Abstract
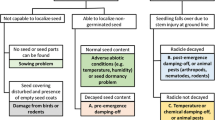
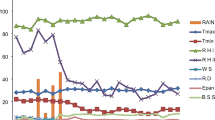
To compare C. monogyna seed germination and the seedling emergence of two populations from Quchan and Bardaskan in Khorasan-e-Razavi-I province in north east of Iran, experiments were carried out. From the results, the highest germination percentages for Quchan (98%) and Bardaskan (97%) populations were observed at alternating 25/15 and 30/20 °C day/night temperatures, respectively. Germination percentage of both populations was 42–92% over a wide range of constant temperatures (10–35 °C). The NaCl concentration of 399.5 and 443.6 mM caused 50% inhibition of germination for Quchan and Bardaskan populations, respectively. The osmotic potential for 50% inhibition of germination was − 0.63 for Quchan and − 0.66 MPa for Bardaskan populations. The maximum emergence of Quchan (74%) and Bardaskan (80%) populations were recorded in the seeds placed at 0.5 cm with emergence failure at 5 cm soil depth. In another experiment in which seeds were after-ripened at different depths of soil in the field, seed germination was lower on the soil surface and 20 cm soil depth compared to 2, 5, 10 and 15 cm depth; and the highest seed emergence in the field was observed at 5 cm soil depth, 270 days after seed burial. The information of this study would be useful for determining the potential of this parasitic species to spread to new areas and arrange management strategies in different environmental conditions.





Similar content being viewed by others
References
Ahmed S, Opena JL, Chauhan BS (2015) Seed germination ecology of doveweed (Murdannia nudiflora) and its implication for management in dry-seeded rice. Weed Sci 63:491–501
Amini R, Gholami F, Ghenepour S (2017) Effects of environmental factors and burial depth on seed germination and emergence of two populations of Caucalis platycarpos. Weed Res 57:247–256
Benvenuti S, Macchia M, Miele S (2001) Quantitative analysis of emergence of seedlings from buried weed seeds with increasing soil depth. Weed Sci 49:528–535
Benevenuti S, Dinelli G, Bonetti A, Catixone P (2005) Germination ecology’ emergence and host detection in Cuscuta campestris. Weed Res 45:270–278
Booth BD, Murphy SD, Swanton CJ (2003) Weed ecology in natural and agricultural systems. CAB International, Wallingford, pp 93–94
Chauhan BS, Johnson DE (2010) The role of seed ecology in improving weed management strategies in the tropics. Advances in Agron 105:221–262
Chauhan BS, Gill G, Preston C (2006) Factors affecting seed germination of annual sowthistle (Sonchus oleraceus) in southern Australia. Weed Sci 54:854–860
Costea M, Stefanovic S (2010) Evolutionary history and taxonomy of the Cuscuta umbellate complex (Convolvulaceae): Evidence of extensive hybridization from discordant nuclear and plastid phylogenies. Taxon 59:1783–1800
Cudney DW, Orloff SB, Reints JS (1992) An integrated weed management procedure for the control of dodder (Cuscuta indecora) in alfalfa (Medicago sativa). Weed Tech 6:603–606
Dawson JH (1987) Cuscuta (Convolvulaceae) and its control. In: Parasitic Flowering Plants. Proc. 4th International. Symposium, Marburg, Germany, pp 137–149
de Cauwer B, Devos R, Cearhout S, Bulcke R, Reheul D (2014) Seed dormancy, germination, emergence and seed longevity in Galinsoga parviflora and G. quadriradiata. Weed Res 54:38–47
Ebrahimi E, Eslami SV (2012a) Effect of different treatments on dormancy breaking and seed germination of Eastern dodder (Cuscuta monogyna Vahl) and African rocket (Malcolmia africana L. (R.BR.)). Plant Prot 26:191–198
Ebrahimi E, Eslami SV (2012b) Effect of environmental factors on seed germination and emergence of invasive Ceratocarpus arenarius. Weed Res 52:50–59
Eslami SV (2011) Comparative germination and emergence ecology of two populations of common lambsquarters (Chenopodium album L.) from Iran and Denmark. Weed Sci 59:90–97
Fathoulla CN, Mosleh MS (2008) Biological and anatomical study of different Cuscuta species. Kurdistan Conf Biol 11:22–39
