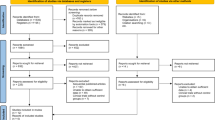
Abstract
Robot-assisted (RA) technology has been shown to be a safe aid in spine surgery, this meta-analysis aims to compare surgical parameters and clinical indexes between robot-assisted cortical bone trajectory (CBT) and fluoroscopy-assisted (FA) cortical bone trajectory in spinal surgery. We searched databases such as PubMed, Web of Science, the Cochrane Library, and the China National Knowledge Infrastructure. The study selection process was guided by the PICOS (Patient/Problem, Intervention, Comparison, Outcome, Study Design) strategy. The risk of bias in non-randomized comparative studies was assessed using the risk of bias in non-randomized studies of interventions (ROBINS-I) tool. We performed this meta-analysis using RevMan 5.3 software (Cochrane Collaboration, Copenhagen, Denmark), and the level of statistical significance was set at P < 0.05. Six articles involving 371 patients and 1535 screws were included in this meta-analysis. RA-CBT outperformed FA-CBT in terms of various parameters, such as accuracy of pedicle screw position (both Gertzbein-Robbins scale and Ding scale), avoidance of superior facet joint violation (FJV), and reduction of neurological injury. Our meta-analysis offered a thorough evaluation of the efficacy and safety of RA-CBT in spinal surgery. The findings revealed that RA-CBT produced statistically significant results in terms of pedicle screw position accuracy and superior facet joint violation prevention. In terms of surgical parameters and clinical indexes, future research and clinical practice should investigate the efficacy of RA-CBT further. The study was registered in the PROSPERO (CRD42023466280).






Similar content being viewed by others
Data availability
The data sets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- RA:
-
Robot-assisted
- FA:
-
Fluoroscopy-assisted
- CBT:
-
Cortical bone trajectory
- BMI:
-
Body mass index
- BMD:
-
Bone mineral density
- ASD:
-
Adjacent segmental degeneration
- QCT:
-
Quantitative computed tomography
- RCT:
-
Randomized controlled trial
- WMD:
-
Weighted mean difference
- SMD:
-
Standardized mean difference
- CI:
-
Confidence interval
- OR:
-
Odds ratio
- M-H:
-
Mantel–Haenszel
- IV:
-
Inverse variance
- Df:
-
Degrees of freedom
References
Santoni BG, Hynes RA, McGilvray KC et al (2009) Cortical bone trajectory for lumbar pedicle screws. Spine J 9:366–373. https://doi.org/10.1016/j.spinee.2008.07.008
Kim KT, Song MG, Park YJ et al (2022) Cortical trajectory screw fixation in lumbar spine surgery: a review of the existing literature. Asian Spine J 16:127–140. https://doi.org/10.31616/asj.2020.0575
Cofano F, Marengo N, Ajello M et al (2020) The era of cortical bone trajectory screws in spine surgery: a qualitative review with rating of evidence. World Neurosurg 134:14–24. https://doi.org/10.1016/j.wneu.2019.10.079
Wang J, He X, Sun T (2019) Comparative clinical efficacy and safety of cortical bone trajectory screw fixation and traditional pedicle screw fixation in posterior lumbar fusion: a systematic review and meta-analysis. Eur Spine J 28:1678–1689. https://doi.org/10.1007/s00586-019-05999-y
Zhang T, Guo N, Chen T et al (2019) Comparison of outcomes between cortical screws and traditional pedicle screws for lumbar interbody fusion: a systematic review and meta-analysis. J Orthop Surg Res 14:269. https://doi.org/10.1186/s13018-019-1311-x
Guo S, Zhu K, Yan MJ et al (2022) Cortical bone trajectory screws in the treatment of lumbar degenerative disc disease in patients with osteoporosis. World J Clin Cases 10:13179–13188. https://doi.org/10.12998/wjcc.v10.i36.13179
Hung CW, Wu MF, Hong RT et al (2016) Comparison of multifidus muscle atrophy after posterior lumbar interbody fusion with conventional and cortical bone trajectory. Clin Neurol Neurosurg 145:41–45. https://doi.org/10.1016/j.clineuro.2016.03.005
Zheng Z, Zhang L, Zhu Y et al (2023) Percutaneous cortical bone trajectory screw fixation versus traditional open pedicle screw fixation for type A thoracolumbar fractures without neurological deficit. J Robot Surg 17:233–241. https://doi.org/10.1007/s11701-022-01426-5
Lopez IB, Benzakour A, Mavrogenis A et al (2023) Robotics in spine surgery: systematic review of literature. Int Orthop 47:447–456. https://doi.org/10.1007/s00264-022-05508-9
