Abstract
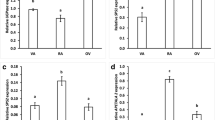
Activities of some key enzymes of carbon metabolism sucrose synthase, acid and alkaline invertase, phosphoenol pyruvate carboxylase, malic enzyme and isocitrate dehydrogenase were investigated in relation to the carbohydrate status in lentil pods. Sucrose remained the dominant soluble sugar in the pod wall and seed, with hexoses (glucose and fructose) present at significantly lower levels. Sucrose synthase is the predominant sucrolytic enzyme in the developing seeds of lentil (Lens culinaris L.). Acid invertase was associated with pod elongation and showed little activity in seeds. Sucrose breakdown was dominated by alkaline invertase during the development of podwall, while both the sucrose synthase and alkaline invertase were active in the branch of inflorescence. A substantial increase of sucrolytic enzymes was observed at the time of maximum seed filling stage (10–20 DAF) in lentil seed. The pattern of activity of sucrose synthase highly paralleled the phase of rapid seed filling and therefore, can be correlated with seed sink strength. It seems likely that the fruiting structures of lentil utilize phosphoenol pyruvate carboxylase for recapturing respired carbon dioxide. Higher activities of isocitrate dehydrogenase and malic enzyme in the seed at the time of rapid seed filling could be effectively linked to the deposition of protein reserves.
Similar content being viewed by others
Abbreviations
- DAF:
-
days after flowering
- SS:
-
sucrose synthase
- SPS:
-
sucrose phosphate synthase
- PEP:
-
phosphoenol pyruvate
- ICD:
-
isocitrate dehydrogenase
- PCR:
-
photosynthetic carbon reduction
- BI:
-
branch of inflorescence
- PW:
-
podwall
References
Chopra J., Kaur N., Gupta A. K. 1998. Carbohydrate status and sucrose metabolism in mungbean roots and nodules. Phytochem. 49: 1891–1895.
Chopra J., Kaur N., Gupta A. K. 2000. Ontogenic changes in enzymes of carbon metabolism in relation to carbohydrate status in developing mungbean reproductive structures. Phytochem. 53: 539–548.
Chopra J., Kaur N., Gupta A. K. 2002. A comparative developmental pattern of enzymes of carbon metabolism and pentose phosphate pathway in mungbean and lentil nodules. Acta Physiol. Plant. 24: In Press
Christeller J. T., Laing W. A., Sutton W. D. 1977. Carbon dioxide fixation by lupin root nodules. I. Characterization, association with phosphoenol-pyruvate carboxylase, and correlation with nitrogen fixation during nodule development. Plant Physiol. 60: 47–50.
Claussen W., Hawker J. S., Loveys B. R. 1985. Sucrose synthase activity, invertase activity, net photosynthetic rates and carbohydrate content of detached stems and shoots (sinks). J. Plant Physiol. 119: 123–132.
Echeverria E., Humphreys T. 1984. Involvement of sucrose synthase in sucrose catabolism. Phytochem. 23: 2173–2178.
Edwards J., apRees T. 1986. Sucrose partitioning in developing embryos of round and wrinkled varieties of Pisum sativum. Phytochem. 25: 2027–2032.
Estruch J. J., Beltren J. P. 1991. Changes in invertase activities precede ovary growth induced by gibberellic acid in Pisum sativum. Physiol. Plant. 81: 319–326.
Gupta V. K., Singh R., Prasad B. N., Ghimire G. P. S., Agarwal V. P. 1992. In: B.N. Prasad and G.P.S. Ghimire, (Eds). Role of biotechnology in Agriculture. Podwall metabolism in relation to dry matter and protein accumulation in developing seeds of chickpea (Cicer arietinum L.). pp. 21–36.
Heim U., Weber H., Baumlein H., Wobus U. 1993. A sucrose-synthase gene of Vicia faba L: expression pattern in developing seeds in relation to starch synthesis and metabolic regulation. Planta 191: 394–401.
Huber S. C., Akazawa T. 1986. A novel sucrose synthase pathway for sucrose degradation in cultured sycamore cells. Plant Physiol. 81: 1008–1013.
