Abstract
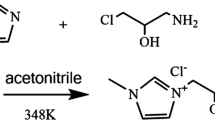
The coupling reaction of propylene and CO2 to form propylene carbonate (PC) was promoted by an ionic liquid (IL) covalently bound to polyethylene glycol (PEG). The supported ionic liquid, which has both acidic and basic components, proved to be an active catalyst for PC synthesis under mild conditions. The effects of different cations and anions, reaction temperature, CO2 pressure, and reaction time were investigated. It was demonstrated that the acid group in the catalyst plays an important role in the reaction. With this system, a high PC yield (95%) was achieved under mild conditions (3.0 MPa, 120°C and 4 h) without a co-solvent. In addition, the catalyst was readily recovered and reused. Based on the experimental results, a plausible mechanism for the catalyst was proposed.
Similar content being viewed by others
References
Sakakura T, Choi J C, Yasuda H. Transformation of carbon dioxide. Chemical Reviews, 2007, 107(6): 2365–2387
Dai W L, Luo S L, Yin S F, Au C T. The direct transformation of carbon dioxide to organic carbonates over heterogeneous catalysts. Applied Catalysis A: General, 2009, 366(1): 2–12
Zhang S J, Chen Y H, Li F W, Lu X M, Dai W B, Mori R. Fixation and conversion of CO2 using ionic liquids. Catalysis Today, 2006, 115(1–4): 61–69
Sako T, Fukai T, Sahashi R, Sone M, Matsuno M. Cycloaddition of oxirane group with carbon dioxide in the supercritical homogeneous state. Industrial & Engineering Chemistry Research, 2002, 41(22): 5353–5358
Caló V, Nacci A, Monopoli A, Fanizzi A. Cyclic carbonate formation from carbon dioxide and oxiranes in tetrabutylammonium halides as solvents and catalysts. Organic Letters, 2002, 4(15): 2561–2563
Chang T, Jing HW, Jin L L, Qiu WY. Quaternary onium tribromide catalyzed cyclic carbonate synthesis from carbon dioxide and epoxides. Journal of Molecular Catalysis A: Chemical, 2007, 264(1–2): 241–247
He L N, Yasuda T, Sakakura T. New procedure for recycling homogeneous catalyst: propylene carbonate synthesis under supercritical CO2. Green Chemistry, 2003, 5(1): 92–94
Jiang J L, Gao F X, Hua R M, Qiu X Q. Re(CO)5Br-catalyzed coupling of epoxides with CO2 affording cyclic carbonates under solvent-free conditions. Journal of Organic Chemistry, 2005, 70(1): 381–383
Kim H S, Kim J J, Kwon H N, Chung M J, Lee B G, Jang H G. Welldefined highly active heterogeneous catalyst system for the coupling reactions of carbon dioxide and epoxides. Journal of Catalysis, 2002, 205(1): 226–229
Kawanami H, Sasaki A, Matsui K, Ikushima Y. A rapid and effective synthesis of propylene carbonate using a supercritical CO2-ionic liquid system. Chemical Communications, 2003, 2003(7): 896–897
Wang J Q, Yue X D, Cai F, He L N. Solventless synthesis of cyclic carbonates from carbon dioxide and epoxides catalyzed by silicasupported ionic liquids under supercritical conditions. Catalysis Communications, 2007, 8(2): 167–172
Peng J J, Deng Y Q. Cycloaddition of carbon dioxide to propylene oxide catalyzed with ionic liquid. New Journal of Chemistry, 2001, 25(4): 639–641
Yang H Z, Gu Y L, Deng Y Q, Shi F. Electrochemical activation of carbon dioxide in ionic liquid: synthesis of cyclic carbonates at mild reaction conditions. Chemical Communications, 2002, 2002(3): 274–275
Kim H S, Kim J J, Kim H, Jang H G. Imidazolium zinc tetrahalide-catalyzed coupling reaction of CO2 and ethylene oxide or propylene oxide. Journal of Catalysis, 2003, 220(1): 44–46
Li F, Xiao L, Xia C, Hu B. Chemical fixationof CO2 with highly efficient ZnCl2/[BMIm]Br catalyst system. Tetrahedron Letters, 2004, 45(45): 8307–8310
Sun J M, Fujita S I, Arai M. Development in the green synthesis of cyclic carbonate from carbon dioxide using ionic liquids. Journal of Organometallic Chemistry, 2005, 690(15): 3490–3497
Xiao L F, Li F W, Peng J J, Xia C G. Immobilized ionic liquid zinc chloride: heterogeneous catalyst for synthesis of cyclic carbonates from carbon dioxide and epoxides. Journal of Molecular Catalysis A: Chemical, 2006, 253(1–2): 265–269
Sun J, Zhang S J, Cheng W G, Ren G Y. Hydroxyl-functionalized ionic liquid: a novel efficient catalyst for chemical fixation of CO2 to cyclic carbonate. Tetrahedron Letters, 2008, 49(22): 3588–3591
