Abstract
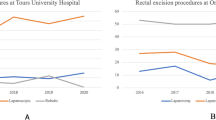
The number of robotic-assisted procedures for rectal cancer is rising. The risk of this procedure when performed by surgeon with limited robotic experience is unknown and the precise duration of the learning curve debated. We, therefore, aimed to analyze the learning curve and its related safety in a single center before the development of mentoring programs. We prospectively recorded all robotic procedures performed for colorectal cancer between 2015 and 2020 by a single surgeon. Operative times for partial and total proctectomy were analyzed. The learning curve was defined by comparison with the standard duration of the laparoscopic procedure performed in expert centers (published in GRECCAR 5 and GRECCAR 6 trials) and calculated using a cumulative summation for learning curve test (LC-CUSUM). Among the 174 patients operated for colorectal cancer, we analyzed the outcomes of the 89 patients operated by partial and total robotic proctectomy. To reach repeatedly the same surgical duration as laparoscopic procedure for partial or complete proctectomy, the LC-CUSUM identified a learning curve of 57 patients. A severe morbidity in this population, defined by Clavien–Dindo classification ≥ 3, was observed in 15 cases (16.8%) with an anastomotic leak rate of 13.5%. The rate of completeness of mesorectal excision was 90% and the mean number of harvested lymph nodes was 15 (± 9). Using operative time as end-point, the learning curve of rectal cancer robotic surgery identified a cut-off of 57 patients. The technic remained safe with acceptable morbidity and oncological outcomes.


Similar content being viewed by others
Data availability
Data supporting the findings of this study are available from the corresponding author on request.
References
Hazebroek EJ, Color Study Group (2002) COLOR: a randomized clinical trial comparing laparoscopic and open resection for colon cancer. Surg Endosc 16:949–953. https://doi.org/10.1007/s00464-001-8165-z. (PMID: 12163961)
Green BL, Marshall HC, Collinson F, Quirke P, Guillou P, Jayne DG, Brown JM (2013) Long-term follow-up of the Medical Research Council CLASICC trial of conventional versus laparoscopically assisted resection in colorectal cancer. Br J Surg 100:75–82. https://doi.org/10.1002/bjs.8945. (PMID: 23132548)
Jayne DG, Brown JM, Thorpe H, Walker J, Quirke P, Guillou PJ (2005) Bladder and sexual function following resection for rectal cancer in a randomized clinical trial of laparoscopic versus open technique. Br J Surg 92:1124–1132. https://doi.org/10.1002/bjs.4989. (PMID: 15997446)
Stevenson ARL, Solomon MJ, Lumley JW, Hewett P, Clouston AD, Gebski VJ, Davies L, Wilson K, Hague W, Simes J, ALaCaRT Investigators (2015) Effect of laparoscopic-assisted resection vs open resection on pathological outcomes in rectal cancer: the ALaCaRT randomized clinical trial. JAMA 314:1356–1363. https://doi.org/10.1001/jama.2015.12009. (PMID: 26441180)
Fleshman J, Branda M, Sargent DJ, Boller AM, George V, Abbas M, Peters WR, Maun D, Chang G, Herline A, Fichera A, Mutch M, Wexner S, Whiteford M, Marks J, Birnbaum E, Margolin D, Larson D, Marcello P, Posner M, Read T, Monson J, Wren SM, Pisters PWT, Nelson H (2015) Effect of laparoscopic-assisted resection vs open resection of stage II or III rectal cancer on pathologic outcomes: the ACOSOG Z6051 randomized clinical trial. JAMA 314:1346–1355. https://doi.org/10.1001/jama.2015.10529. (PMID: 26441179)
Corcione F, Esposito C, Cuccurullo D, Settembre A, Miranda N, Amato F, Pirozzi F, Caiazzo P (2005) Advantages and limits of robot-assisted laparoscopic surgery: preliminary experience. Surg Endosc 19:117–119. https://doi.org/10.1007/s00464-004-9004-9. (PMID: 15549629)
