Abstract
Objective
To observe the effects of electroacupuncture (EA) on uterine prostaglandin F2α (PGF2α), cyclooxygenase 2 (COX-2) and nuclear factor κB (NF-κB) in rats with primary dysmenorrhea (PD) and to discuss the possible mechanism in EA intervening PD.
Methods
Forty Sprague-Dawley female rats were randomly divided into a blank group, a model group, an EA group and an ibuprofen group, with 10 rats in each group. The PD model was established using estradiol benzoate combined with oxytocin in the model group, EA group and ibuprofen group. At the same time of modeling, rats in the EA group were given EA at Guanyuan (CV 4) and Sanyinjiao (SP 6) once a day for 20 min each time for 10 consecutive days. Ibuprofen was intragastrically administered once a day for 10 consecutive days in the ibuprofen group. The same amount of normal saline was intragastrically administered once a day for 10 consecutive days in the blank group and model group. The number of writhing of rats in each group within 30 min was compared on the 11th day just after the interventions. The uterine homogenate supernatant was separated and the PGF2α level was detected by enzyme-linked immunosorbent assay. Western blot was applied for the detection of the expression levels of COX-2, phospho-NF-κB p65 and NF-κB p65 proteins in uterine tissues.
Results
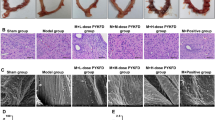
Compared with the blank group, the number of writhing in the model group increased significantly (P<0.01), and the expression levels of PGF2α, COX-2, phospho-NF-κB p65 and NF-κB p65 proteins in uterine tissues were significantly increased (all p<0.01). Compared with the model group, the number of writhing in the EA group and ibuprofen group were significantly reduced (both P<0.01), and the expression levels of PGF2α and COX-2 protein in uterine tissues were significantly reduced (both P<0.01). Compared with the model group, the phospho-NF-κB p65 level in uterine tissues in the EA group was significantly reduced (P<0.01). Compared with the ibuprofen group, the phospho-NF-κB p65 level in the EA group was significantly reduced (P<0.01).
Conclusion
The mechanism of EA for PD rats may be related to inhibiting the phosphorylation of NF-κB and reducing the levels of COX-2 and PGF2α in uterine tissues.
摘要
目的:观察电针对原发性痛经(PD)大鼠子宫组织前列腺素F2α(PGF2α)、环氧化酶2(COX-2)和核因子κB (NF-κB) 的影响, 探讨电针干预PD的可能机制。方法:将40只Sprague-Dawley雌性大鼠随机分为正常组、模型组、电针组 和布洛芬组, 每组10只。模型组、电针组和布洛芬组采用苯甲酸雌二醇联合缩宫素建立PD大鼠模型。造模同时, 电 针组大鼠给予电针关元和三阴交治疗, 每日1次, 每次20 min, 连续10 d; 布洛芬组给予布洛芬灌胃干预, 每日1次, 连续10 d; 正常组和模型组给予等量的生理盐水灌胃, 每日1次, 连续10 d。于第11天比较各组大鼠30 min内扭体 次数; 酶联免疫吸附测定法检测子宫匀浆上清液中PGF2α水平; Western blot检测子宫组织COX-2、磷酸化NF-κB p65 (phospho-NF-κB p65)及NF-κB p65的蛋白表达水平。结果:与正常组相比, 模型组大鼠扭体次数明显增多(P<0.01), 子宫组织PGF2α含量、COX-2、phospho-NF-κB p65及NF-κB p65蛋白水平明显升高(均P<0.01)。与模型组相比, 电针 组和布洛芬组扭体次数明显减少(均P<0.01), 子宫组织PGF2α含量和COX-2蛋白水平明显降低(均P<0.01)。与模型 组相比, 电针组大鼠子宫组织phospho-NF-κB p65蛋白水平明显降低(P<0.01)。与布洛芬组相比, 电针组大鼠子宫 组织phospho-NF-κB p65蛋白水平明显降低(P<0.01)。结论:电针干预PD大鼠的作用机制可能与抑制子宫组织中 NF-κB磷酸化, 降低子宫组织COX-2和PGF2α水平有关。
Similar content being viewed by others
References
Iacovides S, Avidon I, Baker FC. What we know about primary dysmenorrhea today: a critical review. Hum Reprod Update, 2015, 21(6): 762–778.
Chan WY. Prostaglandins and nonsteroidal anti-inflammatory drugs in dysmenorrhea. Annu Rev Pharmacol Toxicol, 1983, 23: 131–149.
Li Q, Xie P, Bai CX, Cui JY, Jiang C, Wu YS. Mechanisms of Fufang Shixiao formula for experimental primary dysmenorrhea. Zhongguo Zhongxiyi Jiehe Xiaohua Zazhi, 2016, 36(9): 1087–1090.
Marjoribanks J, Ayeleke RO, Farquhar C, Proctor M. Nonsteroidal anti-inflammatory drugs for dysmenorrhoea. Cochrane Database Sy st Rev, 2015, (7): CD001751.
Park SM, Min BG, Jung JY, Jegal KH, Lee CW, Kim KY, Kim YW, Choi YW, Cho IJ, Ku SK, Kim SC. Combination of pelargonium sidoides and coptis chinensis root inhibits nuclear factor kappa b-mediated inflammatory response in vitro and in vivo. BMC Complement Altern Med, 2018, 18(1): 20.