Kim Y J, Varma R S. Tetrahaloindate(III)-based ionic liquids in the coupling reaction of carbon dioxide and epoxides to generate cyclic carbonates: H-bonding and mechanistic studies. Journal of Organic Chemistry, 2005, 70(20): 7882–7891
Tian J S, Miao C X, Wang J Q, Cai F, Du Y, Zhao Y, He L N. Efficient synthesis of dimethy carbonate from methanol, propylene oxide and CO2 catalyzed by recyclable inorganic base/phosphonium halide-functionalized polyethylene glycol. Green Chemistry, 2007, 9(6): 566–571
Dai W L, Chen L, Yin S F, Luo S L, Au C T. 3-(2-Hydroxyl-ethyl)-1-propylimidazolium bromide immobilized on SBA-15 as efficient catalyst for the synthesis of cyclic carbonates via the coupling of carbon dioxide with epoxides. Catalysis Letters, 2010, 135(3–4): 295–304
Wang J Q, Kong D L, Chen J Y, Cai F, He L N. Synthesis of cyclic carbonates from epoxides and carbon dioxide over silica-supported quaternary ammonium salts under supercritical conditions. Journal of Molecular Catalysis A: Chemical, 2006, 249(1–2): 143–148
Du Y, Wang J Q, Chen J Y, Cai F, Tian J S, Kong D L, He L N. A poly(ethylene glycol)-supported quaternary ammonium salt for highly efficient and environmentally friendly chemical fixation of CO2 with epoxides under supercritical conditions. Tetrahedron Letters, 2006, 47(8): 1271–1275
Sun J, Cheng W G, Fan W, Wang Y H, Meng Z Y, Zhang S J. Reusable and efficient polymer-supported task-specific ionic liquid catalyst for cycloaddition of epoxide with CO2. Catalysis Today, 2009, 148(3–4): 361–367
Ulusoy M, Cetinkaya E, Cetinkaya B. Conversion of carbon dioxide to cyclic carbonates using diimine Ru(II) complexes as catalysts. Applied Organometallic Chemistry, 2009, 23(2): 68–74
Xie Y, Zhang Z F, Jiang T, He J L, Han B X, Wu T B, Ding K L. CO2 cycloaddition reactions catalyzed by an ionic liquid grafted onto a highly cross-linked polymer matrix. Angewandte Chemie International Edition, 2007, 46(38): 7255–7258
Heldebrant D J, Witt H N, Walsh S M, Ellis T, Rauscher J, Jessop P G. Liquid polymers as solvents for catalytic reductions. Green Chemistry, 2006, 8(9): 807–815
Gourgouillon D, Avelino H J, Fareleira J, Ponte M N. Simultaneous viscosity and density measurement of supercritical CO2-satureted PEG 400. Journal of Supercritical Fluids, 1998, 13(1–3): 177–185
Harrison K L, Johnston K P, Sanchez I C. Effect of surfactants on the interfacial tension between supercritical carbonate dioxide and polyethylene glocol. Langmuir, 1996, 12(11): 2637–2644
Dariva C, Coelho L A F, Oliveira J V. A kinetic approach for predicting diffusivities in dense fluid mixtures. Fluid Phase Equilibria, 1999, 158–160(b): 1045–1054
Kawanami H, Sasaki A, Matsui K, Ikushima Y. A rapid and effective synthesis of propylene carbonate using a supercritical CO2-ionic liquid system. Chemical Communications, 2003, 7(7): 896–897
Nomura R, Kimura M, Teshima S, Ninagawa A, Matsuda H. Directsynthesis of cyclic carbonates in the presence of organometallic compounds. Catalyses by systems from IVA, VA, and VIA group compounds and Lewis base. Bulletin of the Chemical Society of Japan, 1982, 55(10): 3200–3203
Udayakumar S, Lee M K, Shim H L, Park D W. Functionalization of organic ions on hybrid MCM-41 for cycloaddition reaction: the effective conversion of carbon dioxide. Applied Catalysis A: General, 2009, 365(1): 88–95
Udayakumar S, Park S W, Park D W, Choi B S. Immobilization of ionic liquid on hybrid MCM-41 system for the chemical fixation of carbon dioxide on cyclic carbonate. Catalysis Communications, 2008, 9(7): 1563–1570
Zhu A L, Jiang T, Han B X, Zhang J C, Xie Y, Ma X M. Supported choline chloride/urea as a heterogeneous catalyst for chemical fixation of carbon dioxide to cyclic carbonates. Green Chemistry, 2007, 9(2): 169–172
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yao, R., Wang, H. & Han, J. Polyethylene glycol-supported ionic liquid as a highly efficient catalyst for the synthesis of propylene carbonate under mild conditions. Front. Chem. Sci. Eng. 6, 239–245 (2012). https://doi.org/10.1007/s11705-012-1297-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11705-012-1297-y