Abstract
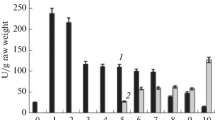
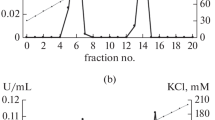
In the investigated 14 day old triticale seedlings a much higher GDH activity was observed in roots than in leaves. The enzyme from the roots was purified up to the state of homogeneity (about 400 fold). The purified enzyme showed a higher activity in the presence of reduced coenzyme forms (NAD(P)H) than their oxidated forms. In the presence of NAD(P)H the enzyme showed absolute specificity to 2-oxoglutarate and in cooperation with NAD(P)+ to L-glutamate. The Km values determined for particular substrates indicate a high affinity of NADPH-GDH to ammonium ions.
Optimum pH, temperature and thermostability of GDH depended on the type and form of the coenzyme. Molecular mass of purified enzyme was 257 kDa. It seems that native GDH is composed of six identical subunits of the molecular mass 42.5 kDa.
Similar content being viewed by others
References
Alekhina N.D., Klyuikova A.J., Gerasimova S. 1984. Adaptative responses in glutamine synthetase and glutamate dehydrogenase from the roots of winter wheat seedlings grown at a various temperature. Fiziol. Rast. 31: 344–350.
Barash I., Sadn T., Mor H. 1973. Induction of a specific isoenzyme of glutamate dehydrogenase by ammonia in oat leaves. Nature New. Biol., 244: 150–152.
Becker T.W., Carrayol E., Hirel B. 2000. Glutamine synthetase and glutamate dehydrogenase isoforms in maize leaves: localization, relative proportion and their role in ammonium assimilation or nitrogen transport. Planta, 211: 800–806.
Bielawski W., Rafalski A. 1979. Glutamate dehydrogenase and glutamine synthetase in rye seedlings supplied with ammonium and nitrate. Acta Biochem. Pol. 26: 383–396.
Bielawski W., Kwinta J., Kączkowski J. 1989. Comparison of some cereal seedlings on the ability of glutamine synthetase induction. Acta Physiol. Plant. 11: 147–156.
Bielawski W. 1994. Effect of some compounds on glutamine synthetase isoforms activity from Triticale seedling leaves. Acta Physiol. Plant. 15: 211–218.
Czosnowski J. 1974. Metabolism of excised embrios of Lupinus luteus L VI. An electrophoretic analysis of some dehydrogenases in cultured as compared with the normal seedling axes. Acta Soc. Bot. Pol. 43: 117–127.
Fereira R., Davies D. 1989. Nitrogen supply and light intensity on properties of glutamate dehydrogenase and glycolate oxidase in Lemna. Phytochemistry 28: 349–354.
Garcia R.N., Mendoza E.M.T., Menancio-Hautea D.I., Robles R.P. 1994. Kinetic properties and variation in activity levels of root nodule ammonia and carbon assimilation enzymes in mung bean, Vigna radiata L. Wilczek. Philip. J. Crop. Sci. 19: 11–18.
Gulati A., Jaiwal J.K., 1996. Effect of NaCl on nitrate reductase, glutamate dehydrogenase and glutamate synthase in Vigna radiata calli. Biol. Plant. 38: 177–183.
Heeschen V., Gerendas J., Richter C.P., Rudolph H. 1997. Glutamate dehydrogenase of Sphagnum. Phytochemistry 45: 881–887.
Inokuchi R., Itagaki T., Wiskich J.T., Nakayama K., Okada M. 1997. An NADP-glutamate dehydrogenase from green alga Bryopsis maxima. Purification and properties. Plant Cell Physiol. 38: 372–335.
Itagaki T., Dry I.B., Wiskich J. 1988. Purification and properties of NAD-glutamate dehydrogenase from turnip mitochondria. Phytochemistry. 27: 3373–3378.
Kumar R.G., Shah K., Dubey R.S. 2000. Salinity induced behavioural changes in malate dehydrogenase and glutamate dehydrogenase activities in rice seedlings of differing salt tolerance. Plant Sci. 156: 23–34.