Delaney CP, Lynch AC, Senagore AJ, Fazio VW (2003) Comparison of robotically performed and traditional laparoscopic colorectal surgery. Dis Colon Rectum 46:1633–1639. https://doi.org/10.1007/bf02660768. (PMID: 14668588)
Mirnezami AH, Mirnezami R, Venkatasubramaniam AK, Chandrakumaran K, Cecil TD, Moran BJ (2010) Robotic colorectal surgery: hype or new hope? A systematic review of robotics in colorectal surgery. Colorectal Dis 12:1084–1093. https://doi.org/10.1111/j.1463-1318.2009.01999.x. (PMID: 19594601)
Miskovic D, Ahmed J, Bissett-Amess R, Gómez Ruiz M, Luca F, Jayne D, Figueiredo N, Heald RJ, Spinoglio G, Parvaiz A, European Academy for Robotic Colorectal Surgery (EARCS) (2019) European consensus on the standardization of robotic total mesorectal excision for rectal cancer. Colorectal Dis 21:270–276. https://doi.org/10.1111/codi.14502. (PMID: 30489676)
f714568d-e0e8–4021-bae3–2a922a10a9a3.pdf. Available from: https://isrg.gcs-web.com/static-files/f714568d-e0e8-4021-bae3-2a922a10a9a3
Feng Q, Yuan W, Li T, REAL Study Group et al (2022) Robotic versus laparoscopic surgery for middle and low rectal cancer (REAL): short-term outcomes of a multicentre randomised controlled trial. Lancet Gastroenterol Hepatol. 7(11):991–1004
Jayne D, Pigazzi A, Marshall H, Croft J, Corrigan N, Copeland J, Quirke P, West N, Rautio T, Thomassen N, Tilney H, Gudgeon M, Bianchi PP, Edlin R, Hulme C, Brown J (2017) Effect of robotic-assisted vs conventional laparoscopic surgery on risk of conversion to open laparotomy among patients undergoing resection for rectal cancer: the ROLARR randomized clinical trial. JAMA 318:1569–1580. https://doi.org/10.1001/jama.2017.7219. (PMID: 29067426)
Jayne D, Pigazzi A, Marshall H, Croft J, Corrigan N, Copeland J, Quirke P, West N, Edlin R, Hulme C, Brown J. Robotic-assisted surgery compared with laparoscopic resection surgery for rectal cancer: the ROLARR RCT. Southampton (UK): NIHR Journals Library Available from: http://www.ncbi.nlm.nih.gov/books/NBK547017/
Rouanet P, Gourgou S, Gogenur I, Jayne D, Ulrich A, Rautio T, Spinoglio G, Bouazza N, Moussion A, Gomez RM (2019) Rectal surgery evaluation trial: protocol for a parallel cohort trial of outcomes using surgical techniques for total mesorectal excision with low anterior resection in high-risk rectal cancer patients. Colorectal Dis 21:516–522. https://doi.org/10.1111/codi.14581. (PMID: 30740878)
Corrigan N, Marshall H, Croft J, Copeland J, Jayne D, Brown J (2018) Exploring and adjusting for potential learning effects in ROLARR: a randomised controlled trial comparing robotic-assisted vs standard laparoscopic surgery for rectal cancer resection. Trials 19:339. https://doi.org/10.1186/s13063-018-2726-0. (PMID: 29945673)
Odermatt M, Ahmed J, Panteleimonitis S, Khan J, Parvaiz A (2017) Prior experience in laparoscopic rectal surgery can minimise the learning curve for robotic rectal resections: a cumulative sum analysis. Surg Endosc 31:4067–4076. https://doi.org/10.1007/s00464-017-5453-9. (PMID: 28271267)
Hanly EJ, Marohn MR, Bachman SL, Talamini MA, Hacker SO, Howard RS, Schenkman NS (2004) Multiservice laparoscopic surgical training using the daVinci surgical system. Am J Surg 187:309–315. https://doi.org/10.1016/j.amjsurg.2003.11.021. (PMID: 14769327)
Guend H, Widmar M, Patel S, Nash GM, Paty PB, Guillem JG, Temple LK, Garcia-Aguilar J, Weiser MR (2017) Developing a robotic colorectal cancer surgery program: understanding institutional and individual learning curves. Surg Endosc 31:2820–2828. https://doi.org/10.1007/s00464-016-5292-0. (PMID: 27815742)
Soomro NA, Hashimoto DA, Porteous AJ, Ridley CJA, Marsh WJ, Ditto R, Roy S (2020) Systematic review of learning curves in robot-assisted surgery. BJS Open 4:27–44. https://doi.org/10.1002/bjs5.50235. (PMID: 32011823)