Iglesias A. A., Andreo C. S. 1990. Kinetic and structural properties of NADP-malic enzyme from sugarcane leaves. Plant Physiol. 92: 66–72.
Latzko E., Kelly G. J. 1983. The many-faceted function of phosphoenolpyruvate carboxylase in C3 plants. Physiol. Veg. 21: 805–815.
Lee Y. P. and Takahashi T. 1966. An improved colorimetric determination of amino acid with the use of nihydrin. Anal. Biochem. 14: 71–77.
Lowell C. A., Kuo T. M. 1989. Oligosaccharide metabolism and accumulation in developing soybean seeds. Crop Sci. 29: 459–465.
Lowry O. H., Rosebrough N. J., Farr A. L., Randall R. J. 1951. Protein measurement with Folin phenol reagent. J. Biol. Chem. 93: 265–275.
Minamikawa T., Yamauchi D., Wade S., Takeuchi H. 1992. Expression of α-amylase in Phaseolus vulgaris and Vigna mungo plants. Plant Cell Physiol. 33: 253–258.
Murray D. R. 1987. Nutritive role of seed coats in developing legume seeds. Am. J. Bot. 74: 1122–1137.
Nelson N. 1944. A photometric adaptation of Somogyi method for the determination of glucose. J. Biol. Chem. 157: 375–380.
Peoples M. B., Atkins C. A., Pate J. S., Murray D. R. 1985. Nitrogen nutrition and metabolic interconversions of nitrogenous solutes in developing cowpea fruit. Plant Physiol. 77: 382–388.
Ross H. A., McRae D., Davies H. V. 1996. Sucrolytic enzyme activities in cotyledons of the faba bean. Developmental changes and purification of alkaline invertase. Plant Physiol. 111: 329–338.
Sharma N., Kaur N., Gupta A. K. 1998. Effect of chlorocholine chloride sprays on carbohydrate composition and activities of sucrose metabolizing enzymes in potato (Solanum tuberosum L.). Plant Growth Regulation 26: 97–103.
Singal H. R., Sheoran I. S., Singh R. 1986. Products of photosynthetic 14CO2 fixation and related enzyme activities in fruiting structures of chickpea. Physiol. Plant. 66: 457–462.
Solanki I. S., Kapoor A. C., Singh U. 1999. Nutritional parameters and yield evaluation of newly developed genotypes of lentil. Plant food for human nutrition 54: 79–87.
Sung S. S., Sheih W. J., Geiger D. R., Black C. C. 1994. Growth, sucrose synthase and invertase activities of developing Phaseolus vulgaris L. fruits. Plant Cell Environ. 17: 419–426.
Sung S. S., Xu D. P., Black C. C. 1989. Identification of actively filling sucrose sinks. Plant Physiol. 89: 1117–1121.
Tezuka T., Yamamoto Y., Kondo N. 1990. Activation of oxygen uptake and NAD-specific isocitrate dehydrogenase in mitochondria isolated from cotyledons of castor bean by cis, trans-abscisic acid. Plant Physiol. 92: 147–150.
Van Handel E. 1968. Direct microdetermination of sucrose. Anal. Biochem. 22: 280–283.
Weber H., Borisjuk L., Wobus U. 1997. Sugar import and metabolism during seed development. Trends Pl. Sci. 2: 169–174.
Williard L. M., Slattery M. 1945. The colorimetric determination of fructosan in plant material. J. Biol. Chem. 157: 161–167.
Wolswinkel P., Ammerlaan A. 1984. Turgor-sensitive sucrose and amino acid transport into developing seeds of Pisum sativum. Effect of high sucrose or mannitol concentration in experiments with empty ovules. Physiol. Plant. 61: 172–182.
Xu D. P., Sung S. J. S., Black C. C. 1989. Sucrose metabolism in lima bean seeds. Plant Physiol. 89: 1106–1116.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chopra, J., Kaur, N. & Gupta, A.K. Changes in the activities of carbon metabolizing enzymes with pod development in lentil (Lens culinaris L.). Acta Physiol Plant 25, 185–191 (2003). https://doi.org/10.1007/s11738-003-0052-x
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11738-003-0052-x