Kelly RW, King AE, Critchley HO. Cytokine control in human endometrium. Reproduction, 2001, 121(1): 3–19.
Kim YS, Kim EK, Nawarathna W, Dong X, Shin WB, Park JS, Moon SH, Park PJ. Immune-stimulatory effects of Althaea rosea flower extracts through the MAPK signaling pathway in RAW264.7 cells. Molecules, 2017, 22(5): 679.
Yu SY, Lü ZT, Zhang Q, Yang S, Wu X, Hu YP, Zeng F, Liang FR, Yang J. Electroacupuncture is beneficial for primary dysmenorrhea: the evidence from meta-analysis of randomized controlled trials. Evid Based Complement Alternat Med, 2017, 2017: 1791258.
Chen PB, Chen J, Cui J, Yang XF. Effects of the acupoint catgut embedding on nerve-endocrine-immune network in dysmenorrhea rats. Zhen Ci Yan Jiu, 2018, 43(1): 30–34.
Chen PB, Qi SS, Cui J, Yang XF, Chen J, Wang XG, Yang ZH, Feng L, Xuan J. Herbal-cake-partitioned moxibustion of Shenque (CV 8) has a relative specific effect in relieving abdominal pain and in regulating neuroendocrine-immune network in primary dysmenorrhea rats. Zhen Ci Yan Jiu, 2019, 44(2): 120–124.
Liu Y, Wang YQ, Chen LY, Mo BQ, Wu XX, Xiao Y, Tang B. Effect of electroacupuncture on NF-kB and NLRP3 inflammasome in uterine tissues of rats with primary dysmenorrhea. J Acupunct Tuina Sci, 2019, 17(4): 215–222.
Xiao H, Shi T, Han L, Tong R, Liao Z. Mechanism of penehyclidine hydrochloride on a dysmenorrhea rat model. Drug Dev Res, 2019, 80(3): 325–332.
Camlibel M, Erdur B, Yilmaz A, Ozen M, Uyanik A. Comparison of the effects of piroxicam and diclofenac sodium as treatments for primary dysmenorrhea. Med Sci Monit, 2019, 25: 157–164.
Wu LL, Su CH, Liu CF. Effects of noninvasive electroacupuncture at Hegu (LI 4) and Sanyinjiao (SP 6) acupoints on dysmenorrhea: a randomized controlled trial. J Altern Complement Med, 2012, 18(2): 137–142.
Luo F, Huang X, Liu X, Wang L, Xu N. Comparative efficacy and safety of NSAIDs-controlled acupuncture in the treatment of patients with primary dysmenorrhea: a Bayesian network meta-analysis. J Int Med Res, 2019, 47(1): 19–30.
Xu T, Hui L, Juan YL, Min SG, Hua WT. Effects of moxibustion or acupoint therapy for the treatment of primary dysmenorrhea: a meta-analysis. Altern Ther Health Med, 2014, 20(4): 33–42.
Sun L, Liu LN, Li JC, Lv YZ, Zong SB, Zhou J, Wang ZZ, Kou JP, Xiao W. The essential oil from the twigs of Cinnamomum cassia Presl inhibits oxytocin-induced uterine contraction in vitro and in vivo. J Ethnopharmacol, 2017, 206: 107–114.
Sun L, Liu L, Zong S, Wang Z, Zhou J, Xu Z, Ding G, Xiao W, Kou J. Traditional Chinese medicine Guizhi Fuling capsule used for therapy of dysmenorrhea via attenuating uterus contraction. J Ethnopharmacol, 2016, 191: 273–279.
Chen Y, Cao Y, Xie Y, Zhang X, Yang Q, Li X, Sun J, Qiu P, Cao W, Wang S. Traditional Chinese medicine for the treatment of primary dysmenorrhea: how do Yuanhu painkillers effectively treat dysmenorrheal. Phytomedicine, 2013, 20(12): 1095–1104.
Acknowledgments
This work was supported by National Natural Science Foundation of China (国家自然科学基金项目, No. 82004490); Hunan Provincial Innovation Foundation For Postgraduate (湖南省研究生科研创新项目, No. CX2018B515); Open Fund of the Domestic First-class Discipline Construction Project of Chinese Medicine of Hunan University of Chinese Medicine (湖南中医药大学 中医学一流学科开放基金 No. 2018ZYX05); First-level Discipline Basic Medicine Construction Project During the 13th Five-year Plan Period of Chinese Medicine of Hunan University of Chinese Medicine 湖南中医药大学“十三 五”一级学科基础医学建设项目, No. 06).
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest
The authors declare that there is no potential conflict of interest in this article.
Statement of Human and Animal Rights
The treatment of animals conformed to the ethical criteria in this experiment.
Rights and permissions
About this article
Cite this article
Liu, Y., Tang, Wj., Wang, Yq. et al. Effects of electroacupuncture on uterine prostaglandin F2α, cyclooxygenase 2 and nuclear factor κB in rats with primary dysmenorrhea. J. Acupunct. Tuina. Sci. 18, 418–424 (2020). https://doi.org/10.1007/s11726-020-1209-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11726-020-1209-5