Abstract
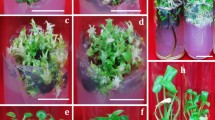
The occurrence of strong polarity towards shoot bud induction and the effect of cytokinin(s) on each segment of stem axis, encapsulation and storability of de novo Shoot buds of Curculigo orchioides Gaertn. (Hypoxidaceae) have been documented in the present communication. Maximum number of shoot buds arising de novo from the stem discs (cross section) explanted from proximal end on MS medium fortified with BAP and KIN 1 mg/L each. Stem discs from distal end were less efficient in shoot bud induction. A combination of two cytokinins (BAP and KIN) as a synergistic effect on shoot buds induction from each segment of stem axis. Stem discs in inverted position produced shoot buds from the lower surface, showing strong polarity within the explant. Further, storability and shoot development of sodium alginate encapsulated shoot buds of Curculigo orchioides were tested on half-strength Murashige and Skoog (MS) basal medium fortified with coconut water (10% v/v). The frequency of regeneration from encapsulated shoot buds was affected significantly by concentration of sodium alginate and the duration of exposure to calcium chloride. Shoot buds encapsulated with 2.5% sodium alginate dissolved in MS basal salts solution recorded significantly higher shoot development than other treatments. A relatively short (5 min) incubation with calcium chloride solution provided uniform encapsulation of shoot buds that gave the highest percentage (68%) of shoot development. Encapsulated shoot buds could be stored at 4°C for 50 days without reduction in viability as oppose to non-encapsulated shoot buds, which showed 9.5% viability after 20 days at 4°C. Encapsulated shoot bud developed into normal shoots. Based on the present observations an improved protocol may be developed for the rapid multiplication and conservation of the endangered species—C. orchioides.




Similar content being viewed by others
Abbreviations
- MS:
-
Murashige and Skoog medium
- BAP:
-
6-Benzylaminopurine
- KIN:
-
Kinetin
- CaCl2 :
-
Calcium chloride
References
Ara H, Jaiswal U, Jaiswal VS (2000) Synthetic seed: prospects and limitations. Curr Sci 78(12):1438–1444
Augustine AC, Souza LD (1997) Micropropagation and conservation of medicinal plants. In: Ravishankar GA, Venkataraman LV (eds) Biotechnological applications of plant tissue and cell culture. Oxford and IBH publication, Calcutta, pp 116–118
Chopra RN, Nayar SL, Chopra LC (1956) In glossary of indian medicinal plants. CSIR, New Delhi, pp 84
Dhar ML, Dhar MN, Dhawan BN, Mehrota DN, Ray C (1968) Screening of Indian plants for biological activity part-I. Indian J Exp Biol 6:232–249
Ipekci Z, Gozukirmizi N (2003) Direct embryogenesis and synthetic seed production from Paulownia elongate. Plant Cell Rep 22:16–24. doi:10.1007/s00299-003-0650-5
Jagadishchandra KS, Rachappaji S, Gowda KRD, Tharasaraswathi KJ (1999) In vitro propagation Pisonia alba (C) Spanogae (Lettuce tree)—a threatened species. Phytomorphology 49:43–47
Lakshmiprasad B, Shanthamma C (1979) Culture of stem bits of Cenchrus glaucus. Curr Sci 48(16):741–742
Misra TN, Singh RS, Upadhya J, Tripathi DNM (1984) Aliphatic compounds from Curculigo orchioides. Phytochem 23:1643–1645. doi:10.1016/S0031-9422(00)83459-7
Misra TN, Singh RS, Tripathi Sharma SC (1990) Curcligol a cycloartane triferpene alcohol from Curculigo orchioides. Phytochemistry 29:929–931. doi:10.1016/0031-9422(90)80048-L
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassay with tobacco tissue culture. Physiol Plant 15:473–497. doi:10.1111/j.1399-3054.1962.tb08052.x
Pattnaik S, Chand PK (2000) Morphogenic response of alginate encapsulated axillary buds from in vitro shoot cultures of six mulberry. Plant Cell Rep 6:177–195
Redenbaugh K, Viss P, David S, Fujii JA (1987) Scale-up artificial seeds plant tissue culture Alan R. Liss. Inc., New York, pp 473–493
Sage DO, Lynn J, Hammatt N (2000) Somatic embriyogenesis in Narcissus pseudonarcissus Cvs Golden Harvest and St. Keverne. Plant Sci 150:209–216. doi:10.1016/S0168-9452(99)00190-9
Sanyal Dutta, Gupta S, Jana MK, Kunolu MC (1998) Shoot organogenesis and plantlet regeneration from leaf callus cultures of tuberose (Polianthes tubesosa L.). Plant Tissue Cult Biotechnol 4:81–86
Srivastava TL, Tiwari KP, Rubysharma (1998) Medicinal plants of Madhya Pradesh; immediate need of conservation. J Trop Forestry 15:144–151
Suchitra B, Mehar Z, Kumar S (1999) In vitro multiplication of Centella astitica. Curr Sci 76:147–148
Suri SS, Jian S, Ramawat KG (1999) Plant regeneration and bullbills formation in vitro from leaf and stem explants of Curculigo orchioides an endangered medicinal plant. Sci Hortic (Amsterdam) 79:127–134. doi:10.1016/S0304-4238(98)00118-6
Xu, JP, Xu RS, Li XY (1992) Glycosides of cycloartane sapogenine from Curculigo orchioides. Phytochemistry 31:233–236. doi:10.1016/0031-9422(91)83043-K
Acknowledgment
Senior author grateful to University of Mysore for providing facility and financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by E. Lojkowska.
Rights and permissions
About this article
Cite this article
Nagesh, K.S., Shanthamma, C. & Bhagyalakshmi, N. Role of polarity in de novo shoot bud initiation from stem disc explants of Curculigo orchioides Gaertn. and its encapsulation and storability. Acta Physiol Plant 31, 699–704 (2009). https://doi.org/10.1007/s11738-009-0281-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11738-009-0281-8