Zhang HQ, Wang CC, Zhang RJ et al (2022) Predictors of accurate intrapedicular screw placement in single-level lumbar (L4–5) fusion: robot-assisted pedicle screw, traditional pedicle screw, and cortical bone trajectory screw insertion. BMC Surg 22:284. https://doi.org/10.1186/s12893-022-01733-6
Khan A, Rho K, Mao JZ et al (2020) Comparing cortical bone trajectories for pedicle screw insertion using robotic guidance and three-dimensional computed tomography navigation. World Neurosurg 141:e625–e632. https://doi.org/10.1016/j.wneu.2020.05.257
Ueno J, Akazawa T, Torii Y et al (2022) Accuracy and screw insertion time of robotic-assisted cortical bone trajectory screw placement for posterior lumbar interbody fusion: a comparison of early, middle, and late phases. Cureus 14:e32574. https://doi.org/10.7759/cureus.32574
Page MJ, McKenzie JE, Bossuyt PM et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. PLoS Med 18:e1003583. https://doi.org/10.1371/journal.pmed.1003583
Gertzbein SD, Robbins SE (1990) Accuracy of pedicular screw placement in vivo. Spine (Phila Pa 1976) 15:11–14
Ding H, Han B, Hai Y et al (2021) The feasibility of assessing the cortical bone trajectory screw placement accuracy using a traditional pedicle screw insertion evaluation system. Clin Spine Surg 34:E112–E120. https://doi.org/10.1097/BSD.0000000000001059
Yson SC, Sembrano JN, Sanders PC et al (2013) Comparison of cranial facet joint violation rates between open and percutaneous pedicle screw placement using intraoperative 3-D CT (O-arm) computer navigation. Spine (Phila Pa 1976) 38:E251-258
Moshirfar A, Jenis LG, Spector LR et al (2006) Computed tomography evaluation of superior-segment facet-joint violation after pedicle instrumentation of the lumbar spine with a midline surgical approach. Spine (Phila Pa 1976) 31:2624–2629
Sterne JA, Hernan MA, Reeves BC et al (2016) ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 355:i4919. https://doi.org/10.1136/bmj.i4919
Guyatt GH, Oxman AD, Schunemann HJ et al (2011) GRADE guidelines: a new series of articles in the Journal of Clinical Epidemiology. J Clin Epidemiol 64:380–382. https://doi.org/10.1016/j.jclinepi.2010.09.011
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558. https://doi.org/10.1002/sim.1186
Zhang XN, Liu YZ, Li Y et al (2023) Clinical comparative study of robot-assisted and fluoroscopy-assisted free-hand cortical bone trajectory screw placement in the treatment of single-level degenerative lumbar diseases. J Cap Med Univ 44:836–844. https://doi.org/10.3969/j.issn.1006-7795.2023.05.019
Lin S, Hu J, Wan L et al (2022) Orthopedic robot-assisted cortical bone trajectory screw internal fixation for the treatment of lumbar degenerative diseases. Chin J Tissue Eng Res 26:2356–2360. https://doi.org/10.12307/2022.590
Ma S, Liu B, Li N et al (2023) Comparison of robot-assisted and free-hand cortical bone trajectory screw in lumbar spinal surgery for senile osteoporosis. Chin J Tissue Eng Res 27:4332–4336. https://doi.org/10.12307/2023.399
Chen H, Liu S, Zhang J et al (2020) A comparative study on treatment of lumbar degenerative disease with osteoporosis by manual and robot-assisted cortical bone trajectory screws fixation. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 34:1142–1148. https://doi.org/10.7507/1002-1892.202001070
Li Y, Chen L, Liu Y et al (2022) Accuracy and safety of robot-assisted cortical bone trajectory screw placement: a comparison of robot-assisted technique with fluoroscopy-assisted approach. BMC Musculoskelet Disord 23:328. https://doi.org/10.1186/s12891-022-05206-y
Le X, Tian W, Shi Z et al (2018) Robot-assisted versus fluoroscopy-assisted cortical bone trajectory screw instrumentation in lumbar spinal surgery: a matched-cohort comparison. World Neurosurg 120:e745–e751. https://doi.org/10.1016/j.wneu.2018.08.157
Inoue T, Mizutamari M, Hatake K (2021) Surgical invasiveness of single-segment posterior lumbar interbody fusion: comparing perioperative blood loss in posterior lumbar interbody fusion with traditional pedicle screws, cortical bone trajectory screws, and percutaneous pedicle screws. Asian Spine J 15:856–864. https://doi.org/10.31616/asj.2020.0296
Joseph JR, Smith BW, Liu X et al (2017) Current applications of robotics in spine surgery: a systematic review of the literature. Neurosurg Focus 42:E2. https://doi.org/10.3171/2017.2.FOCUS16544
Shi W, Aini M, Dang L et al (2022) Feasibility and improvement of a three-dimensional printed navigation template for modified cortical bone trajectory screw placement in the lumbar spine. Front Surg 9:1028276. https://doi.org/10.3389/fsurg.2022.1028276