Pernar LIM, Robertson FC, Tavakkoli A, Sheu EG, Brooks DC, Smink DS (2017) An appraisal of the learning curve in robotic general surgery. Surg Endosc 31:4583–4596. https://doi.org/10.1007/s00464-017-5520-2. (PMID: 28411345)
Sng KK, Hara M, Shin J-W, Yoo B-E, Yang K-S, Kim S-H (2013) The multiphasic learning curve for robot-assisted rectal surgery. Surg Endosc 27:3297–3307. https://doi.org/10.1007/s00464-013-2909-4. (PMID: 23508818)
Harrysson IJ, Cook J, Sirimanna P, Feldman LS, Darzi A, Aggarwal R (2014) Systematic review of learning curves for minimally invasive abdominal surgery: a review of the methodology of data collection, depiction of outcomes, and statistical analysis. Ann Surg 260:37–45. https://doi.org/10.1097/SLA.0000000000000596. (PMID: 24670849)
Hopper AN, Jamison MH, Lewis WG (2007) Learning curves in surgical practice. Postgrad Med J 83:777–779. https://doi.org/10.1136/pgmj.2007.057190. (PMID: 18057179)
Biau DJ, Williams SM, Schlup MM, Nizard RS, Porcher R (2008) Quantitative and individualized assessment of the learning curve using LC-CUSUM. Br J Surg 95:925–929. https://doi.org/10.1002/bjs.6056. (PMID: 18498126)
Biau DJ, Porcher R (2010) A method for monitoring a process from an out of control to an in control state: application to the learning curve. Stat Med 29:1900–1909. https://doi.org/10.1002/sim.3947. (PMID: 20680983)
Bokhari MB, Patel CB, Ramos-Valadez DI, Ragupathi M, Haas EM (2011) Learning curve for robotic-assisted laparoscopic colorectal surgery. Surg Endosc 25:855–860. https://doi.org/10.1007/s00464-010-1281-x. (PMID: 20734081)
Barrie J, Jayne DG, Wright J, Murray CJC, Collinson FJ, Pavitt SH (2014) Attaining surgical competency and its implications in surgical clinical trial design: a systematic review of the learning curve in laparoscopic and robot-assisted laparoscopic colorectal cancer surgery. Ann Surg Oncol 21:829–840. https://doi.org/10.1245/s10434-013-3348-0. (PMID: 24217787)
Jiménez-Rodríguez RM, Rubio-Dorado-Manzanares M, Díaz-Pavón JM, Reyes-Díaz ML, Vazquez-Monchul JM, Garcia-Cabrera AM, Padillo J, De la Portilla F (2016) Learning curve in robotic rectal cancer surgery: current state of affairs. Int J Colorectal Dis 31:1807–1815. https://doi.org/10.1007/s00384-016-2660-0. (PMID: 27714517)
Herron DM, Marohn M, SAGES-MIRA Robotic Surgery Consensus Group (2008) A consensus document on robotic surgery. Surg Endosc 22:313–325. https://doi.org/10.1007/s00464-007-9727-5. (discussion 311–312; PMID: 18163170)
Denost Q, Rouanet P, Faucheron J-L, Panis Y, Meunier B, Cotte E, Meurette G, Kirzin S, Sabbagh C, Loriau J, Benoist S, Mariette C, Sielezneff I, Lelong B, Mauvais F, Romain B, Barussaud M-L, Germain C, Picat M-Q, Rullier E, Laurent C (2017) To drain or not to drain infraperitoneal anastomosis after rectal excision for cancer: the GRECCAR 5 randomized trial. Ann Surg 265:474–480. https://doi.org/10.1097/SLA.0000000000001991. (PMID: 27631776)
Lefèvre J, Mineur L, Cachanado M, Denost Q, Rouanet P, de Chaisemartin C, Meunier B, Mehrdad J, Cotte E, Desrame J, Karoui M, Benoist S, Kirzin S, Berger A, Panis Y, Piessen G, Saudemont A, Prudhomme M, Peschaud F, Dubois A, Loriau J, Tuech J-J, Meurette G, Lupinacci R, Goasguen N, Creavin B, Simon T, Parc Y (2019) Does a longer waiting period after neoadjuvant radio-chemotherapy improve the oncological prognosis of rectal cancer? Three years’ follow-up results of the Greccar-6 randomized multicenter trial. Ann Surg 270:747–754. https://doi.org/10.1097/SLA.0000000000003530. (PMID: 31634178)
Dindo D, Demartines N, Clavien P-A (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213. https://doi.org/10.1097/01.sla.0000133083.54934.ae. (PMID: 15273542)
