV-GRS, RV-GLS, LA-TGSA and RA-TGSA were reduced in 64.1%, 38.0%, 38.0%, 31.5%, 39.1% and 41.3% of patients, respectively. Body mass index (BMI) (OR 1.76, 95% CI 1.18–2.64), neutrophil-to-lymphocyte ratio (NLR) (OR 4.93, 95% CI 1.15–31.8) and LSM (OR 9.26, 95% CI 2.24–38.3) were independently associated to impaired LV-GLS. BMI ≥ 29.3 kg/m2, NLR ≥ 1.8 and LSM ≥ 5.5 kPa were the best cut-off values for detecting outcome. LSM ≥ 5.5 kPa identifies NAFLD patients with subclinical myocardial dysfunction.


Similar content being viewed by others
Abbreviations
- 2D:
-
Two-dimensional
- ALP:
-
Alkaline phosphatase
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- BMI:
-
Body mass index
- CAP:
-
Controlled attenuation parameter
- CI:
-
Confidence interval
- CO:
-
Cardiac output
- DBP:
-
Diastolic blood pressure
- EaI:
-
Arterial elastance index
- EAT:
-
Epicardial adipose tissue
- EesI:
-
End-systolic elastance index
- eGFR:
-
Estimated glomerular filtration rate
- ESP:
-
End-systolic pressure
- GCS:
-
Global circumferential strain
- GGT:
-
Gamma-glutamyl transferase
- GLS:
-
Global longitudinal strain
- GRS:
-
Global radial strain
- GSA + :
-
Positive global atrial strain
- GSA-:
-
Negative global atrial strain
- HDL:
-
High-density lipoprotein
- HFpEF:
-
Heart failure with preserved ejection fraction
- ICC:
-
Intraclass correlation coefficient
- INR:
-
International normalized ratio
- IQR:
-
Interquartile range
- IVC:
-
Inferior vena cava
- LA:
-
Left atrial
- LAVi:
-
Left atrial volume index
- LDL:
-
Low-density lipoprotein
- LV:
-
Left ventricular
- LVFP:
-
Left ventricular fillimng pressure
- LSM:
-
Liver stiffness measurement
- LVEDVi:
-
Left ventricular end-diastolic volume index
- LVESVi:
-
Left ventricular end-systolic volume index
- LVEF:
-
Left ventricular ejection fraction
- LVMi:
-
Left ventricular mass index
- LVOT:
-
Left ventricular outflow tract
- MACE:
-
Major adverse cardiovascular events
- MAP:
-
Mean arterial pressure
- MAPSE:
-
Mitral annular plane systolic excursion
- NAFLD:
-
Non-alcoholic fatty liver disease
- NLR:
-
Neutrophil-to-lymphocyte ratio
- NPV:
-
Negative predictive value
- OR:
-
Odds ratio
- PPV:
-
Positive predictive value
- RA:
-
Right atrial
- RDW:
-
Red cell distribution width
- ROC:
-
Receiver operating characteristics
- RV:
-
Right ventricular
- RWT:
-
Relative wall thickness
- SBP:
-
Systolic blood pressure
- SPAP:
-
Systolic pulmonary artery pressure
- STE:
-
Speckle-tracking echocardiography
- SVi:
-
Stroke volume index
- TAPSE:
-
Tricuspid annular plane systolic excursion
- TE:
-
Transient elastography
- TGSA:
-
Total global atrial strain
- TPRi:
-
Total peripheral resistance index
- TTE:
-
Transthoracic echocardiography
- VAC:
-
Ventricular arterial coupling
References
Younossi Z, Koenig A, Abdelatif D et al (2016) Peak epidemiology of nonalcoholic fatty liver disease-meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology 64:73–84
Temple JL, Cordero P, Li J, Nguyen V, Oben JA (2016) A guide to non-alcoholic fatty liver disease in childhood and adolescence. Int J Mol Sci 17:947
Kim D, Choi SY, Park EH et al (2012) Nonalcoholic fatty liver disease is associated with coronary artery calcification. Hepatology 56:605–613
VanWagner LB, Ning H, Lewis CE et al (2014) Associations between nonalcoholic fatty liver disease and subclinical atherosclerosis in middle-aged adults: the Coronary Artery Risk Development in Young Adults Study. Atherosclerosis 235:599–605
Salah HM, Pandey A, Soloveva A et al (2021) Relationship of nonalcoholic fatty liver disease and heart failure with preserved ejection fraction. JACC Basic Transl Sci 6:918–932
Wong VW, Vergniol J, Grace Lai-Hung Wong GL et al (2010) Diagnosis of fibrosis and cirrhosis using liver stiffness measurement in nonalcoholic fatty liver disease. Hepatology 51(454):462
Petta S, Sebastiani G, Viganò M et al (2021) Monitoring occurrence of liver-related events and survival by transient elastography in patients with nonalcoholic fatty liver disease and compensated advanced chronic liver disease. Clin Gastroenterol Hepatol 19:806–815
Konstam MA, Abboud FM (2017) Ejection fraction: misunderstood and overrated (changing the paradigm in categorizing heart failure). Circulation 135:717–719
Potter E, Marwick TH (2018) Assessment of left ventricular function by echocardiography: the case for routinely adding global longitudinal strain to ejection fraction. JACC Cardiovasc Imaging 11:260–274
