Abstract
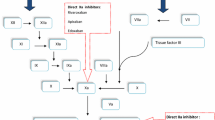
Venous thromboembolism (VTE), which includes deep vein thrombosis and pulmonary embolism, is a major cause of morbidity and mortality in patients undergoing major orthopedic surgery. Routine thromboprophylaxis has been the standard of care over the last 20 years. Currently available options for the prevention of VTE in major orthopedic surgery include low molecular weight heparins, vitamin K antagonists, and, more recently, the synthetic pentasaccharide fondaparinux. Although effective, all these drugs have several limitations and new oral antithrombotics offering predictable, effective and safe anticoagulation are strongly needed. This overview focuses on the most advanced oral direct inhibitors to factor Xa rivaroxaban, apixaban, LY517717 and YM150; specifically, the results of phase II and III studies and the designs of ongoing clinical trials in patients undergoing elective hip and knee replacement are reviewed.



Similar content being viewed by others
References
Silverstein MD, Heit JA, Mohr DN et al (1998) Trends in the incidence of deep vein thrombosis and pulmonary embolism: a 25-year population-based study. Arch Intern Med 158:585–593
Ageno W, Squizzato A, Garcia D, Imberti D (2006) Epidemiology and risk factors of venous thromboembolism. Semin Thromb Hemost 32:651–658
Cohen AT, Agnelli G, Anderson FA et al (2007) Venous thromboembolism (VTE) in Europe. The number of VTE events and associated morbidity and mortality. Conflict of interest statementThromb Haemost 98:756–764
Geerts WH, Pineo GF, Heit JA et al (2004) Prevention of venous thromboembolism: the 7th ACCP conference on antithrombotic and thrombolytic therapy. Chest 126:338S–400S
Pellegrini VD, Donaldson CT, Farber DC et al (2005) Prevention of readmission for venous thromboembolic disease after total hip arthroplasty. Clin Orthop 441:56–62
Pellegrini VD, Donaldson CT, Farber DC et al (2006) The Mark Coventry Award: prevention of readmission for venous thromboembolism after total knee arthroplasty. Clin Orthop 452:21–27
Geerts WH, Bergqvist D, Pineo GM et al (2008) Prevention of venous thromboembolism. American College of Chest Physicians evidence-based clinical practice guidelines, 8th edn. Chest 133:381S–453S
Weitz JI, Hirsh J, Samama MM (2008) New antithrombotic drug. American College of Chest Physicians evidence-based clinical practice guidelines, 8th edn. Chest 133:234S–256S
Turpie AG, Bauer KA, Eriksson BI et al (2004) Superiority of fondaparinux over enoxaparin in preventing venous thromboembolism in major orthopedic surgery using different efficacy end points. Chest 126:501–508
Warkentin TE, Greinacher A, Koster A et al (2008) Treatment and prevention of heparin-induced thrombocytopenia. American College of Chest Physicians evidence-based clinical practice guidelines, 8th edn. Chest 133:330S–380S
Furie B, Furie BC (1990) Molecular basis of vitamin K-dependent γ-carboxylation. Blood 75:1753–1762
Harenberg J (2008) Indirect and direct anticoagulants predominantly inhibiting factor Xa. Therapy 5:177–192
Bauer KA (2006) New anticoagulants: anti IIa vs anti Xa—is one better? J Thromb Thrombolysis 21:67–72
Turpie AG (2007) Oral, direct factor Xa inhibitors in development for the prevention and treatment of thromboembolic diseases. Arterioscler Thromb Vasc Biol 27:1238–1247
Strassburger J, Wilmen A, Pohlmann J et al (2005) In vitro and in vivo studies of the novel antithrombotic agent BAY 59–7939—an oral, direct factor Xa inhibitor. J Thromb Haemost 3:514–521
Kubitza D, Becka M, Wensing G et al (2005) Safety, pharmacodynamics, and pharmacokinetics of BAY 59–7939—an oral, direct factor Xa inhibitor—after multiple dosing in healthy male subjects. Eur J Clin Pharmacol 61:873–880
Gross PL, Weitz JI (2008) New anticoagulants for treatment of venous thromboembolism. Arterioscler Thromb Vasc Biol 28:380–386
Kubitza D, Becka M, Zuehlsdorf M et al (2007) Body weight has limited influence on the safety, tolerability, pharmacokinetics, or pharmacodynamics of rivaroxaban (BAY 59–7939) in healthy subjects. J Clin Pharmacol 47:218–226
Kubitza D, Becka M, Mueck W et al (2006) The effect of extreme age, and gender, on the pharmacology and tolerability of rivaroxaban an oral, direct factor Xa inhibitor. Blood 108 (Abstract 905)
Halabi A, Maatouk H, Klause N et al (2006) Effects of renal impairment on the pharmacology of rivaroxaban (BAY 59-7939) an oral, direct, factor Xa inhibitor. Blood 108 (Abstract 913)
Kubitza D, Becka M, Zuehlsdorf M et al (2006) Effect of food, an antacid, and the H2 antagonist ranitidine on the absorption of BAY 59–7939 (rivaroxaban), an oral, direct factor Xa inhibitor, in healthy subjects. J Clin Pharmacol 46:549–558