Kumar S., Ma B., Tsai Chung-Jung, Nussinov R. 2000. Electrostatic strengths of salt bridges in thermophilic and mesophilic glutamate dehydrogenase monomers. Proteins: Structure, Function and Genetics 38: 368–383.
Kwinta J., Bartoszewicz K., Bielawski W. 1999. Glutamate dehydrogenase and glutamate synthetase activities during development of Triticale grains. Acta Physiol. Plant., 21: 271–275.
Kwinta J., Bielawski W. 1998. Glutamate dehydrogenase in higher plants. Acta Phys. Plant. 20: 453–463.
Laemmli U.K., 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature, 227: 680–685.
Lewis O., James D.M., Hewitt E.J. 1982. Nitrogen assimilation in barley (Hordeum vulgare L. cv. Mazurka) in response to nitrate and ammonium nutrition. Ann. Bot. 49: 39–49.
Lineweaver M., Burk D.J. 1934. The determination of enzyme dissociation constans. J. Am. Chem. Soc., 56: 658–666.
Loulakakis K.A., Roubelakis-Angelakis K.A. 1990. Intracellular localization and properties of NADH-glutamate dehydrogenase from Vitis vinifera L.: Purification and characterization of the major leaf isoenzyme. J. Exp. Bot. 41: 1223- 1230.
Loulakakis K.A., Roubelakis-Angelakis K.A. 1991. Plant NADH-glutamate dehydrogenase consist of two subunit polipeptides and their participation in the seven isoenzymes occurs in an ordered ratio. Plant. Physiol. 97: 104–111.
Loulakakis K.A., Roubelakis-Angelakis K.A. 1996. The seven NADH-glutamate dehydrogenase izoenzymes exhibit similar anabolic and katabolic activities. Physiol. Plant. 96: 29–35.
Lowry H.O., Rosebrough N.J., Farr A.L., Randall R.J. 1951. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193: 265–275.
Loyola-Vargas V.M., Yanez A., Caldera J., Oropeza C., Quiroz R.M.L., Scorer K.N. 1988. Nitrogen metabolism in Canavalia ensiformis (L.) D.C.II. Changing activities of nitrogen-assimilating enzymes during growth. J. Plant Physiol. 132: 289–293.
Moller J.M., Rasmuson A.G. 1998. The role of NADP in themitochondrial matrix. Trends Plant Sci. 3: 21–27.
Oaks A. 1995. Evidence for deamination by glutamate dehydrogenase in higher plants- reply. Can. J. Bot. 73: 1116–1117.
O’Farell P.Z., Goodman H.M., O’Farell P.H. 1977. High resolution two-dimensional electrophoresis of basic as well as acidic proteins. Cell, 12: 1133–1142.
O’Neal D., Joy K.W. 1973 b. Glutamine synthetase of pea leaves. I. Purification and pH optima. Arch. Biochem. Biophys. 159: 113–122.
Orzechowski S., Kwinta J., Gworek B., Bielawski W. 1997. Biochemical indicators of environmental contaminations with heavy metals. Pol. J. Env. Studies. 6: 29–32.
Robinson S.A., Stewart G.R., Phillips R. 1992. Regulation of glutamate dehydrogenase in relation to carbon limitation and protein catabolism in carrot cell suspension cultures. Plant Physiol. 98: 1190–1195.
Srivastava H.S., Singh R.P. 1987. Role and regulation of L-glutamate dehydrogenase activity in higher plants. Phytochemistry 26: 597–610.
Syntichaki K.M., Loulakakis K.A., Ruobelakis-Angelakis K.A. 1996. The amino acid sequence similarity of plant glutamate dehydrogenase to the extermophilic archaeal enzyme conforms to its stress-related function. Gene 168: 87–92.
Turano F.J., Dashner R., Upadhyaya A., Caldwell C.R. 1996. Purification of mitochondrial glutamate dehydrogenase from dark-grown soybean seedlings. Plant. Physiol. 112: 1357–1364.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kwinta, J., Bartoszewicz, K. & Bielawski, W. Purification and characteristics of glutamate dehydrogenase (GDH) from triticale roots. Acta Physiol Plant 23, 399–405 (2001). https://doi.org/10.1007/s11738-001-0049-2
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11738-001-0049-2