Sonaglioni A, Lonati C, Lombardo M et al (2019) Incremental prognostic value of global left atrial peak strain in women with new-onset gestational hypertension. J Hypertens 37:1668–1675
Sonaglioni A, Nicolosi GL, Granato A et al (2021) Reduced myocardial strain parameters in subjects with pectus excavatum: impaired myocardial function or methodological limitations due to chest deformity? Semin Thorac Cardiovasc Surg 33:251–262
Sonaglioni A, Nicolosi GL, Lombardo M et al (2021) Influence of chest conformation on myocardial strain parameters in healthy subjects with mitral valve prolapse. Int J Cardiovasc Imaging 37:1009–1022
Karabay CY, Kocabay G, Kalayci A et al (2014) Impaired left ventricular mechanics in nonalcoholic fatty liver disease: a speckle-tracking echocardiography study. Eur J Gastroenterol Hepatol 26:325–331
Baktır AO, Şarlı B, Altekin RE et al (2015) Non alcoholic steatohepatitis is associated with subclinical impairment in left ventricular function measured by speckle tracking echocardiography. Anatol J Cardiol 15:137–142
VanWagner LB, Wilcox JE, Colangelo LA et al (2015) Association of nonalcoholic fatty liver disease with subclinical myocardial remodeling and dysfunction: a population-based study. Hepatology 62:773–783
Zamirian MMD, Samiee EMD, Moaref AMD et al (2018) Assessment of subclinical myocardial changes in non-alcoholic fatty liver disease: a case-control study using speckle tracking echocardiography. Iran J Med Sci 43:466–472
VanWagner LB, Wilcox JE, Ning H et al (2020) Longitudinal association of non-alcoholic fatty liver disease with changes in myocardial structure and function: the CARDIA study. J Am Heart Assoc 9:e014279
Kocabay G, Karabay CY, Colak Y et al (2014) Left atrial deformation parameters in patients with non-alcoholic fatty liver disease: a 2D speckle tracking imaging study. Clin Sci (Lond) 126:297–304
Chang W, Wang Y, Sun L et al (2019) Evaluation of left atrial function in type 2 diabetes mellitus patients with nonalcoholic fatty liver disease by two-dimensional speckle tracking echocardiography. Echocardiography 36:1290–1297
Parvanescu T, Vitel A, Sporea I et al (2021) Significant association between left ventricular diastolic dysfunction, left atrial performance and liver stiffness in patients with metabolic syndrome and non-alcoholic fatty liver disease. Diabetes Metab Syndr Obes 14:1535–1545
Sunbul M, Kivrak T, Durmus E et al (2015) Nonalcoholic steatohepatitis score is an independent predictor of right ventricular dysfunction in patients with nonalcoholic fatty liver disease. Cardiovasc Ther 33:294–299
Galderisi M, Cosyns B, Edvardsen T et al (2017) Standardization of adult transthoracic echocardiography reporting in agreement with recent chamber quantification, diastolic function, and heart valve disease recommendations: an expert consensus document of the European Association of Cardiovascular Imaging. Eur Heart J Cardiovasc Imaging 18:1301–1310
Chalasani N, Younossi Z, Lavine JE et al (2018) The diagnosis and management of nonalcoholic fatty liver disease: practice guidance from the American Association for the Study of liver diseases. Hepatology 67:328–357
Degos F, Perez P, Roche B et al (2010) Diagnostic accuracy of FibroScan and comparison to liver fibrosis biomarkers in chronic viral hepatitis: a multicenter prospective study (the FIBROSTIC study). J Hepatol 53:1013–1021
Zarski JP, Sturm N, Guechot J et al (2012) Comparison of nine blood tests and transient elastography for liver fibrosis in chronic hepatitis C: the ANRS HCEP-23 study. J Hepatol 56:55–62
Levey AS, Bosch JP, Lewis JB et al (1999) A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 130:461–470
Castera L, Forns X, Alberti A (2008) Non-invasive evaluation of liver fibrosis using transient elastography. J Hepatol 48:835–847
Karlas T, Petroff D, Sasso M et al (2017) Individual patient data meta-analysis of controlled attenuation parameter (CAP) technology for assessing steatosis. J Hepatol 66:1022–1030
Lim JK, Flamm SL, Singh S et al (2017) American gastroenterological association institute guideline on the role of elastography in the evaluation of liver fibrosis. Gastroenterology 152:1536–1543
Wu S, Yang Z, Zhou J et al (2019) Systematic review: diagnostic accuracy of non-invasive tests for staging liver fibrosis in autoimmune hepatitis. Hepatol Int 13:91–101