Ghorbani R, Seel W, Leifert C (1999) Effects of environmental factors on germination and emergence of Amaranthus retroflexus. Weed Sci 47:505–510
Goldwaser MH, Rubin B, Eizenberg H (2016) Field Dodder (Cuscuta campestris)- a new model describing temperature-dependent seed germination. Weed Sci 64:53–60
Guan B, Zhuo D, Zhang H, Tian Y, Japhet W, Wang P (2009) Germination responses of Medicago ruthenica seeds to salinity, alkalinity and temperature. Arid Environ 73:135–138
Hadas A (1976) Water uptake and germination of leguminous seeds under changing external water potential in osmotic solutions. J Exp Bot 27:480–489
Holml DJ, Panch J, Harbeeger J (1997) World weeds: natural histories and distribution. John Willey & Sons, New York, USA
Hu XW, Wang YR, Wu YP (2009) Effects of the pericarp on imbibition, seed germination, and seedling establishment in seeds of Hedysarum scoparium Fisch. et Mey. Ecological Res 24:559–564
Huarth HR, Zorraquin MRP, Bursztyn EM, Zapiola ML (2016) Effects of environmental factors on seed germination and seedling emergence of common teasel (Dipsacus fullonum). Weed Sci 64:421–429
Hutchison JM, Ashton FM (1979) Effect of desiccation and scarification on the permeability and structure of the seed coat of Cuscuta campestris. Am J Bot 66:40–46
Karimi H (2001) Weeds of Iran. Tehran, Iran, p 419
Krsmanovic MS, Bozic D, Pavlovic M, Radivojevic L, Vrbnicanin S (2013) Temperature effects on Cuscuta campestris seed germination. Pestic Phyt 28:187–193
Kuijy J (1969) The biology of parasitic flowering plant. University of California Press, Berkeley, CA, p 246
Lanini WT, Kogan M (2005) Biology and management of Cuscuta in crops. INV Agric 32:165–179
Malik MS, Norsworthy J, Riley MB, William B (2010) Temperature and light requirements for Wild Radish (Raphanus raphanistrum) germination over a 12-month period following maturation. Weed Sci 58:136–140
Mennan H, Ngouajio M (2006) Seasonal cycles in germination and seedling emergence of summer and winter populations of catchweed bedstraw (Galium aparine) and wild mustard (Brassica kaber). Weed Sci 54:114–120
Michel BE (1983) Evaluation of the water potentials of solutions of polyethylene glycol 8000 both in the absence and presence of other solutes. Plant Physiol 72:66–70
Nazari S (2014) Introducing Cuscuta monogyna as Oak Trees Parasite, its Biology, and Method to Fight in Lorestan province. Bull Environ Pharmacol Life Sci 3:164–168
Norsworthy JK, Olivera MJ (2006) Sicklepod (Senna obtusifolia) germination and emergence as affected by environmental factors and burial depth. Weed Sci 54:903–909
Nosratti I, Abbasi R, Bagheri A, Bromandan P (2017) Seed germination and seedling emergence of Iberian starthistle (Centaurea iberica). Weed Biol and Manag 17:144–149
Rashed Mohassel MH, Najafi H, Akbarzadeh MD (2009) Weed biology and control, 2nd edn. Ferdowsi University of Mashhad Press, p 404
Richards LA (1954) Diagnosis and Improvement of Saline and Alkali Soils. USDA Agricultural Handbook 60, Washington
Salimi H, Shahraeen N (2000) Study on comparison of seed dormancy and germination of three species of dodder. Rostaniha 1:33–36
Tsivion Y (1981) Suppression of axillary buds of its host by parasitic Cuscuta I. competition among sinks and indirect inhibition. New Phytol 87:91–99
Waisel Y (1972) Biology of halophytes. Academic Press, New York and London
Zand E, Baghestani MA, Shimi P, Nezamabadi N, Mousavi MR, Mousavi SK (2017) Chemical weed control guideline for major crops of Iran. Ferdowsi University of Mashhad Press, p 224
Acknowledgements
We would like to thank Ferdowsi University of Mashhad for its financial support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflicts of interest have been declared.
Additional information
Communicated by H. Peng.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ebrahimi, E., Darbandi, E.I., Mohassel, M.H.R. et al. Seed germination and seedling emergence in two populations of eastern dodder (Cuscuta monogyna Vahl.): evaluation of environmental factors and burial depth. Acta Physiol Plant 44, 26 (2022). https://doi.org/10.1007/s11738-021-03354-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11738-021-03354-7