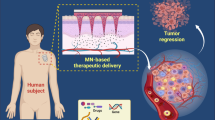
Abstract
Benefiting from the advantage of taking place in biological environments without interfering with an innate biochemical process, the bioorthogonal reaction that commonly contains the “bond formation” and “bond cleavage” system has been widely used in targeted therapy for a variety of tumors. Herein, several prominent cases based on the bioorthogonal reaction that tailoring the metabolic glycoengineering tactics to modified cells for cancer immunotherapy, and the innovative tactics for reducing the metal ions’ toxic and side effects with microneedle patches will be highlighted. Based on these applications, the complexities, potential pitfalls, and opportunities of bioorthogonal chemistry in future cancer therapy will be evaluated.

Similar content being viewed by others
References
Li L, Wang J, Kong H, Zeng Y, Liu G. Functional biomimetic nanoparticles for drug delivery and theranostic applications in cancer treatment. Science and Technology of Advanced Materials, 2018, 19(1): 771–790
Lu Y, Aimetti A A, Langer R, Gu Z. Bioresponsive materials. Nature Reviews Materials, 2017, 2(1): 16075
Hanahan D, Weinberg R A. Hallmarks of cancer: the next generation. Cell, 2011, 144(5): 646–674
Liang T X Z, Chen Z W, Li H J, Gu Z. Bioorthogonal catalysis for biomedical applications. Trends in Chemistry, 2022, 4(2): 157–168
Taiariol L, Chaix C, Farre C, Moreau E. Click and bioorthogonal chemistry: the future of active targeting of nanoparticles for nanomedicines? Chemical Reviews, 2022, 122(1): 340–384
Bird R, Lemmel S, Yu X, Zhou Q. Bioorthogonal chemistry and its applications. Bioconjugate Chemistry, 2021, 32(12): 2457–2479
Kostenkova K, Scalese G, Gambino D, Crans D C. Highlighting the roles of transition metals and speciation in chemical biology. Current Opinion in Chemical Biology, 2022, 69: 102155
Gurruchaga-Pereda J, Martínez-Martínez V, Rezabal E, Lopez X, Garino C, Mancin F, Cortajarena A L, Salassa L. Flavin bioorthogonal photocatalysis toward platinum substrates. ACS Catalysis, 2020, 10(1): 187–196
Deb T, Tu J, Franzini R M. Mechanisms and substituent effects of metal-free bioorthogonal reactions. Chemical Reviews, 2021, 121(12): 6850–6914
Taylor M T, Blackman M L, Dmitrenko O, Fox J M. Design and synthesis of highly reactive dienophiles for the tetrazine-trans-cyclooctene ligation. Journal of the American Chemical Society, 2011, 133(25): 9646–9649
Bednarek C, Wehl I, Jung N, Schepers U, Bräse S. The staudinger ligation. Chemical Reviews, 2020, 120(10): 4301–4354
Li J, Chen P R. Development and application of bond cleavage reactions in bioorthogonal chemistry. Nature Chemical Biology, 2016, 12(3): 129–137
Wang H, Mooney D J. Metabolic glycan labelling for cancer-targeted therapy. Nature Chemistry, 2020, 12(12): 1102–1114
Thirumurugan P, Matosiuk D, Jozwiak K. Click chemistry for drug development and diverse chemical-biology applications. Chemical Reviews, 2013, 113(7): 4905–4979
Ren E, Liu C, Lv P, Wang J, Liu G. Genetically engineered cellular membrane vesicles as tailorable shells for therapeutics. Advanced Science, 2021, 8(21): 2100460
Soriano del Amo D, Wang W, Jiang H, Besanceney C, Yan A C, Levy M, Liu Y, Marlow F L, Wu P. Biocompatible copper(I) catalysts for in vivo imaging of glycans. Journal of the American Chemical Society, 2010, 132(47): 16893–16899
Sletten E M, Bertozzi C R. From mechanism to mouse: a tale of two bioorthogonal reactions. Accounts of Chemical Research, 2011, 44(9): 666–676
Völker T, Meggers E. Transition-metal-mediated uncaging in living human cells—an emerging alternative to photolabile protecting groups. Current Opinion in Chemical Biology, 2015, 25: 48–54
Rakhit R, Navarro R, Wandless T J. Chemical biology strategies for posttranslational control of protein function. Chemistry & Biology, 2014, 21(9): 1238–1252
Yusop R M, Unciti-Broceta A, Johansson E M V, Sánchez-Martín R M, Bradley M. Palladium-mediated intracellular chemistry. Nature Chemistry, 2011, 3(3): 239–243
Li J, Yu J T, Zhao J Y, Wang J, Zheng S Q, Lin S X, Chen L, Yang M Y, Jia S, Zhang X Y, Chen P R. Palladium-triggered deprotection chemistry for protein activation in living cells. Nature Chemistry, 2014, 6(4): 352–361
Weiss J T, Dawson J C, Macleod K G, Rybski W, Fraser C, Torres-Sánchez C, Patton E E, Bradley M, Carragher N O, Unciti-Broceta A. Extracellular palladium-catalysed dealkylation of 5-fluoro-1-propargyl-uracil as a bioorthogonally activated prodrug approach. Nature Communications, 2014, 5(1): 3277
Yang W, Nan H X, Xu Z F, Huang Z X, Chen S, Li J Y, Li J, Yang H H. DNA-templated glycan labeling for monitoring receptor spatial distribution in living cells. Analytical Chemistry, 2021, 93(36): 12265–12272
