Abstract
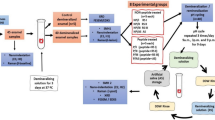
Dental caries is the most common oral disease with high incidence, widely spread and can seriously affect the health of oral cavity and the whole body. Current caries prevention measures such as fluoride treatment, antimicrobial agents, and traditional Chinese herbal, have limitations to some extent. Here we design and synthesize a novel peptide based on the amelogenin, and assess its ability to promote the remineralization of initial enamel caries lesions. We used enamel blocks to form initial lesions, and then subjected to 12-day pH cycling in the presence of peptide, NaF and HEPES buffer. Enamel treated with peptide or NaF had shallower, narrower lesions, thicker remineralized surfaces and less mineral loss than enamel treated with HEPES. This peptide can promote the remineralization of initial enamel caries and inhibit the progress of caries. It is a promising anti-caries agent with various research prospects and practical application value.
Similar content being viewed by others
References
Brambilla E. Fluoride- is it capable of fighting old and new dental diseases? An overview of existing fluoride compounds and their clinical applications. Caries Research, 2001, 35(Suppl 1): 6–9
Sheng J, Liu Z. Induction of fluoride-resistant mutant of S. mutans and the measurement of its acidogenesis in vitro. Chinese Journal of Stomatological Research, 2000, 35(2): 95–98 (in Chinese)
Li L. The biochemistry and physiology of metallic fluoride: action, mechanism, and implications. Critical Reviews in Oral Biology and Medicine, 2003, 14(2): 100–114
Dickens SH, Flaim GM, Takagi S. Mechanical properties and biochemical activity of remineralizing resin-based Ca-PO4 cements. Dental Materials, 2003, 19(6): 558–566
Van Loveren C. Sugar alcohols: what is the evidence for cariespreventive and caries-therapeutic effects? Caries Research, 2004, 38(3): 286–293
Wang XJ, Huang H, Yang F, et al. Ectopic study of tissueengineered bone complex with enamel matrix proteins, bone marrow stromal cells in porous calcium phosphate cement scaffolds, in nude mice. Cell Proliferation, 2011, 44(3): 274–282
Mrozik KM, Gronthos S, Menicanin D, et al. Effect of coating Straumann® Bone Ceramic with Emdogain on mesenchymal stromal cell hard tissue formation. Clinical Oral Investigations, 2012, 16(3): 867–878
Rathe F, Junker R, Chesnutt BM, et al. The effect of enamel matrix derivative (Emdogain) on bone formation: a systematic review. Tissue Engineering Part B: Reviews, 2009, 15(3): 215–224
Suzuki S, Nagano T, Yamakoshi Y, et al. Enamel matrix derivative gel stimulates signal transduction of BMP and TGF-β. Journal of Dental Research, 2005, 84(6): 510–514
Windisch P, Sculean A, Klein F, et al. Comparison of clinical, radiographic, and histometric measurements following treatment with guided tissue regeneration or enamel matrix proteins in human periodontal defects. Journal of Periodontology, 2002, 73(4): 409–417
Zhang L, Zou L, Li J, et al. Effect of enamel organic matrix on the potential of Galla chinensis to promote the remineralization of initial enamel carious lesions in vitro. Biomedical Materials, 2009, 4(3): 034102
Zhang L, Xue J, Li J, et al. Effects of Galla chinensis on inhibition of demineralization of regular bovine enamel or enamel disposed of organic matrix. Archives of Oral Biology, 2009, 54(9): 817–822
Chen H, Clarkson BH, Sun K, et al. Self-assembly of synthetic hydroxyapatite nanorods into an enamel prism-like structure. Journal of Colloid and Interface Science, 2005, 288(1): 97–103
Yamagishi K, Onuma K, Suzuki T, et al. Materials chemistry: a synthetic enamel for rapid tooth repair. Nature, 2005, 433(7028): 819
Wang ZW, Zhao YP, Zhou CR, et al. The study on the enamel remineralization by enamel matrix proteins’ inducing. Journal of Sichuan University (Medical Science Edition), 2008, 39(4): 579–582 (in Chinese)
Ishizaki NT, Matsumoto K, Kimura Y, et al. Histopathological study of dental pulp tissue capped with enamel matrix derivative. Journal of Endodontics, 2003, 29(3): 176–179
Xiang C, Ran J, Yang Q, et al. Effects of enamel matrix derivative on remineralization of initial enamel carious lesions in vitro. Archives of Oral Biology, 2013, 58(4): 362–369
Ran JM, Ieong CC, Xiang CY, et al. In vitro inhibition of bovine enamel demineralization by enamel matrix derivative. Scanning, doi: 10.1002/sca.21085
Moradian-Oldak J. Amelogenins: assembly, processing and control of crystal morphology. Matrix Biology, 2001, 20(5–6): 293–305
Du C, Falini G, Fermani S, et al. Supramolecular assembly of amelogenin nanospheres into birefringent microribbons. Science, 2005, 307(5714): 1450–1454
Fincham AG, Moradian-Oldak J, Diekwisch TG, et al. Evidence for amelogenin “nanospheres” as functional components of secretory-stage enamel matrix. Journal of Structural Biology, 1995, 115(1): 50–59
Moradian-Oldak J. Protein-mediated enamel mineralization. Frontiers in Bioscience, 2012, 17(7): 1996–2023
Fan Y, Sun Z, Moradian-Oldak J. Controlled remineralization of enamel in the presence of amelogenin and fluoride. Biomaterials, 2009, 30(4): 478–483
