Abstract
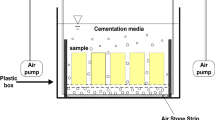
A heterotrophic Bacillus sp. strain (5C-1) was isolated from Heshang cave, an oligotrophic karst cave in the middle reaches of Yangtze River, and identified by BIOLOG and 16S rDNA sequencing. Bacterially induced formation of calcium carbonate by 5C-1 was investigated in several comparative experimental sets with or without the cell and extracellular enzymes. The temporal variations of both the amount of the precipitates and the pH values of the solution were measured by a spectrophotometer and a pH meter, respectively. The morphological characteristics of the calcium carbonate precipitates were observed with environmental scanning electronic microscopy (ESEM). The growth of 5C-1 was found to greatly promote the pH value of the liquid medium in the first 2 days, which favors the formation of calcium carbonate. No precipitates were formed with the pH value lower than 8.6, though the pH value was demonstrated to be not the only factor controlling the formation of the calcium carbonate. The accumulation of extracellular polysaccharide substance was observed to favor the precipitate formation. Only when both factors reached a threshold did the precipitates form with the addition of CaCl2. Cells and extracellular enzymes were not the factors that limit the precipitate formation in our microbial systems. The precipitates of a variety of morphological features including dumb bells, peanuts, irregular and spherical and rhombic forms were mainly observed in our microbial systems but not in the chemical control system. Interestingly, imprints of bacterial cells and spores were observed to be present on the surface of the precipitates of a peanut or a dumb bell form, probably indicative of the microbial escaping mechanism during the mineralization of calcium carbonate.
Similar content being viewed by others
References
Aloisi G, Gloter A M, Wallmann L K, Guyot F, Zuddas P (2006). Nucleation of calcium carbonate on bacterial nanoglobules. Geology, 34 (12): 1017–1020
Baumgartner L K, Reid R P, Dupraz C, Decho AW, Buckley D H, Spear J R, Przekop K M, Visscher P T (2006). Sulfate reducing bacteria in microbial mats: changing paradigms, new discoveries. Sedimentary Geology, 185: 131–145
Ben C K, Rodriguez-Navarro C, Gonzalez-Muñoz M T, Arias J M, Cultrone G, Rodriguez-Gallego M (2004). Precipitation and growth morphology of calcium carbonate induced by Myxococcus xanthus: implications for recognition of bacterial carbonates. Journal of Sedimentary Research, 74 (6): 868–876
Benzerara K, Tyliszczak T, Brown Jr G E (2006). Study of Interactions Between Microbes and Minerals by Scanning Transmission X-Ray Microscopy (STXM). SLAC-PUB-12231, Stanford, California
Bontognali T R R, Vasconcelos C, Warthmann R J, Dupraz C, Bernasconi S M, McKenzie J A(2008). Microbes produce nanobacteria-like structures, avoiding cell entombment. Geology, 36 (8): 663–666
Boquet E, Boronat A, Ramos-Cormenzana A (1973). Production of calcite (calcium carbonate) crystals by soil bacteria is a general phenomenon. Nature, 246: 527–529
Braissant O, Decho AW, Dupraz C, Glunk C, Przekop K M, Visscher P (2007). Exopolymeric substances of sulfate-reducing bacteria: Interactions with calcium at alkaline pH and implication for formation of carbonate minerals. Geobiology, 5 (4): 401–411
Dupraz C, Visscher P T, Baumgartner L K, Reid R P (2004).Microbe-mineral interactions: early carbonate precipitation in a hypersaline lake (Eleuthera Island, Bahamas). Sedimentology, 51(4): 745–765
Edwards K J, Bach W, McCollom T M (2005). Geomicrobiology in oceanography: microbe-mineral interactions at and below the seafloor. Trends in Microbiology, 13 (9): 449–456
Lower S K, Tadanier C J, Hochella M F Jr (2001). Dynamics of the Mineral-Microbe Interface: Use of Biological Force Microscopy in Biogeochemistry and Geomicrobiology. Geomicrobiology Journal, 18 (1): 63–76
Perdria J N, Warr L N, Perdrial N, Lett Marie-Claire, Elsass F (2009). Interaction between smectite and bacteria: Implications for bentonite as backfill material in the disposal of nuclear waste. Chemical Geology, 264 (1–4): 281–294
Ransom B, Bennett R H, Baerwald R, Hulbert M H, Burkett Patti-jo (1999). In situ conditions and interactions between microbes and minerals in fine-grained marine sediments: a TEM microfabric perspective. American Mineralogist, 84 (1–2): 183–192
Riding R E (2000). Microbial carbonate: the geological record of calcified algal mats and biofilms. Sedimentology, 47 (Suppl. 1): 179–214
Riding R E (2009). An atmospheric stimulus for cyanobacterial-bioinduced calcification ca. 350 million years ago? Palaios, 24 (10): 685–696
Rodriguez-Navarro C, Jimenez-Lopez C, Rodriguez-Navarro A, Gonzalez-Muñoz M T, Rodriguez-Gallego M (2007). Bacterially mediated mineralization of vaterite. Geochimica et Cosmochimica Acta, 71 (5): 1197–1213
Visscher P T, Reid R P, Bebout B M (2000). Microscale observations of sulfate reduction: correlation of microbial activity with lithified micritic laminae in modern marine stromatolites. Geology, 28 (10): 919–922
Wright D T (1999). The role of sulphate-reducing bacteria and cyanobacteria in dolomite formation in distal ephemeral lakes of the Coorong region, South Australia. Sedimentary Geolology, 126 (1–4): 147–157
Wright D T, Wacey D (2005). Precipitation of dolomite using sulphatereducing bacteria from the Coorong Region, South Australia: significance and implications. Sedimentology, 52 (5): 987–1008
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, H., Zeng, C., Liu, Q. et al. Calcium carbonate precipitation induced by a bacterium strain isolated from an oligotrophic cave in Central China. Front. Earth Sci. China 4, 148–151 (2010). https://doi.org/10.1007/s11707-010-0017-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11707-010-0017-5