Kubitza D, Becka M, Mueck W et al (2006) Safety, tolerability, pharmacodynamics, and pharmacokinetics of rivaroxaban—an oral, direct factor Xa inhibitor—are not affected by aspirin. J Clin Pharmacol 46:981–990
Turpie A, Fisher W, Bauer K et al (2005) BAY 59–7939: an oral, direct factor Xa inhibitor for the prevention of venous thromboembolism in patients after total knee replacement: a phase II dose-ranging study. J Thromb Haemost 3:2479–2486
Eriksson BI, Borris LC, Dahl OE et al (2006) Oral, direct factor Xa inhibition with BAY 59–7939 for the prevention of venous thromboembolism after total hip replacement. J Thromb Haemost 4:121–128
Eriksson BI, Borris LC, Dahl OE et al (2006) A once-daily, oral, direct factor Xa inhibitor, rivaroxaban (BAY 59–7939), for thromboprophylaxis after total hip replacement. Circulation 114:2374–2381
Eriksson BI, Borris LC, Dahl OE et al (2007) Dose-escalation study of rivaroxaban (BAY 59–7939)—an oral, direct factor Xa inhibitor—for the prevention of venous thromboembolism in patients undergoing total hip replacement. Thromb Res 120:685–693
Mueck W, Borris LC, Dahl OE et al (2008) Population pharmacokinetics and pharmacodynamics of once- and twice-daily rivaroxaban for the prevention of venous thromboembolism in patients undergoing total hip replacement. Thromb Haemost 100:453–461
Eriksson BI, Borris LC, Friedman RJ et al (2008) Rivaroxaban versus enoxaparin for thromboprophylaxis after hip arthroplasty. N Engl J Med 358:2765–2775
Kakkar AK, Brenner B, Eriksson BI et al (2008) Extended duration of rivaroxaban versus short term enoxaparin for the prevention on venous thromboembolism after total hip arthroplasty: a double blind, randomized controlled trial. Lancet 372:29–37
Lassen MR, Ageno W, Borris LC et al (2008) Rivaroxaban versus enoxaparin for thromboprophylaxis after total knee arthroplasty. N Engl J Med 358:2776–2786
Turpie A, Lassen M, Davidson B et al (2009) Rivaroxaban versus enoxaparin for thromboprophylaxis after total knee arthroplasty (RECORD4): a randomised trial. Lancet 373:1673–1680
Turpie A, Lassen M, Kakkar AJ et al (2008) A pooled analysis of four pivotal studies of rivaroxaban for the prevention of venous thromboembolism after orthopaedic surgery: effect on symptomatic venous thromboembolism, death and bleeding. American Society of Haematology 2008 annual meeting, December 6–9, 2008, San Francisco, Abstract 36
Imberti D, Dall’Asta C (2008) Clinical benefits of rivaroxaban for thromboprophylaxis after major orthopaedic surgery. Adv Orthop 1:2–8
Pinto DJ, Orwat MJ, Koch S et al (2007) Discovery of 1-(4-methoxyphenyl)7-oxo-6-(4-(2-oxopiperidin-1-yl)phenyl)-4,5,6,7-tetrahydro-1H-pyrazolo[3,4-c]pyridine-3-carboxamide apixaban, BMS-562247), a highly potent, selective, efficacious, and orally bioavailable inhibitor of blood coagulation factor Xa. J Med Chem 50:5339–5556
Wong PC, Crain EJ, Xin B et al (2007) Apixaban, an oral, direct and selective factor Xa inhibitor: in vitro, antithrombotic and antihemostatic studies. J Thromb Haemost 6:820–829
Raghavan N, Frost CE, Yu Z et al (2008) Apixaban metabolism and pharmacokinetics following oral administration to humans. Drug Metab Dispos [Epub ahead of print]
Lassen MR, Davidson BL, Gallus A et al (2007) The efficacy of apixaban, an oral, direct factor Xa inhibitor, as thromboprophylaxis in patients following total knee replacement. J Thromb Haemost 5:2368–2375
Lassen M, Gallus A, Pineo G et al (2008) Randomized double-blind comparison of apixaban with enoxaparin for thromboprophyaxis after knee replacement: the ADVANCE-1 trial. American Society of Haematology 2008 annual meeting, December 6–9, 2008, San Francisco, Abstract 31
Agnelli G, Haas SK, Krueger KA (2007) A phase II study of the oral factor Xa inhibitor LY517717 for prevention of venous thromboembolism after hip or knee replacement. J Thromb Haemost 5:746–753
Eriksson BI, Turpie AGG, Lassen MR et al (2007) A dose escalation study of YM150, an oral direct factor Xa inhibitor, in the prevention of venous thromboembolism in elective primary hip replacement. J Thromb Haemost 5:1660–1665
Gruber A, Marzec UM, Buetehorn U et al (2007) Recombinant factor VIIa reverses the effects of a high dose of rivaroxaban in baboons. Pathophysiol Haemost Thromb 36(Suppl 1):A39 (abstract)
Perzborn E, Tinel H (2007) FEIBA reverses the effects of a high dose of rivaroxaban in rats. Pathophysiol Haemost Thromb 36(Suppl 1):A40 (abstract)
Conflict of interest statement
The authors declare that they have no conflict of interest related to the publication of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Imberti, D., Dall’Asta, C. & Pierfranceschi, M.G. Oral factor Xa inhibitors for thromboprophylaxis in major orthopedic surgery: a review. Intern Emerg Med 4, 471–477 (2009). https://doi.org/10.1007/s11739-009-0293-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11739-009-0293-9