Hu Q Y, Sun W J, Wang J Q, Ruan H T, Zhang X D, Ye Y Q, Shen S, Wang C, Lu W Y, Cheng K, Dotti G, Zeidner J F, Wang J, Gu Z. Conjugation of haematopoietic stem cells and platelets decorated with anti-PD-1 antibodies augments anti-leukaemia efficacy. Nature Biomedical Engineering, 2018, 2(11): 831–840
Pawlak J B, Gential G P P, Ruckwardt T J, Bremmers J S, Meeuwenoord N J, Ossendorp F A, Overkleeft H S, Filippov D V, van Kasteren S I. Bioorthogonal deprotection on the dendritic cell surface for chemical control of antigen cross-presentation. Angewandte Chemie International Edition, 2015, 54(19): 5628–5631
Wu D, Yang K K, Zhang Z K, Feng Y X, Rao L, Chen X Y, Yu G C. Metal-free bioorthogonal click chemistry in cancer theranostics. Chemical Society Reviews, 2022, 51(4): 1336–1376
Völker T, Dempwolff F, Graumann P L, Meggers E. Progress towards bioorthogonal catalysis with organometallic compounds. Angewandte Chemie International Edition, 2014, 53(39): 10536–10540
Lim R K, Lin Q. Photoinducible bioorthogonal chemistry: a spatiotemporally controllable tool to visualize and perturb proteins in live cells. Accounts of Chemical Research, 2011, 44(9): 828–839
Chang P V, Prescher J A, Sletten E M, Baskin J M, Miller I A, Agard N J, Lo A, Bertozzi C R. Copper-free click chemistry in living animals. Proceedings of the National Academy of Sciences of the United States of America, 2010, 107(5): 1821–1826
Laughlin S T, Bertozzi C R. Metabolic labeling of glycans with azido sugars and subsequent glycan-profiling and visualization via Staudinger ligation. Nature Protocols, 2007, 2(11): 2930–2944
Li W J, Pan H, He H M, Meng X Q, Ren Q, Gong P, Jiang X, Liang Z G, Liu L L, Zheng M B, Shao X, Ma Y, Cai L. Bioorthogonal T cell targeting strategy for robustly enhancing cytotoxicity against tumor cells. Small, 2019, 15(4): e1804383
Prescher J A, Bertozzi C R. Chemical technologies for probing glycans. Cell, 2006, 126(5): 851–854
Ren E, Chu C C, Zhang Y M, Wang J Q, Pang X, Lin X N, Liu C, Shi X X, Dai Q X, Lv P, Wang X, Chen X, Liu G. Mimovirus vesicle-based biological orthogonal reaction for cancer diagnosis. Small Methods, 2020, 4(9): 2000291
Wang H, Wang R B, Cai K M, He H, Liu Y, Yen J, Wang Z Y, Xu M, Sun Y W, Zhou X, Yin Q, Tang L, Dobrucki I T, Dobrucki L W, Chaney E J, Boppart S A, Fan T M, Lezmi S, Chen X, Yin L, Cheng J. Selective in vivo metabolic cell-labeling-mediated cancer targeting. Nature Chemical Biology, 2017, 13(4): 415–424
Shim M K, Yoon H Y, Ryu J H, Koo H, Lee S, Park J H, Kim J H, Lee S, Pomper M G, Kwon I C, Kim K. Cathepsin B-specific metabolic precursor for in vivo tumor-specific fluorescence imaging. Angewandte Chemie International Edition, 2016, 55(47): 14698–14703
Xie R, Dong L, Huang R B, Hong S L, Lei R X, Chen X. Targeted imaging and proteomic analysis of tumor-associated glycans in living animals. Angewandte Chemie International Edition, 2014, 53(51): 14082–14086
Wang H, Gauthier M, Kelly J R, Miller R J, Xu M, O’Brien W D Jr, Cheng J J. Targeted ultrasound-assisted cancer-selective chemical labeling and subsequent cancer imaging using click chemistry. Angewandte Chemie International Edition, 2016, 55(18): 5452–5456
Wang H, Sobral M C, Zhang D K Y, Cartwright A N, Li A W, Dellacherie M O, Tringides C M, Koshy S T, Wucherpfennig K W, Mooney D J. Metabolic labeling and targeted modulation of dendritic cells. Nature Materials, 2020, 19(11): 1244–1252
Chen Z W, Li H J, Bian Y J, Wang Z J, Chen G J, Zhang X D, Miao Y M, Wen D, Wang J Q, Wan G, Zeng Y, Abdou P, Fang J, Li S, Sun C J, Gu Z. Bioorthogonal catalytic patch. Nature Nanotechnology, 2021, 16(8): 933–941
Yu J C, Wang J Q, Zhang Y Q, Chen G J, Mao W W, Ye Y Q, Kahkoska A R, Buse J B, Langer R, Gu Z. Glucose-responsive insulin patch for the regulation of blood glucose in mice and minipigs. Nature Biomedical Engineering, 2020, 4(5): 499–506
Wang C Q, Zhang H, Zhang T, Zou X Y, Wang H, Rosenberger J E, Vannam R, Trout W S, Grimm J B, Lavis L D, Thorpe C, Jia X, Li Z, Fox J M. Enabling in vivo photocatalytic activation of rapid bioorthogonal chemistry by repurposing silicon-rhodamine fluorophores as cytocompatible far-red photocatalysts. Journal of the American Chemical Society, 2021, 143(28): 10793–10803
Acknowledgments
This work was supported by the Major State Basic Research Development Program of China (Grant No. 2017YFA0205201) and the National Natural Science Foundation of China (Grant Nos. 81925019 and U1705281).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Lin, L., Jiang, L., Ren, E. et al. Bioorthogonal chemistry based on-demand drug delivery system in cancer therapy. Front. Chem. Sci. Eng. 17, 483–489 (2023). https://doi.org/10.1007/s11705-022-2227-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11705-022-2227-2