Kim HJ, Choi G-S, Park JS, Park SY (2014) Multidimensional analysis of the learning curve for robotic total mesorectal excision for rectal cancer: lessons from a single surgeon’s experience. Dis Colon Rectum 57:1066–1074. https://doi.org/10.1097/DCR.0000000000000174. (PMID: 25101602)
Gerbaud F, Valverde A, Danoussou D, Goasguen N, Oberlin O, Lupinacci RM (2019) Experience with transitioning from laparoscopic to robotic right colectomy. JSLS. https://doi.org/10.4293/JSLS.2019.00044. (PMID: 31787837)
Kowalewski K-F, Schmidt MW, Proctor T, Pohl M, Wennberg E, Karadza E, Romero P, Kenngott HG, Müller-Stich BP, Nickel F (2018) Skills in minimally invasive and open surgery show limited transferability to robotic surgery: results from a prospective study. Surg Endosc 32:1656–1667. https://doi.org/10.1007/s00464-018-6109-0. (PMID: 29435749)
Sian TS, Tierney GM, Park H, Lund JN, Speake WJ, Hurst NG, Al Chalabi H, Smith KJ, Tou S (2018) Robotic colorectal surgery: previous laparoscopic colorectal experience is not essential. J Robot Surg 12:271–275. https://doi.org/10.1007/s11701-017-0728-7. (PMID: 28721636)
Vanlander AE, Mazzone E, Collins JW, Mottrie AM, Rogiers XM, van der Poel HG, Van Herzeele I, Satava RM, Gallagher AG (2020) Orsi consensus meeting on european robotic training (OCERT): results from the first multispecialty consensus meeting on training in robot-assisted surgery. Eur Urol. https://doi.org/10.1016/j.eururo.2020.02.003. (e-pub ahead of print 20 February 2020)
Hanna GB, Mackenzie H, Miskovic D, Ni M, Wyles S, Aylin P, Parvaiz A, Cecil T, Gudgeon A, Griffith J, Robinson JM, Selvasekar C, Rockall T, Acheson A, Maxwell-Armstrong C, Jenkins JT, Horgan A, Cunningham C, Lindsey I, Arulampalam T, Motson RW, Francis NK, Kennedy RH, Coleman MG, on behalf of Lapco program (2022) Laparoscopic colorectal surgery outcomes improved after national training program (LAPCO) for specialists in England. Ann Surg 275(6):1149–1155. https://doi.org/10.1097/SLA.0000000000004584
Symer M, Sedrakyan A, Yeo H (2019) Case sequence analysis of the robotic colorectal resection learning curve. Dis Colon Rectum 62:1071–1078. https://doi.org/10.1097/DCR.0000000000001437. (PMID: 31318771)
Chen W, Sailhamer E, Berger DL, Rattner DW (2007) Operative time is a poor surrogate for the learning curve in laparoscopic colorectal surgery. Surg Endosc 21:238–243. https://doi.org/10.1007/s00464-006-0120-6. (PMID: 17180745)
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis were performed by SZ, RM and LM. The first draft of the manuscript was written by SZ and LM and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. SZ: methodology, project administration, original draft; writing- review & editing, data curation. IS: validation; resources; original draft editing. RM: validation; data curation; resources. EF-d-S: validation; data curation; resources. CH: validation; resources; original draft editing. MG: validation; resources; original draft editing. MF: validation; conceptualization; visualization; original draft writing; original draft editing. LM: methodology; conceptualization; data curation; project administration; supervision; original draft; writing, review & editing.
Corresponding author
Ethics declarations
Conflict of interest
Leonor Benhaim is a trainer for Intuitive Surgical®; other authors have no conflict of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zaepfel, S., Marcovei, R., Fernandez-de-Sevilla, E. et al. Robotic-assisted surgery for mid and low rectal cancer: a long but safe learning curve. J Robotic Surg 17, 2099–2108 (2023). https://doi.org/10.1007/s11701-023-01624-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11701-023-01624-9