Zhang L, Tian N, Yang J et al (2020) Risk of pedicle and spinous process violation during cortical bone trajectory screw placement in the lumbar spine. BMC Musculoskelet Disord 21:536. https://doi.org/10.1186/s12891-020-03535-4
Weiser L, Huber G, Sellenschloh K et al (2017) Insufficient stability of pedicle screws in osteoporotic vertebrae: biomechanical correlation of bone mineral density and pedicle screw fixation strength. Eur Spine J 26:2891–2897. https://doi.org/10.1007/s00586-017-5091-x
Liu L, Zhang S, Liu G et al (2019) Early clinical outcome of lumbar spinal fixation with cortical bone trajectory pedicle screws in patients with osteoporosis with degenerative disease. Orthopedics 42:e465–e471. https://doi.org/10.3928/01477447-20190604-01
Ding H, Hai Y, Liu Y et al (2022) Cortical trajectory fixation versus traditional pedicle-screw fixation in the treatment of lumbar degenerative patients with osteoporosis: a prospective randomized controlled trial. Clin Interv Aging 17:175–184. https://doi.org/10.2147/CIA.S349533
Shi W, Aierken G, Wang S et al (2021) Application study of three-dimensional printed navigation template between traditional and novel cortical bone trajectory on osteoporosis lumbar spine. J Clin Neurosci 85:41–48. https://doi.org/10.1016/j.jocn.2020.11.038
Di Perna G, Marengo N, Matsukawa K et al (2023) Three-dimensional patient-matched template guides are able to increase mean diameter and length and to improve accuracy of cortical bone trajectory screws: a 5-year international experience. World Neurosurg 170:e542–e549. https://doi.org/10.1016/j.wneu.2022.11.066
Agyei JO, Khan A, Jowdy PK et al (2020) Robot-assisted cortical bone trajectory insertion of pedicle screws: 2-dimensional operative video. Oper Neurosurg (Hagerstown) 18:E171. https://doi.org/10.1093/ons/opz216
Buza JA 3rd, Good CR, Lehman RA Jr et al (2021) Robotic-assisted cortical bone trajectory (CBT) screws using the Mazor X Stealth (MXSE) system Edition: workflow and technical tips for safe and efficient use. J Robot Surg 15:13–23
Nagata K, Glassman SD, Brown ME et al (2023) Risk factors of screw malposition in robot-assisted cortical bone trajectory: analysis of 1344 consecutive screws in 256 patients. Spine (Phila Pa 1976). https://doi.org/10.1097/BRS.0000000000004827
Jia L, Yu Y, Khan K et al (2018) Superior facet joint violations during single level minimally invasive transforaminal lumbar interbody fusion: a preliminary retrospective clinical study. Biomed Res Int 2018:6152769. https://doi.org/10.1155/2018/6152769
Zhou LP, Zhang RJ, Li HM et al (2020) Comparison of cranial facet joint violation rate and four other clinical indexes between robot-assisted and freehand pedicle screw placement in spine surgery: a meta-analysis. Spine (Phila Pa 1976) 45:E1532–E1540
Le XF, Shi Z, Wang QL et al (2020) Rate and risk factors of superior facet joint violation during cortical bone trajectory screw placement: a comparison of robot-assisted approach with a conventional technique. Orthop Surg 12:133–140. https://doi.org/10.1111/os.12598
Rho K, OConnor TE, Lucas JM et al (2021) Minimally invasive robot-guided dual cortical bone trajectory for adjacent segment disease. Cureus 13:e16822
Acknowledgements
The authors acknowledged the Department of Statistics, Public Health College of Jilin University for their suggestions concerning the statistical analysis in this manuscript.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Q-SZ and Y-HZ designed the study protocol. XW and H-XL managed the literature search and data acquisition. XW and Y-HZ performed the statistical analysis and drafted the manuscript. Q-SZ provided critical revisions to the manuscript. Q-SZ resolved ambiguities during the study and gave final approval of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Informed consent
This article does not contain any studies with human or animal subjects performed by any authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, X., Li, Hx., Zhu, Qs. et al. Effectiveness and safety of robot-assisted versus fluoroscopy-assisted cortical bone trajectory screw instrumentation in spinal surgery: a systematic review and meta-analysis. J Robotic Surg 18, 78 (2024). https://doi.org/10.1007/s11701-024-01866-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11701-024-01866-1