Wong VW, Irles M, Wong GL et al (2019) Unified interpretation of liver stiffness measurement by M and XL probes in non-alcoholic fatty liver disease. Gut 68:2057–2064
Lang RM, Badano LP, Mor-Avi V et al (2015) Recommendations for cardiac chamber quantification by echocardiography in adults: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 28:1–39
Nagueh SF, Smiseth OA, Appleton CP et al (2016) Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr 29:277–314
Rudski LG, Lai WW, Afilalo J et al (2010) Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography endorsed by the European Association of Echocardiography, a registered branch of the European Society of Cardiology, and the Canadian Society of Echocardiography. J Am Soc Echocardiogr 23:685–713
Nishimura RA, Otto CM, Bonow RO et al (2017) 2017 AHA/ACC focused update of the 2014 AHA/ACC guideline for the management of patients with valvular heart disease: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation 135:e1159–e1195
Iacobellis G, Willens HJ, Barbaro G, Sharma AM (2008) Threshold values of high-risk echocardiographic epicardial fat thickness. Obesity (Silver Spring) 16:887–892
Redfield MM, Jacobsen SJ, Borlaug BA et al (2005) Age- and gender-related ventricular-vascular stiffening: a community-based study. Circulation 112:2254–2262
Sunagawa K, Maughan WL, Burkhoff D, Sagawa K (1983) Left ventricular interaction with arterial load studied in isolated canine ventricle. Am J Physiol 245:H773–H780
Chantler PD, Lakatta EG (1985) Najjar SS (2008) Arterial-ventricular coupling: mechanistic insights into cardiovascular performance at rest and during exercise. J Appl Physiol 105:1342–1351
Hill LK, Sollers Iii JJ, Thayer JF (2013) Resistance reconstructed estimation of total peripheral resistance from computationally derived cardiac output – biomed 2013. Biomed Sci Instrum 49:216–223
Muraru D, Onciul S, Peluso D et al (2016) Sex- and method-specific reference values for right ventricular strain by 2-dimensional speckle-tracking echocardiography. Circ Cardiovasc Imaging 9:e003866
Sonaglioni A, Vincenti A, Baravelli M et al (2019) Prognostic value of global left atrial peak strain in patients with acute ischemic stroke and no evidence of atrial fibrillation. Int J Cardiovasc Imaging 35:603–613
Sugimoto T, Dulgheru R, Bernard A et al (2017) Echocardiographic reference ranges for normal left ventricular 2D strain: results from the EACVI NORRE study. Eur Heart J Cardiovasc Imaging 18:833–840
Wang TKM, Grimm RA, Rodriguez LL et al (2021) Defining the reference range for right ventricular systolic strain by echocardiography in healthy subjects: a meta-analysis. PLoS ONE 16:e0256547
Yingchoncharoen T, Agarwal S, Popović ZB, Marwick TH (2013) Normal ranges of left ventricular strain: a meta-analysis. J Am Soc Echocardiogr 26:185–191
Sugimoto T, Robinet S, Dulgheru R et al (2018) Echocardiographic reference ranges for normal left atrial function parameters: results from the EACVI NORRE study. Eur Heart J Cardiovasc Imaging 19:630–638
Brand A, Bathe M, Hübscher A et al (2018) Normative reference data, determinants, and clinical implications of right atrial reservoir function in women assessed by 2D speckle-tracking echocardiography. Echocardiography 35:1542–1549
Suto M, Tanaka H, Mochizuki Y et al (2017) Impact of overweight on left ventricular function in type 2 diabetes mellitus. Cardiovasc Diabetol 16:145
Ng ACT, Prevedello F, Dolci G et al (2018) Impact of diabetes and increasing body mass index category on left ventricular systolic and diastolic function. J Am Soc Echocardiogr 31:916–925
Kishi S, Armstrong AC, Gidding SS et al (2014) Association of obesity in early adulthood and middle age with incipient left ventricular dysfunction and structural remodeling: the CARDIA study (Coronary Artery Risk Development in Young Adults). JACC Heart Fail 2:500–508
Wang YC, Liang CS, Gopal DM et al (2015) Preclinical systolic and diastolic dysfunctions in metabolically healthy and unhealthy obese individuals. Circ Heart Fail 8:897–904
Dini FL, Fabiani I, Miccoli M et al (2018) Prevalence and determinants of left ventricular diastolic dysfunction in obese subjects and the role of left ventricular global longitudinal strain and mass normalized to height. Echocardiography 35:1124–1131
Blomstrand P, Sjöblom P, Nilsson M et al (2018) Overweight and obesity impair left ventricular systolic function as measured by left ventricular ejection fraction and global longitudinal strain. Cardiovasc Diabetol 17:113