Fan Y, Nelson JR, Alvarez JR, et al. Amelogenin-assisted ex vivo remineralization of human enamel: Effects of supersaturation degree and fluoride concentration. Acta Biomaterialia, 2011, 7(5): 2293–2302
Porter SM. Seawater chemistry and early carbonate biomineralization. Science, 2007, 316(5829): 1302
Paine ML, Snead ML. Tooth developmental biology: disruptions to enamel-matrix assembly and its impact on biomineralization. Orthodontics & Craniofacial Research, 2005, 8(4): 239–251
Ieong CC, Zhou XD, Li JY, et al. Possibilities and potential roles of the functional peptides based on enamel matrix proteins in promoting the remineralization of initial enamel caries. Medical Hypotheses, 2011, 76(3): 391–394
Cate JMT, Duijsters PP. Influence of fluoride in solution on tooth demineralization. I. Chemical data. Caries Research, 1983, 17(3): 193–199
White DJ. Reactivity of fluoride dentifrices with artificial caries. I. Effects on early lesions: F uptake, surface hardening and remineralization. Caries Research, 1987, 21(2): 126–140
Snead ML, Zhu DH, Lei Y, et al. A simplified genetic design for mammalian enamel. Biomaterials, 2011, 32(12): 3151–3157
Lacruz RS, Smith CE, Bringas P Jr, et al. Identification of novel candidate genes involved in mineralization of dental enamel by genome-wide transcript profiling. Journal of Cellular Physiology, 2012, 227(5): 2264–2275
Aoba T, Tanabe T, Moreno EC. Proteins in the enamel fluid of immature porcine teeth. Journal of Dental Research, 1987, 66(12): 1721–1726
Iijima M, Moradian-Oldak J. Control of apatite crystal growth in a fluoride containing amelogenin-rich matrix. Biomaterials, 2005, 26(13): 1595–1603
Yamakoshi Y, Hu JC C, Ryu OH, et al. A comprehensive strategy for purifying pig enamel proteins in biomineralization: formation, diversity, evolution and application. In: Proceedings of the 8th International Symposium on Biomineralization. Hadano: Tokai University Press, 2003, 326–332
Luo JJ, Ning TY, Cao Y, et al. Biomimic enamel remineralization by hybridization calcium- and phosphate-loaded liposomes with amelogenin-inspired peptide. Key Engineering Materials, 2012, 512–515: 1727–1730
Roy MD, Stanley SK, Amis EJ, et al. Identification of a highly specific hydroxyapatite-binding peptide using phage display. Advanced Materials, 2008, 20(10): 1830–1836
Ravindranath HH, Chen LS, Zeichner-David M, et al. Interaction between the enamel matrix proteins amelogenin and ameloblastin. Biochemical and Biophysical Research Communications, 2004, 323(3): 1075–1083
Beniash E, Simmer JP, Margolis HC. The effect of recombinant mouse amelogenins on the formation and organization of hydroxyapatite crystals in vitro. Journal of Structural Biology, 2005, 149(2): 182–190
Paine ML, Wang HJ, Luo W, et al. A transgenic animal model resembling amelogenesis imperfecta related to ameloblastin overexpression. The Journal of Biological Chemistry, 2003, 278(21): 19447–19452
Habelitz S, DenBesten PK, Marshall SJ, et al. Self-assembly and effect on crystal growth of the leucine-rich amelogenin peptide. European Journal of Oral Sciences, 2006, 114(Suppl 1): 315–319
Moradian-Oldak J, Tan J, Fincham AG. Interaction of amelogenin with hydroxyapatite crystals: an adherence effect through amelogenin molecular self-association. Biopolymers, 1998, 46 (4): 225–238
Wallwork ML, Kirkham J, Chen H, et al. Binding of dentin noncollagenous matrix proteins to biological mineral crystals: an atomic force microscopy study. Calcified Tissue International, 2002, 71(3): 249–256
Müller H, Zentel R, Janshoff A, et al. Control of CaCO3 crystallization by demixing of monolayers. Langmuir, 2006, 22(26): 11034–11040
Bagheri HG, Sadr A, Espigares J, et al. Leucine rich amelogenin peptide improves the remineralization of enamel lesions. Dental Materials, 2014, 30(suppl): e172–e173
Tartaix PH, Doulaverakis M, George A, et al. In vitro effects of dentin matrix protein-1 on hydroxyapatite formation provide insights into in vivo functions. The Journal of Biological Chemistry, 2004, 279(18): 18115–18120
He G, Dahl T, Veis A, et al. Nucleation of apatite crystals in vitro by self-assembled dentin matrix protein 1. Nature Materials, 2003, 2(8): 552–558
Aggeli A, Bell M, Boden N, et al. Self-assembling peptide polyelectrolyte beta-sheet complexes form nematic hydrogels. Angewandte Chemie International Edition in English, 2003, 42(45): 5603–5606
Kirkham J, Firth A, Vernals D, et al. Self-assembling peptide scaffolds promote enamel remineralization. Journal of Dental Research, 2007, 86(5): 426–430
Hartgerink JD, Beniash E, Stupp SI. Self-assembly and mineralization of peptide-amphiphile nanofibers. Science, 2001, 294(5547): 1684–1688
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, D., Lv, X., Tu, H. et al. Remineralization of initial enamel caries in vitro using a novel peptide based on amelogenin. Front. Mater. Sci. 9, 293–302 (2015). https://doi.org/10.1007/s11706-015-0298-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11706-015-0298-4