Hoang K, Zhao Y, Gardin JM et al (2015) LV mass as a predictor of CVD events in older adults with and without metabolic syndrome and diabetes. JACC Cardiovasc Imaging 8:1007–1015
Lee HJ, Kim HL, Lim WH et al (2019) Subclinical alterations in left ventricular structure and function according to obesity and metabolic health status. PLoS ONE 14:e0222118
Alaaeddine N, Sidaoui J, Hilal G et al (2012) TNF-α messenger ribonucleic acid (mRNA) in patients with nonalcoholic steatohepatitis. Eur Cytokine Netw 23:107–111
Leite NC, Salles GF, Cardoso CR, Villela-Nogueira CA (2013) Serum biomarkers in type 2 diabetic patients with non-alcoholic steatohepatitis and advanced fibrosis. Hepatol Res 43:508–515
Yilmaz H, Yalcin KS, Namuslu M et al (2015) Neutrophil-lymphocyte ratio (NLR) could be better predictor than C-reactive protein (CRP) for liver fibrosis in non-alcoholic steatohepatitis (NASH). Ann Clin Lab Sci 45:278–286
Abdel-Razik A, Mousa N, Shabana W et al (2016) A novel model using mean platelet volume and neutrophil to lymphocyte ratio as a marker of nonalcoholic steatohepatitis in NAFLD patients: multicentric study. Eur J Gastroenterol Hepatol 28:e1-9
Alkhouri N, Tamimi TA, Yerian L et al (2010) The inflamed liver and atherosclerosis: a link between histologic severity of nonalcoholic fatty liver disease and increased cardiovascular risk. Dig Dis Sci 55:2644–2650
Russo C, Sera F, Jin Z et al (2016) Abdominal adiposity, general obesity, and subclinical systolic dysfunction in the elderly: a population-based cohort study. Eur J Heart Fail 18:537–544
Perseghin G (2011) Lipids in the wrong place: visceral fat and nonalcoholic steatohepatitis. Diabetes Care 34:S367–S370
Mazurek T, Zhang L, Zalewski A et al (2003) Human epicardial adipose tissue is a source of inflammatory mediators. Circulation 108:2460–2466
Rao VN, Fudim M, Mentz RJ et al (2020) Regional adiposity and heart failure with preserved ejection fraction. Eur J Heart Fail 22:1540–1550
Khoury T, Mari A, Nseir W et al (2019) Neutrophil-to-lymphocyte ratio is independently associated with inflammatory activity and fibrosis grade in nonalcoholic fatty liver disease. Eur J Gastroenterol Hepatol 31:1110–1115
Spengler EK, Loomba R (2015) Recommendations for diagnosis, referral for liver biopsy, and treatment of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Mayo Clin Proc 90:1233–1246
Funding
This work has been supported by Italian Ministry of Health Ricerca Corrente—IRCCS MultiMedica.
Author information
Authors and Affiliations
Contributions
AS: conceptualization; data curation; investigation; methodology; software; visualization; writing—original draft. FC, AC, LA, ER: conceptualization; data curation; methodology. GLN, ML, MGR, MV: conceptualization; supervision; validation; writing—review and editing.
Corresponding author
Ethics declarations
Conflict of interest
We wish to confirm that there are no conflicts of interest associated with this publication. Andrea Sonaglioni declares that he has no conflict of interest. Federica Cerini declares that she has no conflict of interest. Antonio Cerrone declares that he has no conflict of interest. Lorenzo Argiento declares that he has no conflict of interest. Gian Luigi Nicolosi declares that he has no conflict of interest. Elisabetta Rigamonti declares that she has no conflict of interest. Michele Lombardo declares that he has no conflict of interest. Maria Grazia Rumi declares that she has no conflict of interest. Mauro Viganò declares that he has no conflict of interest.
Ethical approval
All procedures were performed according to the ethical standards of the institutional research committee and to the Declaration of Helsinki (1964) and its subsequent amendments or equivalent ethical standards. A written and informed consent was obtained from each participant and the study protocol was authorized the local Ethics Committee.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Human and animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sonaglioni, A., Cerini, F., Cerrone, A. et al. Liver stiffness measurement identifies subclinical myocardial dysfunction in non-advanced non-alcoholic fatty liver disease patients without overt heart disease. Intern Emerg Med 17, 1425–1438 (2022). https://doi.org/10.1007/s11739-022-02966-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-022-02966-2